Abstract
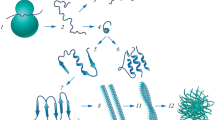
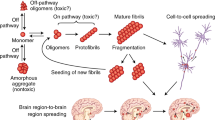
Several lines of evidence suggest that a defective protein folding is a central event in both Alzheimer’s and prion disease. Although the two disorders are very different clinically, neuropathologically, and biochemically, the molecular event that may trigger the disease process appears to be the same: the formation of an altered protein conformer composed of a high content of β-sheet structure. Compounds with the ability to prevent and to reverse protein conformational changes may be useful as novel therapeutic approaches for Alzheimer’s disease, prion disease, and other disorders of defective protein folding.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 28 December 1998 / Accepted: 8 February 1999
Rights and permissions
About this article
Cite this article
Soto, C. Alzheimer’s and prion disease as disorders of protein conformation: implications for the design of novel therapeutic approaches. J Mol Med 77, 412–418 (1999). https://doi.org/10.1007/s001090050371
Issue Date:
DOI: https://doi.org/10.1007/s001090050371