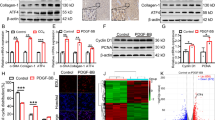
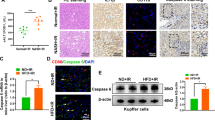
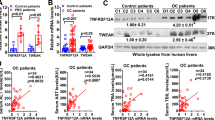
Abstract
The histidine-rich calcium-binding protein (HRC) is a regulator of Ca2 + homeostasis and it plays a significant role in liver fibrosis. Pyroptosis, a specific inflammatory cell death, can lead to hepatic stellate cells (HSCs) activation and liver fibrosis. However, the role of HRC in pyroptosis has not been explored. In this study, we demonstrated that HRC, mainly located in the hepatocyte, was over expressed in fibrotic liver tissues. We further found that enforced expression of HRC in hepatocytes induced pyroptosis and HMGB1 release, and subsequently led to HSCs activation by NLRP3/caspase-1 pathway. In addition, the proliferation and migration of HSCs were also enhanced by HRC overexpression in hepatocytes. Furthermore, NLRP3 inhibitor MCC950 and caspase-1 inhibitor VX-765 alleviated hepatic HRC-mediated hepatocytes pyroptosis and HSCs activation. This study demonstrated that hepatic HRC promoted HSCs activation by inducing hepatocyte pyroptosis, which suggests that HRC may be a promising therapeutic target to prevent liver fibrosis.







Similar content being viewed by others
Data availability
The data used to support the findings of this study are available from the corresponding author upon request.
Abbreviations
- HRC:
-
Histidine-rich calcium-binding protein
- α-SMA:
-
α-Smooth actin
- CCl4:
-
Carbon tetrachloride
- ALT:
-
Aminotransferase
- AST:
-
Aspartic aminotransferase
- LDH:
-
Lactate dehydrogenase
- H&E:
-
Hematoxylin-eosin
- IHC:
-
Immunohistochemistry
- GSDMD:
-
Gasdermin D
- GSDMD-N:
-
N-terminal fragment of GSDMD
- NLRP3:
-
Nod-like receptor protein 3
- IL-1β:
-
Interleukin-1β
- IL-18:
-
Interleukin-18
- HMGB1:
-
High mobility group box 1
- LPS:
-
Lipopolysaccharide
References
Asrani SK, Devarbhavi H, Eaton J et al (2019) Burden of liver diseases in the world. J Hepatol 70:151–171. https://doi.org/10.1016/j.jhep.2018.09.014
Seki E, Schwabe RF (2015) Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology 61:1066–1079. https://doi.org/10.1002/hep.27332
Marrone G, Shah V, Gracia-Sancho J H (2016) Sinusoidal communication in liver fibrosis and regeneration. J Hepatol 65:608–617. https://doi.org/10.1016/j.jhep.2016.04.018
Wree A, Mehal W, Feldstein ZAE (2016) Targeting Cell death and sterile inflammation loop for the treatment of nonalcoholic steatohepatitis. Semin Liver Dis 36:27–36. https://doi.org/10.1055/s-0035-1571272
Galluzzi L, Vitale I, Aaronson SA et al (2018) Molecular mechanisms of cell death: recommendations of the Nomenclature committee on cell death 2018. Cell Death Differ 25:486–541. https://doi.org/10.1038/s41418-017-0012-4
Broz P, Pelegrin P, Shao F (2020) The gasdermins, a protein family executing cell death and inflammation. Nat Rev Immunol 20:143–157. https://doi.org/10.1038/s41577-019-0228-2
Zhang K, Shi Z, Zhang M et al (2020) Silencing lncRNA Lfar1 alleviates the classical activation and pyoptosis of macrophage in hepatic fibrosis. Cell Death Dis 11:132. https://doi.org/10.1038/s41419-020-2323-5
Wree A, Eguchi A, McGeough MD et al (2014) NLRP3 inflammasome activation results in hepatocyte pyroptosis, liver inflammation, and fibrosis in mice. Hepatology 59:898–910. https://doi.org/10.1002/hep.26592
Kong DL, Kong FY, Liu XY et al (2019) Soluble egg antigen of Schistosoma japonicum induces pyroptosis in hepatic stellate cells by modulating ROS production. Parasit Vectors 12:475. https://doi.org/10.1186/s13071-019-3729-8
Palacios-Macapagal D, Connor J, Mustelin T et al (2017) Cutting edge: eosinophils undergo caspase-1-mediated pyroptosis in response to necrotic liver cells. J Immunol 199:847–853. https://doi.org/10.4049/jimmunol.1601162
