Abstract
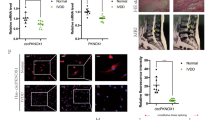
Accumulating evidence suggests that microRNAs (miRNAs) play an important role in intervertebral disc degeneration (IDD), but the precise role of specific miRNAs involved in this disease remains elusive. The purpose of this study was to identify IDD-specific miRNAs, followed by functional validation of results. MiRNA expression profile was determined in nucleus pulposus (NP) tissues from patients with IDD and controls, employing Solexa sequencing and quantitative real-time PCR (qRT-PCR). Biological functions of differential expression miRNAs were further investigated in vitro and in vivo. Luciferase reporter assays and Western blotting were performed to determine miRNA targets. We identified 28 miRNAs that were differentially expressed in patients compared with controls. Following qRT-PCR confirmation, miR-193a-3p was significantly down-regulated in degenerative NP tissues. Moreover, its level was correlated with grade of disc degeneration. Through gain- and loss-of-function studies, miR-193a-3p was demonstrated to significantly promote type II collagen expression in NP cells. Knockdown of MMP14 induced effects on NP cells similar to those induced by miR-193a-3p. Bioinformatics target prediction identified MMP14 as a putative target of miR-193a-3p. Furthermore, luciferase reporter assays and Western blotting demonstrated that miR-193a-3p directly targets MMP14. MiR-193a-3p inhibited IDD in vitro and in vivo. The downregulation of miR-193a-3p induces the expression of MMP14, which promotes loss of type II collagen and thereby contributes to the development of human IDD. Our findings extend the role of miR-193a-3p in the pathogenesis of IDD and provide a potential novel therapeutic target for degenerative disc disease.
Key messages
-
Intervertebral disc degeneration (ICC)-specific miRNA profile generated by next generation sequencing.
-
Downregulation of miR-193a-3p promoted loss of type II collagen by directly targeting MMP14 in IDD.
-
miR-193a-3p inhibited IDD in vitro and in vivo.
-
miR-193a-3p may be a promising candidate for prevention of degenerative disc disease.






Similar content being viewed by others
Change history
16 November 2023
A Correction to this paper has been published: https://doi.org/10.1007/s00109-023-02400-w
References
Williams FM, Popham M, Hart DJ, de Schepper E, Bierma-Zeinstra S et al (2011) GDF5 single-nucleotide polymorphism rs143383 is associated with lumbar disc degeneration in Northern European women. Arthritis Rheum 63:708–712
Samartzis D, Karppinen J, Chan D, Luk KD, Cheung KM (2012) The association of lumbar intervertebral disc degeneration on magnetic resonance imaging with body mass index in overweight and obese adults: a population-based study. Arthritis Rheum 64:1488–1496
Luo X, Pietrobon R, Sun SX, Liu GG, Hey L (2004) Estimates and patterns of direct health care expenditures among individuals with back pain in the United States. Spine 29:79–86
Borenstein D (1992) Epidemiology, etiology, diagnostic evaluation, and treatment of low back pain. Curr Opin Rheumatol 4:226–232
Risbud MV (2014) Role of cytokines in intervertebral disc degeneration: pain and disc content. Nat Rev Rheumatol 10:44–56
Battié MC, Ortega-Alonso A, Niemelainen R, Gill K, Levalahti E, Videman T (2014) Lumbar spinal stenosis is a highly genetic condition partly mediated by disc degeneration. Arthritis Rheum 66:3505–3510
Power KA, Grad S, Rutges JP, Creemers LB, van Rijen MH et al (2011) Identification of cell surface-specific markers to target human nucleus pulposus cells: expression of carbonic anhydrase XII varies with age and degeneration. Arthritis Rheum 63:3876–3886
Tran CM, Smith HE, Symes A, Rittié L, Perbal B, Shapiro IM (2011) Transforming growth factor β controls CCN3 expression in nucleus pulposus cells of the intervertebral disc. Arthritis Rheum 63:3022–3031
Kim JS, Ellman MB, An HS, van Wijnen AJ, Borgia JA, Im HJ (2010) Insulin-like growth factor 1 synergizes with bone morphogenetic protein 7-mediated anabolism in bovine intervertebral disc cells. Arthritis Rheum 62:3706–3715
Hiyama A, Sakai D, Risbud MV, Tanaka M, Arai F, Abe K, Mochida J (2010) Enhancement of intervertebral disc cell senescence by WNT/β-catenin signaling-induced matrix metalloproteinase expression. Arthritis Rheum 62:3036–3047
Wang HQ, Yu XD, Liu ZH, Cheng X, Samartzis D, Jia LT, Wu SX, Huang J, Chen J, Luo ZJ (2011) Deregulated miR-155 promotes Fas-mediated apoptosis in human intervertebral disc degeneration by targeting FADD and caspase-3. J Pathol 225:232–242
Yu X, Li Z, Shen J, Wu WK, Liang J, Weng X, Qiu G (2013) MicroRNA-10b promotes nucleus pulposus cell proliferation through RhoC-Akt pathway by targeting HOXD10 in intervetebral disc degeneration. PLoS One 20:e83080
