Abstract
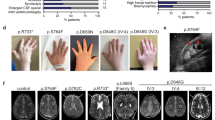
Filamin A, the filamentous protein encoded by the X-linked gene FLNA, cross-links cytoskeletal actin into three-dimensional networks, facilitating its role as a signalling scaffold and a mechanosensor of extrinsic shear forces. Central to these functions is the ability of FLNA to form V-shaped homodimers through its C-terminal located filamin repeat 24. Additionally, many proteins that interact with FLNA have a binding site that includes the C-terminus of the protein. Here, a cohort of patients with mutations affecting this region of the protein is studied, with particular emphasis on the phenotype of male hemizygotes. Seven unrelated families are reported, with five exhibiting a typical female presentation of periventricular heterotopia (PH), a neuronal migration disorder typically caused by loss-of-function mutations in FLNA. One male presents with widespread PH consistent with previous male phenotypes attributable to hypomorphic mutations in FLNA. In stark contrast, two brothers are described with a mild PH presentation, due to a missense mutation (p.Gly2593Glu) inserting a large negatively charged amino acid into the hydrophobic dimerisation interface of FLNA. Co-immunoprecipitation, in vitro cross-linking studies and gel filtration chromatography all demonstrated that homodimerisation of isolated FLNA repeat 24 is abolished by this p.Gly2593Glu substitution but that extended FLNAGly2593Glu repeat 16–24 constructs exhibit dimerisation. These observations imply that other interactions apart from those mediated by the canonical repeat 24 dimerisation interface contribute to FLNA homodimerisation and that mutations affecting this region of the protein can have broad phenotypic effects.
Key messages
• Mutations in the X-linked gene FLNA cause a spectrum of syndromes.
• Genotype-phenotype correlations are emerging but still remain unclear.
• C-term mutations can confer male lethality, survival or connective tissue defects.
• Mutations leading to the latter affect filamin dimerisation.
• This deficit is compensated for by remotely acting domains elsewhere in FLNA.




Similar content being viewed by others
References
Fox JW, Lamperti ED, Eksioglu YZ, Hong SE, Feng Y, Graham DA, Scheffer IE, Dobyns WB, Hirsch BA, Radtke RA et al (1998) Mutations in filamin 1 prevent migration of cerebral cortical neurons in human periventricular heterotopia. Neuron 21:1315–1325
Kyndt F, Gueffet JP, Probst V, Jaafar P, Legendre A, Le Bouffant F, Toquet C, Roy E, McGregor L, Lynch SA et al (2007) Mutations in the gene encoding filamin A as a cause for familial cardiac valvular dystrophy. Circulation 115:40–49
Robertson SP, Twigg SR, Sutherland-Smith AJ, Biancalana V, Gorlin RJ, Horn D, Kenwrick SJ, Kim CA, Morava E, Newbury-Ecob R et al (2003) Localized mutations in the gene encoding the cytoskeletal protein filamin A cause diverse malformations in humans. Nat Genet 33:487–491
Sheen VL, Jansen A, Chen MH, Parrini E, Morgan T, Ravenscroft R, Ganesh V, Underwood T, Wiley J, Leventer R et al (2005) Filamin A mutations cause periventricular heterotopia with Ehlers-Danlos syndrome. Neurology 64:254–262
Sun Y, Almomani R, Aten E, Celli J, van der Heijden J, Venselaar H, Robertson SP, Baroncini A, Franco B, Basel-Vanagaite L et al (2010) Terminal osseous dysplasia is caused by a single recurrent mutation in the FLNA gene. Am J Hum Genet 87:146–153
Hartwig JH, Tyler J, Stossel TP (1980) Actin-binding protein promotes the bipolar and perpendicular branching of actin filaments. J Cell Biol 87:841–848
