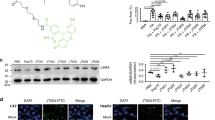
Abstract
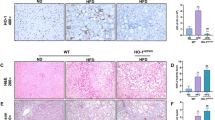
Adiponectin protects against liver fibrosis, but the mechanisms have not been fully elucidated. Here, we showed that adiponectin upregulated inducible nitric oxide synthase (iNOS) messenger RNA (mRNA) and protein expression in hepatic non-parenchymal cells, particularly in hepatic stellate cells (HSCs), and increased nitric oxide (NO2−/NO3−) concentration in HSC-conditioned medium. Adiponectin attenuated HSC proliferation and migration but promoted apoptosis in a NO-dependent manner. More advanced liver fibrosis with decreased iNOS/NO levels was observed in adiponectin knockout mice comparing to wide-type mice when administered with CCI4 while NO donor supplementation rescued the phenotype. Further experiments demonstrated that adiponectin-induced iNOS/NO system activation is mediated through adipoR2-AMPK-JNK/Erk1/2-NF-κB signaling. These data suggest that adiponectin inhibits HSC function, further limiting the development of liver fibrosis at least in part through adiponectin-induced NO release. Therefore, adiponectin-mediated NO signaling may be a novel target for the treatment of liver fibrosis.
Key messages
• Adiponectin activates HSC iNOS/NO and SEC eNOS/NO systems.
• Adiponectin inhibits HSC proliferation and migration but promotes its apoptosis.
• Adiponectin inhibits CCL4-induced liver fibrosis by modulation of liver iNOS/NO.
• Adiponectin stimulates HSC iNOS/NO via adipoR2-AMPK-JNK/ErK1/2-NF-κB pathway.








Similar content being viewed by others
Abbreviations
- AD:
-
Adiponectin
- AD KO:
-
Adiponectin knockout
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- αSMA:
-
Alpha smooth muscle actin
- CCL4:
-
Carbon tetrachloride
- Erk1/2:
-
Extracellular signal-regulated kinase1/2
- HSCs:
-
Hepatic stellate cells
- iNOS:
-
Inducible nitric oxide synthase
- JNK:
-
c-Jun terminal kinase
- l-NAME:
-
NG-nitro-l-arginine methyl ester, hydrochloride
- MAPK:
-
Ras-mitogen-activated protein kinase
- NO:
-
Nitric oxide
- nNOS:
-
Neuronal nitric oxide synthase
- NF-κB:
-
Nuclear factor kappa B
- PDGF:
-
Platelet-derived growth factor
- PDTC:
-
Ammonium pyrrolidinedithiocarbamate
- SECs:
-
Sinusoidal endothelial cells
- siRNA:
-
Small interfering RNA
- SMT:
-
S-methylisothiourea hemisulfate salt
- TGFβ1:
-
Transforming growth factor beta 1
- WT:
-
Wide type
References
Galic S, Oakhill JS, Steinberg GR (2010) Adipose tissue as an endocrine organ. Mol Cell Endocrinol 316:129–139
Masaki T, Chiba S, Tatsukawa H, Yasuda T, Noguchi H, Seike M, Yoshimatsu H (2004) Adiponectin protects LPS-induced liver injury through modulation of TNF-alpha in KK-Ay obese mice. Hepatology 40:177–184
Handy JA, Saxena NK, Fu P, Lin S, Mells JE, Gupta NA, Anania FA (2010) Adiponectin activation of AMPK disrupts leptin-mediated hepatic fibrosis via suppressors of cytokine signaling (SOCS-3). J Cell Biochem 110:1195–1207
Kamada Y, Tamura S, Kiso S, Matsumoto H, Saji Y, Yoshida Y, Fukui K, Maeda N, Nishizawa H, Nagaretani H et al (2003) Enhanced carbon tetrachloride-induced liver fibrosis in mice lacking adiponectin. Gastroenterology 125:1796–1807
Ding X, Saxena NK, Lin S, Xu A, Srinivasan S, Anania FA (2005) The roles of leptin and adiponectin: a novel paradigm in adipocytokine regulation of liver fibrosis and stellate cell biology. Am J Pathol 166:1655–1669
Adachi M, Brenner DA (2008) High molecular weight adiponectin inhibits proliferation of hepatic stellate cells via activation of adenosine monophosphate-activated protein kinase. Hepatology 47:677–685
Grebely J, Feld JJ, Applegate T, Matthews GV, Hellard M, Sherker A, Petoumenos K, Zang G, Shaw I, Yeung B et al (2013) Plasma interferon-gamma-inducible protein-10 (IP-10) levels during acute hepatitis C virus infection. Hepatology 57:2124–2134
Gracia-Sancho J, Lavina B, Rodriguez-Vilarrupla A, Garcia-Caldero H, Fernandez M, Bosch J, Garcia-Pagan JC (2008) Increased oxidative stress in cirrhotic rat livers: a potential mechanism contributing to reduced nitric oxide bioavailability. Hepatology 47:1248–1256
