Abstract
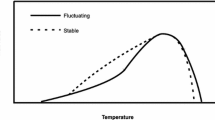
Environmental variability is expected to be important in shaping performance curves, reaction norms of phenotypic traits related to fitness. Models predict that the breadth of performance curves should increase with environmental variability at the expense of maximal performance. In this study, we compared the thermal performance curves of two sympatric populations of the parasitoid Venturia canescens that were observed under contrasting thermal regimes in their respective preferred habitats and differing in their modes of reproduction. Our results confirm the large effect of developmental temperature on phenotypic traits of insects and demonstrate that thelytokous and arrhenotokous wasps respond differently to temperature during development, in agreement with model predictions. For traits related to fecundity, thelytokous parasitoids, which usually occur in stable thermal conditions, exhibit specialist performance curves, maximising their reproductive success under a restricted range of temperature. In contrast, arrhenotokous parasitoids, which occur in variable climates, exhibit generalist performance curves, in keeping with the hypothesis "jack of all temperatures, master of none".



Similar content being viewed by others
References
Ahmad T (1936) The influence of ecological factors on the Mediterranean flour moth, Ephestia kuehniella, and its parasite, Nemeritis canescens. J Anim Ecol 5:67–93
Amat I (2004) Coexistence de la reproduction sexuée et asexuée chez l’hyménoptère parasitoïde Venturia canescens: aspects comportementaux et écologiques. Dissertation, Université Claude Bernard Lyon 1
Amat I, Castelo M, Desouhant E, Bernstein C (2006) The influence of temperature and host availability on the host exploitation strategies of sexual and asexual parasitic wasps of the same species. Oecologia 148:153–161
Amat I, Desouhant E, Bernstein C (2009) Differential use of conspecific-derived information by sexual and asexual parasitic wasps exploiting partially depleted host patches. Behav Ecol Sociobiol 63:563–572
Angilletta MJ (2009) Thermal adaptation: a theoretical and empirical synthesis. University Press, Oxford
Atkinson D (1994) Temperature and organism size—a biological law for ectotherms? Adv Ecol Res 3:1–58
Barke J, Mateo Leach I, Beukeboom LW (2005) Fitness of arrhenotokous and thelytokous Venturia canescens. Proc Exp Appl Entomol Neth 16:27–35
Beukeboom LW, Driessen G, Luckerhoff L, Bernstein C, Lapchin L, Van Alphen JJM (1999) Distribution and relatedness of sexual and asexual Venturia canescens (Hymenoptera). Proc Exper Appl Entomol 10:23–28
Carrière Y, Boivin G (2001) Constraints on the evolution of thermal sensitivity of foraging in Trichogramma: genetic trade-off and plasticity in maternal selection. Am Nat 157:570–581
Casas J, Nisbet RM, Swarbrick S, Murdoch WW (2000) Eggload dynamics and oviposition rate in a wild population of a parasitic wasp. J Anim Ecol 69:185–193
Castelo MK, Corley JC, Desouhant E (2003) Conspecific avoidance during foraging in Venturia canescens (Hymenoptera: Ichneumonidae): the roles of host presence and conspecific densities. J Insect Behav 16:307–318
Chesson P (2000) Mechanisms of maintenance of species diversity. Annu Rev Ecol Syst 31:343–366
Chevin LM, Lande R, Mace GM (2010) Adaptation, plasticity, and extinction in a changing environment: towards a predictive theory. PLoS Biol 8:e1000357
Chown SL, Hoffmann AA, Kristensen TN, Anguilletta MJ, Nils CS, Pertoldi C (2010) Adapting to climate change: a perspective from evolutionary physiology. Clim Res 43:3–15
David JR, Moreteau B, Gauthier JP, Pétavy G, Stockel A, Imasheva AG (1994) Reaction norms of size characters in relation to growth temperature in Drosophila melanogaster: an isofemale lines analysis. Genet Select Evol 26:229–251
Davidowitz G, Nijhout H (2004) The physiological basis of reaction norms: the interaction among growth rate, the duration of growth and body size. Integr Comp Biol 44:443–449
Desouhant E, Driessen G, Amat I, Bernstein C (2005) Host and food searching in a parasitic wasp Venturia canescens: a trade-off between current and future reproduction? Anim Behav 70:145–152
Eliopoulos PA, Harvey JA, Athanassiou G, Stathas GJ (2003) Effect of biotic and abiotic factors on reproductive parameters of the synovigenic endoparasitoid Venturia canescens. Physiol Entomol 28:268–275
