Abstract
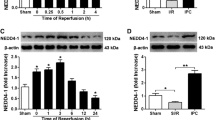
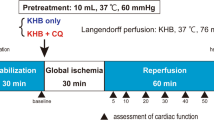
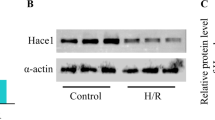
Activation of ERK signaling may promote cardioprotection from ischemia–reperfusion (I/R) injury. ZnT-1, a protein that confers resistance from zinc toxicity, was found to interact with Raf-1 kinase through its C-terminal domain, leading to downstream activation of ERK. In the present study, we evaluated the effects of ZnT-1 in cultured murine cardiomyocytes (HL-1 cells) that were exposed to simulated-I/R. Cellular injury was evaluated by lactate dehydrogenase (LDH) release and by staining for pro-apoptotic caspase activation. Overexpression of ZnT-1 markedly reduced LDH release and caspase activation following I/R. Knockdown of endogenous ZnT-1 augmented the I/R-induced release of LDH and increased caspase activation following I/R. Phospho-ERK levels were significantly increased following I/R in cells overexpressing ZnT-1, while knockdown of ZnT-1 reduced phospho-ERK levels. Pretreatment of cells with the MEK inhibitor PD98059 abolished the protective effect of ZnT-1 following I/R. Accordingly, a truncated form of ZnT-1 lacking the C-terminal domain failed to induce ERK activation and did not protect the cells from I/R injury. In contrast, expression of the C-terminal domain by itself was sufficient to induce ERK activation and I/R protection. Interestingly, the C-terminal of the ZnT-1 did not have protective effect against the toxicity of zinc. In the isolated rat heart, global ischemic injury rapidly increased the endogenous levels of ZnT-1. However, following reperfusion ZnT-1 levels were found to be decreased. Our findings indicate that ZnT-1 may have important role in the ischemic myocardium through its ability to interact with Raf-1 kinase.









Similar content being viewed by others
References
Yellon DM, Hausenloy DJ (2007) Myocardial reperfusion injury. N Engl J Med 357:1121–1135. doi:10.1056/NEJMra071667
Buja LM (2005) Myocardial ischemia and reperfusion injury. Cardiovasc Pathol 14:170
Hausenloy DJ, Yellon DM (2009) Preconditioning and postconditioning: underlying mechanisms and clinical application. Atherosclerosis 204:334–341
Hausenloy DJ, Yellon DM (2007) Reperfusion injury salvage kinase signalling: taking a RISK for cardioprotection. Heart Fail Rev 12:217–234
Iliodromitis EK, Papalois A, Gritsopoulos G, Kremastinos DT, Yellon DM, Hausenloy DJ (2009) Remote ischaemic preconditioning and postconditioning and the reperfusion injury salvage kinase pathway. Heart 95:13
Philipp S, Critz SD, Cui L, Solodushko V, Cohen MV, Downey JM (2006) Localizing extracellular signal-regulated kinase (ERK) in pharmacological preconditioning’s trigger pathway. Basic Res Cardiol 101:159–167
Hausenloy DJ, Yellon DM (2009) Cardioprotective growth factors. Cardiovasc Res 83:179–194. doi:10.1093/cvr/cvp062
Lips DJ, Bueno OF, Wilkins BJ, Purcell NH, Kaiser RA, Lorenz JN, Voisin L, Saba-El-Leil MK, Meloche S, Pouyssegur J et al (2004) MEK1-ERK2 signaling pathway protects myocardium from ischemic injury in vivo. Circulation 109:1938–1941. doi:10.1161/01.CIR.0000127126.73759.23
Bruinsma JJ, Jirakulaporn T, Muslin AJ, Kornfeld K (2002) Zinc ions and cation diffusion facilitator proteins regulate Ras-mediated signaling. Dev Cell 2:567–578
Jirakulaporn T, Muslin AJ (2004) Cation diffusion facilitator proteins modulate Raf-1 activity. J Biol Chem 279:27807–27815
Lazarczyk M, Pons C, Mendoza JA, Cassonnet P, Jacob Y, Favre M (2008) Regulation of cellular zinc balance as a potential mechanism of EVER-mediated protection against pathogenesis by cutaneous oncogenic human papillomaviruses. J Exp Med 205:35–42. doi:10.1084/jem.20071311
Palmiter R, Findley S (1995) Cloning and functional characterization of a mammalian zinc transporter that confers resistance to zinc. EMBO J 14:639–649
Cousins RJ, Liuzzi JP, Lichten LA (2006) Mammalian zinc transport, trafficking, and signals. J Biol Chem 281:24085–24089
Levy S, Beharier O, Etzion Y, Mor M, Buzaglo L, Shaltiel L, Gheber LA, Kahn J, Muslin AJ, Katz A et al (2009) Molecular basis for zinc transporter 1 action as an endogenous inhibitor of L-type calcium channels. J Biol Chem 284:32434–32443
Beharier O, Etzion Y, Katz A, Friedman H, Tenbosh N, Zacharish S, Bereza S, Goshen U, Moran A (2007) Crosstalk between L-type calcium channels and ZnT-1, a new player in rate-dependent cardiac electrical remodeling. Cell Calcium 42:71–82
Kamalov G, Deshmukh PA, Baburyan NY, Gandhi MS, Johnson PL, Ahokas RA, Bhattacharya SK, Sun Y, Gerling IC, Weber KT (2009) Coupled calcium and zinc dyshomeostasis and oxidative stress in cardiac myocytes and mitochondria of rats with chronic aldosteronism. J Cardiovasc Pharmacol 53:414–423. doi:10.1097/FJC.0b013e3181a15e77