Chen Y, Hozawa S, Sawamura S, Sato S, Fukuyama N, Tsuji C, Mine T, Okada Y, Tanino R, Ogushi Y et al (2005) Deficiency of inducible nitric oxide synthase exacerbates hepatic fibrosis in mice fed high-fat diet. Biochem Biophys Res Commun 326:45–51
Lukivskaya O, Patsenker E, Lis R, Buko VU (2008) Inhibition of inducible nitric oxide synthase activity prevents liver recovery in rat thioacetamide-induced fibrosis reversal. Eur J Clin Invest 38:317–325
Vercelino R, Crespo I, de Souza GF, Cuevas MJ, de Oliveira MG, Marroni NP, Gonzalez-Gallego J, Tunon MJ (2010) S-nitroso-N-acetylcysteine attenuates liver fibrosis in cirrhotic rats. J Mol Med (Berl) 88:401–411
Failli P, De FR, Caligiuri A, Gentilini A, Romanelli RG, Marra F, Batignani G, Guerra CT, Laffi G, Gentilini P et al (2000) Nitrovasodilators inhibit platelet-derived growth factor-induced proliferation and migration of activated human hepatic stellate cells. Gastroenterology 119:479–492
Langer DA, Das A, Semela D, Kang-Decker N, Hendrickson H, Bronk SF, Katusic ZS, Gores GJ, Shah VH (2008) Nitric oxide promotes caspase-independent hepatic stellate cell apoptosis through the generation of reactive oxygen species. Hepatology 47:1983–1993
Lee JS, Kang Decker N, Chatterjee S, Yao J, Friedman S, Shah V (2005) Mechanisms of nitric oxide interplay with Rho GTPase family members in modulation of actin membrane dynamics in pericytes and fibroblasts. Am J Pathol 166:1861–1870
Deleve LD, Wang X, Guo Y (2008) Sinusoidal endothelial cells prevent rat stellate cell activation and promote reversion to quiescence. Hepatology 48:920–930
Kawada N, Kuroki T, Uoya M, Inoue M, Kobayashi K (1996) Smooth muscle alpha-actin expression in rat hepatic stellate cell is regulated by nitric oxide and cGMP production. Biochem Biophys Res Commun 229:238–242
Cheng KK, Lam KS, Wang Y, Huang Y, Carling D, Wu D, Wong C, Xu A (2007) Adiponectin-induced endothelial nitric oxide synthase activation and nitric oxide production are mediated by APPL1 in endothelial cells. Diabetes 56:1387–1394
Gonon AT, Widegren U, Bulhak A, Salehzadeh F, Persson J, Sjoquist PO, Pernow J (2008) Adiponectin protects against myocardial ischaemia-reperfusion injury via AMP-activated protein kinase, Akt, and nitric oxide. Cardiovasc Res 78:116–122
Nishimura M, Izumiya Y, Higuchi A, Shibata R, Qiu J, Kudo C, Shin HK, Moskowitz MA, Ouchi N (2008) Adiponectin prevents cerebral ischemic injury through endothelial nitric oxide synthase dependent mechanisms. Circulation 117:216–223
Lago R, Gomez R, Otero M, Lago F, Gallego R, Dieguez C, Gomez-Reino JJ, Gualillo O (2008) A new player in cartilage homeostasis: adiponectin induces nitric oxide synthase type II and pro-inflammatory cytokines in chondrocytes. Osteoarthr Cartil 16:1101–1109
Koskinen A, Juslin S, Nieminen R, Moilanen T, Vuolteenaho K, Moilanen E (2011) Adiponectin associates with markers of cartilage degradation in osteoarthritis and induces production of proinflammatory and catabolic factors through mitogen-activated protein kinase pathways. Arthritis Res Ther 13:R184
Chen H, Montagnani M, Funahashi T, Shimomura I, Quon MJ (2003) Adiponectin stimulates production of nitric oxide in vascular endothelial cells. J Biol Chem 278:45021–45026
Chang J, Li Y, Huang Y, Lam KS, Hoo RL, Wong WT, Cheng KK, Wang Y, Vanhoutte PM, Xu A (2010) Adiponectin prevents diabetic premature senescence of endothelial progenitor cells and promotes endothelial repair by suppressing the p38 MAP kinase/p16INK4A signaling pathway. Diabetes 59:2949–2959
El Hasnaoui-Saadani R, Alayza RC, Launay T, Pichon A, Quidu P, Beaudry M, Leon-Velarde F, Richalet JP, Duvallet A, Favret F (1985) Brain stem NO modulates ventilatory acclimatization to hypoxia in mice. J Appl Physiol 103:1506–1512
Lowenstein CJ, Alley EW, Raval P, Snowman AM, Snyder SH, Russell SW, Murphy WJ (1993) Macrophage nitric oxide synthase gene: two upstream regions mediate induction by interferon gamma and lipopolysaccharide. Proc Natl Acad Sci U S A 90:9730–9734