Li Z, Yu X, Shen J, Chan MT, Wu WK (2015) MicroRNA in intervertebral disc degeneration. Cell Prolif 48:278–283
Maurer B, Stanczyk J, Jüngel A, Akhmetshina A, Trenkmann M, Brock M et al (2010) MicroRNA-29, a key regulator of collagen expression in systemic sclerosis. Arthritis Rheum 62:1733–1743
Nakamachi Y, Kawano S, Takenokuchi M, Nishimura K, Sakai Y et al (2009) MicroRNA-124a is a key regulator of proliferation and monocyte chemoattractant protein 1 secretion in fibroblast-like synoviocytes from patients with rheumatoid arthritis. Arthritis Rheum 60:1294–1304
Zhao B, Yu Q, Li H, Guo X, He X (2014) Characterization of microRNA expression profiles in patients with intervertebral disc degeneration. Int J Mol Med 33:43–50
Liu R, Chen X, Du Y, Yao W, Shen L, Wang C et al (2012) Serum microRNA expression profile as a biomarker in the diagnosis and prognosis of pancreatic cancer. Clin Chem 58:610–618
Wang HY, Yan LX, Shao Q, Fu S, Zhang ZC, Ye W, Zeng YX, Shao JY (2014) Profiling plasma microRNA in nasopharyngeal carcinoma with deep sequencing. Clin Chem 60:773–782
Pfirrmann CW, Metzdorf A, Zanetti M, Hodler J, Boos N (2001) Magnetic resonance classification of lumbar intervertebral disc degeneration. Spine 26:1873–1878
Williams AL, Haughton VM, Meyer GA, Ho KC (1982) Computed tomographic appearance of the bulging annulus. Radiology 142:403–408
Hughes P, Tanner J (1970) The assessment of skeletal maturity in the growing rat. J Anat 106:371–402
Yang F, Leung VY, Luk KD, Chan D, Cheung KM (2009) Injury-induced sequential transformation of notochordal nucleus pulposus to chondrogenic and fibrocartilaginous phenotype in the mouse. J Pathol 218:113–121
Xiao B, Wang Y, Li W, Baker M, Guo J, Corbet K, Tsalik EL, Li QJ, Palmer SM, Woods CW et al (2013) Plasma microRNA signature as a noninvasive biomarker for acute graft-versus-host disease. Blood 122:3365–3375
Dusl M, Senderek J, Müller JS, Vogel JG, Pertl A, Stucka R, Lochmüller H, David R, Abicht A (2015) A 3′-UTR mutation creates a microRNA target site in the GFPT1 gene of patients with congenital myasthenic syndrome. Hum Mol Genet 24:3418–3426
Gandhi R, Healy B, Gholipour T, Egorova S, Musallam A, Hussain MS, Nejad P, Patel B, Hei H, Khoury S et al (2013) Circulating microRNAs as biomarkers for disease staging in multiple sclerosis. Ann Neurol 73:729–740
Qu H, Zheng L, Pu J, Mei H, Xiang X, Zhao X, Li D, Li S, Mao L, Huang K et al (2015) miRNA-558 promotes tumorigenesis and aggressiveness of neuroblastoma cells through activating the transcription of heparanase. Hum Mol Genet 24:2539–2551
Georgantas RW, Streicher K, Greenberg SA, Greenlees LM, Zhu W, Brohawn PZ (2014) Inhibition of myogenic microRNAs 1133 and 206 by inflammatory cytokines links inflammation and muscle degeneration in adult inflammatory myopathies. Arthritis Rheum 66:1022–1033
Matsui Y, Maeda M, Nakagami W, Iwata H (1998) The involvement of matrix metalloproteinases and inflammation in lumbar disc herniation. Spine 23:863–868
Rutges JP, Kummer JA, Oner FC, Verbout AJ, Castelein RJ, Roestenburg HJ, Dhert WJ, Creemers LB (2008) Increased MMP-2 activity during intervertebral disc degeneration is correlated to MMP-14 levels. J Pathol 214:523–530
Bartel DP (2009) MicroRNAs: target recognition and regulatory functions. Cell 136:215–233
Vo NV, Hartman RA, Yurube T, Jacobs LJ, Sowa GA, Kang JD (2013) Expression and regulation of metalloproteinases and their inhibitors in intervertebral disc aging and degeneration. Spine J 13:331–341
Cui Y, Yu J, Urban JP, Young DA (2010) Differential gene expression profiling of metalloproteinases and their inhibitors: a comparison between bovine intervertebral disc nucleus pulposus cells and articular chondrocytes. Spine 35:1101–1108
Acknowledgments
We thank all donors enrolled in the present study.
Authors’ contributions
All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version to be published. Dr. Wang Chen had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis. Wang C, Ji ML, and Zhang XJ were involved in study conception and design. Ji ML, Shi PL, Chen H, and Chang Q were responsible for acquisition of data. Ji ML, Lu J, and Wang SZ analyzed and interpreted the data. Ji ML prepared the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Funding
This work was supported by “The Fundamental Research Funds for the Central Universities (3290005422).
Conflict of interest
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ji, Ml., Zhang, Xj., Shi, Pl. et al. Downregulation of microRNA-193a-3p is involved in invertebral disc degeneration by targeting MMP14. J Mol Med 94, 457–468 (2016). https://doi.org/10.1007/s00109-015-1371-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1371-2