Lan P, Fan Y, Zhao Y et al (2017) TNF superfamily receptor OX40 triggers invariant NKT cell pyroptosis and liver injury. J Clin Invest 127:2222–2234. https://doi.org/10.1172/JCI91075
Arvanitis DA, Vafiadaki E, Sanoudou D et al (2011) Histidine-rich calcium binding protein: the new regulator of sarcoplasmic reticulum calcium cycling. J Mol Cell Cardiol 50:43–49. https://doi.org/10.1016/j.yjmcc.2010.08.021
Swanson KV, Deng M, Ting JP (2019) The NLRP3 inflammasome: molecular activation and regulation to therapeutics. Nat Rev Immunol 19:477–489. https://doi.org/10.1038/s41577-019-0165-0
Liu C, Hu T, Cai Z et al (2020) Nucleotide-binding oligomerization domain-like receptor 3 deficiency attenuated isoproterenol-induced cardiac fibrosis via reactive oxygen species/high mobility group box 1 protein axis. Front Cell Dev Biol 8:713. https://doi.org/10.3389/fcell.2020.00713
Gehrke N, Hovelmeyer N, Waisman A et al (2018) Hepatocyte-specific deletion of IL1-RI attenuates liver injury by blocking IL-1 driven autoinflammation. J Hepatol 68:986–995. https://doi.org/10.1016/j.jhep.2018.01.008
Geng Y, Ma Q, Liu YN et al (2015) Heatstroke induces liver injury via IL-1beta and HMGB1-induced pyroptosis. J Hepatol 63:622–633. https://doi.org/10.1016/j.jhep.2015.04.010
Ge X, Arriazu E, Magdaleno F et al (2018) High mobility group box-1 drives fibrosis progression signaling via the receptor for advanced glycation end products in mice. Hepatology 68:2380–2404. https://doi.org/10.1002/hep.30093
Liu J, Li M, Gong J et al (2017) Knockdown of histidine-rich calcium-binding protein (HRC) suppresses liver fibrosis by inhibiting the activation of hepatic stellate cells. Biol Open 6:29–34. https://doi.org/10.1242/bio.019828
Frank D, Vince JE (2019) Pyroptosis versus necroptosis: similarities, differences, and crosstalk. Cell Death Differ 26:99–114. https://doi.org/10.1038/s41418-018-0212-6
Volchuk A, Ye A, Chi L et al (2020) Indirect regulation of HMGB1 release by gasdermin D. Nat Commun 11:4561. https://doi.org/10.1038/s41467-020-18443-3
Odagiri N, Matsubara T, Sato-Matsubara M et al (2021) Anti-fibrotic treatments for chronic liver diseases: the present and the future. Clin Mol Hepatol 27:413–424. https://doi.org/10.3350/cmh.2020.0187
Cheng QC, Fan J, Deng XW et al (2020) Dihydromyricetin ameliorates chronic liver injury by reducing pyroptosis. World J Gastroenterol 26:6346–6360. https://doi.org/10.3748/wjg.v26.i41.6346
Zhang M, Zhu X, Tong H et al (2019) AVE 0991 attenuates pyroptosis and liver damage after heatstroke by inhibiting the ROS-NLRP3 inflammatory signalling pathway. Biomed Res Int 2019:1806234. https://doi.org/10.1155/2019/1806234
Zhao X, Wang C, Dai S et al (2022) Quercetin protects ethanol-induced hepatocyte pyroptosis via scavenging mitochondrial ROS and promoting PGC-1alpha-regulated mitochondrial homeostasis in L02 cells. Oxid Med Cell Longev 2022:4591134. https://doi.org/10.1155/2022/4591134
Cai X, Wang J, Wang J et al (2020) Intercellular crosstalk of hepatic stellate cells in liver fibrosis: new insights into therapy. Pharmacol Res 155:104720. https://doi.org/10.1016/j.phrs.2020.104720
Bourebaba N, Marycz K (2021) Hepatic stellate cells role in the course of metabolic disorders development - a molecular overview. Pharmacol Res 170
Kawashima R, Tamaki S, Kawakami F et al (2020) Histamine H2-receptor antagonists improve non-steroidal anti-inflammatory drug-induced intestinal dysbiosis. Int J Mol Sci 21. https://doi.org/10.3390/ijms21218166
Wu J, Lin S, Wan B et al (2019) Pyroptosis in liver disease: new insights into disease mechanisms. Aging Dis 10:1094–1108. https://doi.org/10.14336/AD.2019.0116.