Xie QW, Whisnant R, Nathan C (1993) Promoter of the mouse gene encoding calcium-independent nitric oxide synthase confers inducibility by interferon gamma and bacterial lipopolysaccharide. J Exp Med 177:1779–1784
Fruebis J, Tsao TS, Javorschi S, Ebbets-Reed D, Erickson MR, Yen FT, Bihain BE, Lodish HF (2001) Proteolytic cleavage product of 30-kDa adipocyte complement-related protein increases fatty acid oxidation in muscle and causes weight loss in mice. Proc Natl Acad Sci U S A 98:2005–2010
Yamauchi T, Kamon J, Minokoshi Y, Ito Y, Waki H, Uchida S, Yamashita S, Noda M, Kita S, Ueki K et al (2002) Adiponectin stimulates glucose utilization and fatty-acid oxidation by activating AMP-activated protein kinase. Nat Med 8:1288–1295
Shah V, Cao S, Hendrickson H, Yao J, Katusic ZS (2001) Regulation of hepatic eNOS by caveolin and calmodulin after bile duct ligation in rats. Am J Physiol Gastrointest Liver Physiol 280:G1209–G1216
Leifeld L, Fielenbach M, Dumoulin FL, Speidel N, Sauerbruch T, Spengler U (2002) Inducible nitric oxide synthase (iNOS) and endothelial nitric oxide synthase (eNOS) expression in fulminant hepatic failure. J Hepatol 37:613–619
Wang W, Zhao CY, Wang YD, He X, Shen C, Cao W, Zhou JY, Zhen Z (2011) Adiponectin inhibits the activation of hepatic stellate cells induced by TGFb1 via up-regulating the expression of eNOS. Zhonghua Gan Zang Bing Za Zhi 19:917–922
Caligiuri A, Bertolani C, Guerra CT, Aleffi S, Galastri S, Trappoliere M, Vizzutti F, Gelmini S, Laffi G, Pinzani M et al (2008) Adenosine monophosphate-activated protein kinase modulates the activated phenotype of hepatic stellate cells. Hepatology 47:668–676
Wang Y, Lam KS, Xu JY, Lu G, Xu LY, Cooper GJ, Xu A (2005) Adiponectin inhibits cell proliferation by interacting with several growth factors in an oligomerization-dependent manner. J Biol Chem 280:18341–18347
Li J, Billiar TR (1999) Nitric oxide. IV. Determinants of nitric oxide protection and toxicity in liver. Am J Physiol 276:G1069–G1073
Rockey DC, Shah V (2004) Nitric oxide biology and the liver: report of an AASLD research workshop. Hepatology 39:250–257
Chen T, Zamora R, Zuckerbraun B, Billiar TR (2003) Role of nitric oxide in liver injury. Curr Mol Med 3:519–526
Clemens MG (1999) Nitric oxide in liver injury. Hepatology 30:1–5
Shah V, Haddad FG, Garcia-Cardena G, Frangos JA, Mennone A, Groszmann RJ, Sessa WC (1997) Liver sinusoidal endothelial cells are responsible for nitric oxide modulation of resistance in the hepatic sinusoids. J Clin Invest 100:2923–2930
Suematsu M, Goda N, Sano T, Kashiwagi S, Egawa T, Shinoda Y, Ishimura Y (1995) Carbon monoxide: an endogenous modulator of sinusoidal tone in the perfused rat liver. J Clin Invest 96:2431–2437
Utz J, Ullrich V (1991) Carbon monoxide relaxes ileal smooth muscle through activation of guanylate cyclase. Biochem Pharmacol 41:1195–1201
Furchgott RF, Jothianandan D (1991) Endothelium-dependent and -independent vasodilation involving cyclic GMP: relaxation induced by nitric oxide, carbon monoxide and light. Blood Vessels 28:52–61
Acknowledgments
We thank Xin Wang (Flow Cytometry Facility, Westmead Millennium Institute) and Hong Yu (Microscopy Unit, Westmead Millennium Institute) for technical assistance. This study was supported by the National Health and Medical Research Council of Australia (AP1004595 and a Program Grant 1053206) and the Robert W. Storr Bequest to the University of Sydney.
Conflict of interest
The authors declare that they have no competing interests.
Author information
Authors and Affiliations
Corresponding author
Additional information
Zhixia Dong and Lin Su contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 653 kb)
Rights and permissions
About this article
Cite this article
Dong, Z., Su, L., Esmaili, S. et al. Adiponectin attenuates liver fibrosis by inducing nitric oxide production of hepatic stellate cells. J Mol Med 93, 1327–1339 (2015). https://doi.org/10.1007/s00109-015-1313-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1313-z