Ernande B, Dieckmann U (2004) The evolution of phenotypic plasticity in spatially structured environments: implications of intraspecific competition, plasticity costs and environmental characteristics. J Evol Biol 17:613–628
Gilchrist GW (1995) Specialists and generalists in changing environments. I. Fitness landscapes of thermal sensitivity. Am Nat 146:252–270
Godfray HCJ (1994) Parasitoids: behavioral and evolutionary ecology. Princeton University Press, Princeton
Griffith TM, Sultan SE (2006) Plastic and constant developmental traits contribute to adaptive differences in co-occurring Polygonum species. Oikos 114:5–14
Harvey JA, Vet LEM (1997) Venturia canescens parasitizing Galleria mellonella and Anagasta kuehniella: differing suitability of two hosts with highly variable growth potential. Entomol Exp Appl 84:93–100
Harvey JA, Harvey IF, Thompson DJ (1994) Flexible larval growth allows use of a range of host sizes by a parasitoid wasp. Ecology 75:1420–1428
Harvey JA, Harvey IF, Thompson DJ (2001) Lifetime reproductive success in the solitary endoparasitoid, Venturia canescens. J Insect Behav 14:573–593
Hochachka PW, Somero GN (2002) Biochemical adaptation: mechanism and process in physiological evolution. Oxford University Press, Oxford
Honek A (1993) Intraspecific variation in body size and fecundity in insects: a general relationship. Oikos 66:483–492
Huey RB, Hertz PE (1984) Is a jack-of-all-temperatures a master of none? Evolution 38:441–444
Huey RB, Kingsolver JG (1989) Evolution of thermal sensitivity of ectotherm performance. Trends Ecol Evol 4:131–135
Huey RB, Stevenson R (1979) Integrating thermal physiology and ecology of ectotherms: a discussion of approaches. Amer Zool 19:357–366
Jervis MA, Kidd NAC (1986) Host-feeding strategies in hymenopteran parasitoids. Biol Rev 61:395–434
Jervis MA, Heimpel GE, Ferns PN, Harvey JA, Kidd NAC (2001) Life-history strategies in parasitoid wasps: a comparative analysis of ‘ovigeny’. J Anim Ecol 70:442–458
Karan D, Morin JP, Gravot E, Moreteau B, David JR (1999) Body size reaction norms in Drosophila melanogaster: temporal stability and genetic architecture in a natural population. Genet Select Evol 31:491–508
Kazmer DJ, Luck RF (1995) Field tests of the size-fitness hypothesis in the egg parasitoid Trichogramma pretiosum. Ecology 76:412–425
Le Ralec A (1995) Egg contents in relation to host-feeding in some parasitic hymenoptera. Biocontrol 40:87–93
Li D, Zhao Z, Roberts H, Schneider MV, Theopold U, Schmidt O (2003) Genetic analysis of two distinct reproductive strategies in sexual and asexual field populations of an endoparasitic wasp, Venturia canescens. Heredity 90:291–297
Liang KY, Zeger SL (1986) Longitudinal data analysis using generalized linear models. Biometrika 73:13–22
Liefting M, Ellers J (2008) Habitat-specific differences in thermal plasticity in natural populations of a soil arthropod. Biol J Linnean Soc 94:265–271
Liefting M, Hoffmann AA, Ellers J (2009) Plasticity versus environmental canalization: population differences in thermal responses along a latitudinal gradient in Drosophila serrata. Evolution 63:1954–1963
Lind MI, Johansson F (2007) The degree of adaptive phenotypic plasticity is correlated with the spatial environmental heterogeneity experienced by island populations of Rana temporaria. J Evol Biol 20:1288–1297
Liu YQ, Thiel A, Hoffmeister TS (2009a) Odor-mediated patch choice in the parasitoid Venturia canescens: temporal decision dynamics. Entomol Exp Appl 132:110–117
Liu YQ, Bernstein C, Thiel A (2009b) Travel duration, energetic expenditure, and patch exploitation in the parasitic wasp Venturia canescens. Behav Ecol Sociobiol 63:1459–1469
Lucchetta P, Desouhant E, Wajnberg E, Bernstein C (2007) Small but smart: the interaction between environmental cues and internal state modulates host-patch exploitation in a parasitic wasp. Behav Ecol Sociobiol 61:1409–1418
Lynch M, Gabriel W (1987) Environmental tolerance. Am Nat 129:283–303
Mateo Leach I, Hesseling A, Huibers WHC, Witsenboer H, Beukeboom LW, Van De Zande L (2009) Transcriptome and proteome analysis of ovaries of arrhenotokous and thelytokous Venturia canescens. Insect Mol Biol 18:477–482
