Abstract
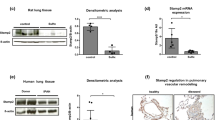
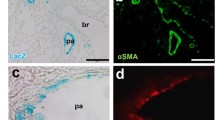
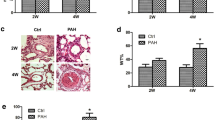
Pulmonary arterial hypertension (PAH) is a vascular remodeling disease characterized by enhanced proliferation and suppressed apoptosis of pulmonary artery smooth muscle cells (PASMC). This apoptosis resistance is characterized by PASMC mitochondrial hyperpolarization [in part, due to decreased pyruvate dehydrogenase (PDH) activity], decreased mitochondrial reactive oxygen species (mROS), downregulation of Kv1.5, increased [Ca++]i, and activation of the transcription factor nuclear factor of activated T cells (NFAT). Inflammatory cells are present within and around the remodeled arteries and patients with PAH have elevated levels of inflammatory cytokines, including tumor necrosis factor-α (TNFα). We hypothesized that the inflammatory cytokine TNFα inhibits PASMC PDH activity, inducing a PAH phenotype in normal PASMC. We exposed normal human PASMC to recombinant human TNFα and measured PDH activity. In TNFα-treated cells, PDH activity was significantly decreased. Similar to exogenous TNFα, endogenous TNFα secreted from activated human CD8+ T cells, but not quiescent T cells, caused mitochondrial hyperpolarization, decreased mROS, decreased K+ current, increased [Ca++]i, and activated NFAT in normal human PASMC. A TNFα antibody completely prevented, while recombinant TNFα mimicked the T cell-induced effects. In vivo, the TNFα antagonist etanercept prevented and reversed monocrotaline (MCT)-induced PAH. In a separate model, T cell deficient rats developed less severe MCT-induced PAH compared to their controls. We show that TNFα can inhibit PASMC PDH activity and induce a PAH phenotype. Our work supports the use of anti-inflammatory therapies for PAH.








Similar content being viewed by others
References
Dromparis P, Sutendra G, Michelakis ED (2010) The role of mitochondria in pulmonary vascular remodeling. J Mol Med 88:1003–1010
McMurtry MS, Bonnet S, Wu X, Dyck JR, Haromy A, Hashimoto K, Michelakis ED (2004) Dichloroacetate prevents and reverses pulmonary hypertension by inducing pulmonary artery smooth muscle cell apoptosis. Circ Res 95:830–840
Michelakis ED, Sutendra G, Dromparis P, Webster L, Haromy A, Niven E, Maguire C, Gammer TL, Mackey JR, Fulton D et al (2010) Metabolic modulation of glioblastoma with dichloroacetate. Sci Transl Med 2:31ra34
Sutendra G, Bonnet S, Rochefort G, Haromy A, Folmes KD, Lopaschuck GD, Dyck JRB, Michelakis ED (2010) Fatty acid oxidation and malonyl-CoA decarboxylase in vascular remodeling of pulmonary hypertension. Sci Transl Med 2:44ra58
Kim JW, Dang CV (2005) Multifaceted roles of glycolytic enzymes. Trends Biochem Sci 30:142–150
Bonnet S, Archer SL, Allalunis-Turner J, Haromy A, Beaulieu C, Thompson R, Lee CT, Lopaschuk GD, Puttagunta L, Bonnet S et al (2007) A mitochondria-K+ channel axis is suppressed in cancer and its normalization promotes apoptosis and inhibits cancer growth. Cancer Cell 11:37–51
Guignabert C, Tu L, Izikki M, Dewachter L, Zadigue P, Humbert M, Adnot S, Fadel E, Eddahibi S (2009) Dichloroacetate treatment partially regresses established pulmonary hypertension in mice with SM22alpha-targeted overexpression of the serotonin transporter. FASEB J 23:4135–4147
Zamzami N, Kroemer G (2001) The mitochondrion in apoptosis: how Pandora’s box opens. Nat Rev Mol Cell Biol 2:67–71
Archer SL, Souil E, Dinh-Xuan AT, Schremmer B, Mercier JC, El Yaagoubi A, Nguyen-Huu L, Reeve HL, Hampl V (1998) Molecular identification of the role of voltage-gated K+ channels, Kv1.5 and Kv2.1, in hypoxic pulmonary vasoconstriction and control of resting membrane potential in rat pulmonary artery myocytes. J Clin Invest 101:2319–2330