White SM, Constantin PE, Claycomb WC (2004) Cardiac physiology at the cellular level: use of cultured HL-1 cardiomyocytes for studies of cardiac muscle cell structure and function. Am J Physiol Heart Circ Physiol 286:H823–H829
Seymour EM, Wu SY, Kovach MA, Romano MA, Traynor JR, Claycomb WC, Bolling SF (2003) HL-1 myocytes exhibit PKC and K(ATP) channel-dependent delta opioid preconditioning. J Surg Res 114:187–194. doi:S0022480403002488
Facundo HT, de Paula JG, Kowaltowski AJ (2005) Mitochondrial ATP-sensitive K + channels prevent oxidative stress, permeability transition and cell death. J Bioenerg Biomembr 37:75–82. doi:10.1007/s10863-005-4130-1
Ruiz-Meana M, Garcia-Dorado D, Miro-Casas E, Abellan A, Soler-Soler J (2006) Mitochondrial Ca2+ uptake during simulated ischemia does not affect permeability transition pore opening upon simulated reperfusion. Cardiovasc Res 71:715–724. doi:10.1016/j.cardiores.2006.06.019
Ljubkovic M, Marinovic J, Fuchs A, Bosnjak ZJ, Bienengraeber M (2006) Targeted expression of Kir6.2 in mitochondria confers protection against hypoxic stress. J Physiol 577:17–29. doi:10.1113/jphysiol.2006.118299
Cavalheiro RA, Marin RM, Rocco SA, Cerqueira FM, da Silva CC, Rittner R, Kowaltowski AJ, Vercesi AE, Franchini KG, Castilho RF (2010) Potent cardioprotective effect of the 4-anilinoquinazoline derivative PD153035: involvement of mitochondrial K(ATP) channel activation. PLoS One 5:e10666. doi:10.1371/journal.pone.0010666
Yitzhaki S, Huang C, Liu W, Lee Y, Gustafsson AB, Mentzer RM Jr, Gottlieb RA (2009) Autophagy is required for preconditioning by the adenosine A1 receptor-selective agonist CCPA. Basic Res Cardiol 104:157–167. doi:10.1007/s00395-009-0006-6
McMahon RJ, Cousins RJ (1998) Regulation of the zinc transporter ZnT-1 by dietary zinc. PNAS 95:4841–4846
Heusch G (2009) No risk, no … cardioprotection? A critical perspective. Cardiovasc Res 84:173–175. doi:10.1093/cvr/cvp298
Skyschally A, van Caster P, Boengler K, Gres P, Musiolik J, Schilawa D, Schulz R, Heusch G (2009) Ischemic postconditioning in pigs: no causal role for RISK activation. Circ Res 104:15–18. doi:10.1161/circresaha.108.186429
Murphy E, Steenbergen C (2008) Mechanisms underlying acute protection from cardiac ischemia-reperfusion injury. Physiol Rev 88:581–609. doi:10.1152/physrev.00024.2007
Ramaraj R, Forman MB, Jackson EK, Lowenstein CJ, Ibanez B, Cimmino G, Badimon JJ, Korantzopoulos PG, Goudevenos JA, Yellon DM et al (2007) Myocardial reperfusion injury. N Engl J Med 357:2408–2410. doi:10.1056/NEJMc072913
Armstrong SC (2004) Protein kinase activation and myocardial ischemia/reperfusion injury. Cardiovasc Res 61:427–436. doi:10.1016/j.cardiores.2003.09.031
Beguin PC, Belaidi E, Godin-Ribuot D, Levy P, Ribuot C (2007) Intermittent hypoxia-induced delayed cardioprotection is mediated by PKC and triggered by p38 MAP kinase and Erk1/2. J Mol Cell Cardiol 42:343–351. doi:10.1016/j.yjmcc.2006.11.008
Przyklenk K, Hata K, Kloner RA (1997) Is calcium a mediator of infarct size reduction with preconditioning in canine myocardium? Circulation 96:1305–1312
Miyawaki H, Zhou X, Ashraf M (1996) Calcium preconditioning elicits strong protection against ischemic injury via protein kinase C signaling pathway. Circ Res 79:137–146
Cain BS, Meldrum DR, Cleveland JC, Meng X, Banerjee A, Harken AH (1999) Clinical L-type Ca(2+) channel blockade prevents ischemic preconditioning of human myocardium. J Mol Cell Cardiol 31:2191–2197
Schulman D, Latchman DS, Yellon DM (2002) Urocortin protects the heart from reperfusion injury via upregulation of p42/p44 MAPK signaling pathway. Am J Physiol Heart Circ Physiol 283:H1481–H1488. doi:10.1152/ajpheart.01089.2001
Brar BK, Jonassen AK, Stephanou A, Santilli G, Railson J, Knight RA, Yellon DM, Latchman DS (2000) Urocortin protects against ischemic and reperfusion injury via a MAPK-dependent pathway. J Biol Chem 275:8508–8514
Acknowledgments
This work was supported by Israel Science Foundation (ISF) grant 992/07 (AM and YE) and the Deutsche Forschungsgemeinschaf (DFG) grant MO 1932/1-1 (AM).
Disclosures
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Authors Ofer beharier and Shani Dror contributed equally to this work. Authors Arie Moran and Yoram Etzion contributed equally to this work.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOC 325 kb)
Rights and permissions
About this article
Cite this article
Beharier, O., Dror, S., Levy, S. et al. ZnT-1 protects HL-1 cells from simulated ischemia–reperfusion through activation of Ras–ERK signaling. J Mol Med 90, 127–138 (2012). https://doi.org/10.1007/s00109-011-0845-0
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0845-0