Gehler S, Baldassarre M, Lad Y, Leight JL, Wozniak MA, Riching KM, Eliceiri KW, Weaver VM, Calderwood DA, Keely PJ (2009) Filamin A-beta1 integrin complex tunes epithelial cell response to matrix tension. Mol Biol Cell 20:3224–3238
Lynch CD, Gauthier NC, Biais N, Lazar AM, Roca-Cusachs P, Yu CH, Sheetz MP (2011) Filamin depletion blocks endoplasmic spreading and destabilizes force-bearing adhesions. Mol Biol Cell 22:1263–1273
Nakamura F, Stossel TP, Hartwig JH (2011) The filamins: organizers of cell structure and function. Cell Adhes Migr 5
Fucini P, Renner C, Herberhold C, Noegel AA, Holak TA (1997) The repeating segments of the F-actin cross-linking gelation factor (ABP-120) have an immunoglobulin-like fold. Nat Struct Biol 4:223–230
Gorlin JB, Yamin R, Egan S, Stewart M, Stossel TP, Kwiatkowski DJ, Hartwig JH (1990) Human endothelial actin-binding protein (ABP-280, nonmuscle filamin): a molecular leaf spring. J Cell Biol 111:1089–1105
Gardel ML, Nakamura F, Hartwig JH, Crocker JC, Stossel TP, Weitz DA (2006) Prestressed F-actin networks cross-linked by hinged filamins replicate mechanical properties of cells. Proc Natl Acad Sci U S A 103:1762–1767
Himmel M, Van Der Ven PF, Stocklein W, Furst DO (2003) The limits of promiscuity: isoform-specific dimerization of filamins. Biochemistry 42:430–439
Weihing RR (1988) Actin-binding and dimerization domains of HeLa cell filamin. Biochemistry 27:1865–1869
Pudas R, Kiema TR, Butler PJ, Stewart M, Ylanne J (2005) Structural basis for vertebrate filamin dimerization. Structure 13:111–119
Kainulainen T, Pender A, D'Addario M, Feng Y, Lekic P, McCulloch CA (2002) Cell death and mechanoprotection by filamin a in connective tissues after challenge by applied tensile forces. J Biol Chem 277:21998–22009
Kasza KE, Nakamura F, Hu S, Kollmannsberger P, Bonakdar N, Fabry B, Stossel TP, Wang N, Weitz DA (2009) Filamin A is essential for active cell stiffening but not passive stiffening under external force. Biophys J 96:4326–4335
Kesner BA, Ding F, Temple BR, Dokholyan NV (2010) N-terminal strands of filamin Ig domains act as a conformational switch under biological forces. Proteins 78:12–24
Shifrin Y, Arora PD, Ohta Y, Calderwood DA, McCulloch CA (2009) The role of FilGAP-filamin A interactions in mechanoprotection. Mol Biol Cell 20:1269–1279
Nakamura F, Osborn TM, Hartemink CA, Hartwig JH, Stossel TP (2007) Structural basis of filamin A functions. J Cell Biol 179:1011–1025
Seo MD, Seok SH, Im H, Kwon AR, Lee SJ, Kim HR, Cho Y, Park D, Lee BJ (2009) Crystal structure of the dimerization domain of human filamin A. Proteins 75:258–263
Gargiulo A, Auricchio R, Barone MV, Cotugno G, Reardon W, Milla PJ, Ballabio A, Ciccodicola A, Auricchio A (2007) Filamin A is mutated in X-linked chronic idiopathic intestinal pseudo-obstruction with central nervous system involvement. Am J Hum Genet 80:751–758
Guerrini R, Mei D, Sisodiya S, Sicca F, Harding B, Takahashi Y, Dorn T, Yoshida A, Campistol J, Kramer G et al (2004) Germline and mosaic mutations of FLN1 in men with periventricular heterotopia. Neurology 63:51–56
Hehr U, Hehr A, Uyanik G, Phelan E, Winkler J, Reardon W (2006) A filamin A splice mutation resulting in a syndrome of facial dysmorphism, periventricular nodular heterotopia, and severe constipation reminiscent of cerebro-fronto-facial syndrome. J Med Genet 43:541–544
Parrini E, Mei D, Wright M, Dorn T, Guerrini R (2004) Mosaic mutations of the FLN1 gene cause a mild phenotype in patients with periventricular heterotopia. Neurogenetics 5:191–196
Sheen VL, Dixon PH, Fox JW, Hong SE, Kinton L, Sisodiya SM, Duncan JS, Dubeau F, Scheffer IE, Schachter SC et al (2001) Mutations in the X-linked filamin 1 gene cause periventricular nodular heterotopia in males as well as in females. Hum Mol Genet 10:1775–1783