Metzger M, Bernstein C, Desouhant E (2008) Does constrained oviposition influence offspring sex ratio in the solitary parasitoid wasp Venturia canescens ? Ecol Entomol 33:167–174
Moran NA (1992) The evolutionary maintenance of alternative phenotypes. Am Nat 139:971–989
Nelder JA, Weddenburn RWM (1972) Generalized linear models. J Roy Stat Soc A Sta 135:370–384
Pelosse P, Bernstein C, Desouhant E (2007) Differential energy allocation as an adaptation to different habitats in the parasitic wasp Venturia canescens. Evol Ecol 21:669–685
Pelosse P, Amat I, Bernstein C, Desouhant E (2010) The dynamics of energy allocation in adult arrhenotokous and thelytokous Venturia canescens. Entomol Exp Appl 135:68–76
Pigliucci M (1996) How organisms respond to environmental changes: from phenotypes to molecules (and vice versa). Trends Ecol Evol 11:168–173
R Development Core Team (2010) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna
Richard R, Casas J (2009) Stochasticity and controllability of nutrient sources in foraging: host-feeding and egg resorption in parasitoids. Ecol Monogr 79:465–483
Richards CL, Bossdorf O, Muth NZ, Gurevitch J, Pigliucci M (2006) Jack of all trades, master of some? On the role of phenotypic plasticity in plant invasions. Ecol Lett 9:981–993
Roff DA (1992) The evolution of life histories: theory and analysis. Chapman & Hall, New York
Rosenheim JA, Heimpel GE, Mangel M (2000) Egg maturation, egg resorption and the costliness of transient egg limitation in insects. Proc R Soc B-Biol Sci 267:1565–1573
Salt G (1976) The hosts of Nemeritis canescens, a problem in the host specificity of insect parasitoids. Ecol Entomol 1:63–67
Schneider MV, Beukeboom LW, Driessen G, Lapchin L, Bernstein C, Van Alphen JJM (2002) Geographical distribution and genetic relatedness of sympatrical thelytokous and arrhenotokous populations of the parasitoid Venturia canescens (Hymenoptera). J Evol Biol 15:191–200
Sgrô CM, Partridge L (1999) A delayed wave of death from reproduction in Drosophila. Science 286:2521–2524
Sirot E, Bernstein C (1996) Time sharing between host searching and food searching in parasitoids: state-dependent optimal strategies. Behav Ecol 7:189–194
Sultan SE (2001) Phenotypic plasticity for fitness components in Polygonum species of contrasting ecological breadth. Ecology 82:328–343
Sultan SE, Wilczek AM, Bell DL, Hand G (1998) Physiological response to complex environments in annual Polygonum species of contrasting ecological breadth. Oecologia 115:564–578
Thiel A, Driessen G, Hoffmeister TS (2006) Different habitats, different habits? Response to foraging information in the parasitic wasp Venturia canescens. Behav Ecol Sociobiol 59:614–623
Van Tienderen PH (1991) Evolution of generalists and specialist in spatially heterogeneous environments. Evolution 45:1317–1331
Visser ME (1994) The importance of being large: the relationship between size and fitness in females of the parasitoid Aphaereta minuta (Hymenoptera: Braconidae). J Anim Ecol 6:963–978
Waage JK (1979) Foraging for patchily-distributed hosts by the parasitoid, Nemeritis canescens. J Anim Ecol 48:353–371
Wajnberg E, Bernstein C, Van Alphen JJM (2008) Behavioral ecology of insect parasitoids. Blackwell, Oxford
Wilson RS (2001) Geographic variation in thermal sensitivity of jumping performance in the frog Limnodynastes peronii. J Exp Biol 204:4227–4236
Woltereck R (1909) Weitere experimentelle Untersuchungen über Artveränderung, speziell über das Wesen quantitativer Artunter- schiede bei Daphnien. Verh Deutsch Zool Ges 19:110–173
Acknowledgements
We are grateful to François Débias and Sandrine Sauzet for his help in rearing of V. canescens. We would like to thank Isabelle Amat, Jacintha Ellers, Jacques Van Alphen and Berteau Florence for their comments on earlier drafts of the manuscript, and two anonymous reviewers for their comments on the manuscript. Research was supported by the French Ministry.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Communicated by: Sven Thatje
Rights and permissions
About this article
Cite this article
Foray, V., Gibert, P. & Desouhant, E. Differential thermal performance curves in response to different habitats in the parasitoid Venturia canescens . Naturwissenschaften 98, 683–691 (2011). https://doi.org/10.1007/s00114-011-0818-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-011-0818-8