Li W, Deng M, Loughran PA et al (2020) LPS induces active HMGB1 release from hepatocytes into exosomes through the coordinated activities of TLR4 and caspase-11/GSDMD signaling. Front Immunol 11:229. https://doi.org/10.3389/fimmu.2020.00229
Gaul S, Leszczynska A, Alegre F et al (2021) Hepatocyte pyroptosis and release of inflammasome particles induce stellate cell activation and liver fibrosis. J Hepatol 74:156–167. https://doi.org/10.1016/j.jhep.2020.07.041
Deng M, Tang Y, Li W et al (2018) The endotoxin delivery protein HMGB1 mediates caspase-11-dependent lethality in sepsis. Immunity 49(740–753):e7. https://doi.org/10.1016/j.immuni.2018.08.016
Li J, Zeng C, Zheng B et al (2018) HMGB1-induced autophagy facilitates hepatic stellate cells activation: a new pathway in liver fibrosis. Clin Sci (Lond) 132:1645–1667. https://doi.org/10.1042/CS20180177
He Q, Fu Y, Ding X et al (2018) High-mobility group box 1 induces endoplasmic reticulum stress and activates hepatic stellate cells. Lab Invest 98:1200–1210. https://doi.org/10.1038/s41374-018-0085-9
Man SM (2018) Inflammasomes in the gastrointestinal tract: infection, cancer and gut microbiota homeostasis. Nat Rev Gastroenterol Hepatol 15:721–737. https://doi.org/10.1038/s41575-018-0054-1
Mangan MSJ, Olhava EJ, Roush WR et al (2018) Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov 17:688. https://doi.org/10.1038/nrd.2018.149
Tang D, Wang H, Billiar TR et al (2021) Emerging mechanisms of immunocoagulation in sepsis and septic shock. Trends Immunol 42:508–522. https://doi.org/10.1016/j.it.2021.04.001
Guo Q, Wu Y, Hou Y et al (2018) Cytokine secretion and pyroptosis of thyroid follicular cells mediated by enhanced NLRP3, NLRP1, NLRC4, and AIM2 inflammasomes are associated with autoimmune thyroiditis. Front Immunol 9:1197. https://doi.org/10.3389/fimmu.2018.01197
Koh EH, Yoon JE, Ko MS et al (2020) Sphingomyelin synthase 1 mediates hepatocyte pyroptosis to trigger non-alcoholic steatohepatitis. Gut. https://doi.org/10.1136/gutjnl-2020-322509
Yu E, Zhang E, Lv X et al (2022) LDC7559 Exerts neuroprotective effects by inhibiting GSDMD-dependent pyroptosis of microglia in mice with traumatic brain injury. J Neurotrauma. https://doi.org/10.1089/neu.2021.0318
Sun J, Shi L, Xiao T et al (2021) MicroRNA-21, via the HIF-1alpha/VEGF signaling pathway, is involved in arsenite-induced hepatic fibrosis through aberrant cross-talk of hepatocytes and hepatic stellate cells. Chemosphere 266:129177. https://doi.org/10.1016/j.chemosphere.2020.129177
Funding
This study was funded by the National Natural Science Foundation of China (Nos. 81800547, 81672392).
Author information
Authors and Affiliations
Contributions
JM. Liu and DA. Tian designed the study. JW. Wu, SH, Xia and WD. Zhou performed the experiments. P. Han, K. Zhao and MY. Zhang analyzed the data. JW. Wu, JM. Liu and KXP prepared the manuscript. JM. Liu and JZ. Liao provide financial support. All authors contributed to the article and approved the submitted version.
Corresponding authors
Ethics declarations
Ethics approval
The animal study was reviewed and approved by The Institution Ethics Committee of Tongji Medical College, Huazhong University of Science and Technology. This study was performed according to the principles of the Declaration of Helsinki and was approved by the Ethics Committee of Tongji Hospital, Huazhong University of Science and Technology following the ethical and institutional guidelines.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Key message
• Histidine-rich calcium-binding protein (HRC) is overexpressed in fibrotic liver tissues.
• HRC triggers cell pyroptosis and HMGB1 release.
• HRC induces HSCs activation, proliferation and migration.
• HRC mediates hepatocyte pyroptosis and HSCs activation via NLRP3/caspase-1 pathway.
• HRC may be a promising therapeutic target to prevent liver fibrosis.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, J., Zhang, M., Xia, S. et al. Hepatic HRC induces hepatocyte pyroptosis and HSCs activation via NLRP3/caspase-1 pathway. J Mol Med 100, 1787–1799 (2022). https://doi.org/10.1007/s00109-022-02270-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-022-02270-8