Fergelot P, Coupry I, Rooryck C, Deforges J, Maurat E, Sole G, Boute O, Dieux-Coeslier A, David A, Marchal C et al (2012) Atypical male and female presentations of FLNA-related periventricular nodular heterotopia. Eur J Med Genet 55:313–318
Liang CC, Park AY, Guan JL (2007) In vitro scratch assay: a convenient and inexpensive method for analysis of cell migration in vitro. Nat Protoc 2:329–333
Collins TJ (2007) ImageJ for microscopy. Biotechniques 43:25–30
Reinstein E, Frentz S, Morgan T, Garcia-Minaur S, Leventer RJ, McGillivray G, Pariani M, van der Steen A, Pope M, Holder-Espinasse M et al (2013) Vascular and connective tissue anomalies associated with X-linked periventricular heterotopia due to mutations in filamin A. Eur J Hum Genet 21:494–502
Cunningham CC, Gorlin JB, Kwiatkowski DJ, Hartwig JH, Janmey PA, Byers HR, Stossel TP (1992) Actin-binding protein requirement for cortical stability and efficient locomotion. Science 255:325–327
Feng Y, Chen MH, Moskowitz IP, Mendonza AM, Vidali L, Nakamura F, Kwiatkowski DJ, Walsh CA (2006) Filamin A (FLNA) is required for cell-cell contact in vascular development and cardiac morphogenesis. Proc Natl Acad Sci U S A 103:19836–19841
Sheen VL, Feng Y, Graham D, Takafuta T, Shapiro SS, Walsh CA (2002) Filamin A and filamin B are co-expressed within neurons during periods of neuronal migration and can physically interact. Hum Mol Genet 11:2845–2854
Ferland RJ, Batiz LF, Neal J, Lian G, Bundock E, Lu J, Hsiao YC, Diamond R, Mei D, Banham AH et al (2009) Disruption of neural progenitors along the ventricular and subventricular zones in periventricular heterotopia. Hum Mol Genet 18:497–516
Baldassarre M, Razinia Z, Burande CF, Lamsoul I, Lutz PG, Calderwood DA (2009) Filamins regulate cell spreading and initiation of cell migration. PLoS One 4:e7830
Ylanne J, Scheffzek K, Young P, Saraste M (2001) Crystal structure of the alpha-actinin rod reveals an extensive torsional twist. Structure 9:597–604
Stossel TP, Condeelis J, Cooley L, Hartwig JH, Noegel A, Schleicher M, Shapiro SS (2001) Filamins as integrators of cell mechanics and signalling. Nat Rev Mol Cell Biol 2:138–145
Ohta Y, Hartwig JH, Stossel TP (2006) FilGAP, a Rho- and ROCK-regulated GAP for Rac binds filamin A to control actin remodelling. Nat Cell Biol 8:803–814
Gay O, Gilquin B, Nakamura F, Jenkins ZA, McCartney R, Krakow D, Deshiere A, Assard N, Hartwig JH, Robertson SP et al (2011) RefilinB (FAM101B) targets filamin A to organize perinuclear actin networks and regulates nuclear shape. Proc Natl Acad Sci U S A 108:11464–11469
Nakamura F, Heikkinen O, Pentikainen OT, Osborn TM, Kasza KE, Weitz DA, Kupiainen O, Permi P, Kilpelainen I, Ylanne J et al (2009) Molecular basis of filamin A-FilGAP interaction and its impairment in congenital disorders associated with filamin A mutations. PLoS One 4:e4928
Acknowledgments
We are grateful for the willing participation of the family described here. This work was supported by Curekids New Zealand (SR) and the Royal Society of New Zealand Marsden Fund (AJSS and SR).
Conflict of interest
The authors declare no conflicts of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
John Tolmie: In memoriam
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 707 kb)
Rights and permissions
About this article
Cite this article
van Kogelenberg, M., Clark, A.R., Jenkins, Z. et al. Diverse phenotypic consequences of mutations affecting the C-terminus of FLNA. J Mol Med 93, 773–782 (2015). https://doi.org/10.1007/s00109-015-1261-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-015-1261-7