Platoshyn O, Golovina VA, Bailey CL, Limsuwan A, Krick S, Juhaszova M, Seiden JE, Rubin LJ, Yuan JX (2000) Sustained membrane depolarization and pulmonary artery smooth muscle cell proliferation. Am J Physiol Cell Physiol 279:C1540–C1549
Remillard CV, Yuan JX (2004) Activation of K+ channels: an essential pathway in programmed cell death. Am J Physiol Lung Cell Mol Physiol 286:L49–L67
Pastorino JG, Hoek JB, Shulga N (2005) Activation of glycogen synthase kinase 3beta disrupts the binding of hexokinase II to mitochondria by phosphorylating voltage-dependent anion channel and potentiates chemotherapy-induced cytotoxicity. Cancer Res 65:10545–10554
Bonnet S, Rochefort G, Sutendra G, Archer SL, Haromy A, Webster L, Hashimoto K, Bonnet SN, Michelakis ED (2007) The nuclear factor of activated T cells in pulmonary arterial hypertension can be therapeutically targeted. Proc Natl Acad Sci USA 104:11418–11423
Taraseviciene-Stewart L, Nicolls MR, Kraskauskas D, Scerbavicius R, Burns N, Cool C, Wood K, Parr JE, Boackle SA, Voelkel NF (2007) Absence of T cells confers increased pulmonary arterial hypertension and vascular remodeling. Am J Respir Crit Care Med 175:1280–1289
Tuder RM, Groves B, Badesch DB, Voelkel NF (1994) Exuberant endothelial cell growth and elements of inflammation are present in plexiform lesions of pulmonary hypertension. Am J Pathol 144:275–285
Caslin AW, Heath D, Madden B, Yacoub M, Gosney JR, Smith P (1990) The histopathology of 36 cases of plexogenic pulmonary arteriopathy. Histopathology 16:9–19
Cool CD, Rai PR, Yeager ME, Hernandez-Saavedra D, Serls AE, Bull TM, Geraci MW, Brown KK, Routes JM, Tuder RM et al (2003) Expression of human herpesvirus 8 in primary pulmonary hypertension. N Engl J Med 349:1113–1122
Marecki JC, Cool CD, Parr JE, Beckey VE, Luciw PA, Tarantal AF, Carville A, Shannon RP, Cota-Gomez A, Tuder RM et al (2006) HIV-1 Nef is associated with complex pulmonary vascular lesions in SHIV-nef-infected macaques. Am J Respir Crit Care Med 174:437–445
McLaughlin VV, Presberg KW, Doyle RL, Abman SH, McCrory DC, Fortin T, Ahearn G (2004) Prognosis of pulmonary arterial hypertension: ACCP evidence-based clinical practice guidelines. Chest 126:78S–92S
Soon E, Holmes AM, Treacy CM, Doughty NJ, Southgate L, Machado RD, Trembath RC, Jennings S, Barker L, Nicklin P et al (2010) Elevated levels of inflammatory cytokines predict survival in idiopathic and familial pulmonary arterial hypertension. Circulation 122:920–927
Zell R, Geck P, Werdan K, Boekstegers P (1997) TNF-alpha and IL-1 alpha inhibit both pyruvate dehydrogenase activity and mitochondrial function in cardiomyocytes: evidence for primary impairment of mitochondrial function. Mol Cell Biochem 177:61–67
Atkinson EA, Barry M, Darmon AJ, Shostak I, Turner PC, Moyer RW, Bleackley RC (1998) Cytotoxic T lymphocyte-assisted suicide. Caspase 3 activation is primarily the result of the direct action of granzyme B. J Biol Chem 273:21261–21266
Bowker-Kinley MM, Davis WI, Wu P, Harris RA, Popov KM (1998) Evidence for existence of tissue-specific regulation of the mammalian pyruvate dehydrogenase complex. Biochem J 329(Pt 1):191–196
Barath P, Fishbein MC, Cao J, Berenson J, Helfant RH, Forrester JS (1990) Tumor necrosis factor gene expression in human vascular intimal smooth muscle cells detected by in situ hybridization. Am J Pathol 137:503–509
Wilson DW, Segall HJ, Pan LC, Dunston SK (1989) Progressive inflammatory and structural changes in the pulmonary vasculature of monocrotaline-treated rats. Microvasc Res 38:57–80
Lovell DJ, Giannini EH, Reiff A, Cawkwell GD, Silverman ED, Nocton JJ, Stein LD, Gedalia A, Ilowite NT, Wallace CA et al (2000) Etanercept in children with polyarticular juvenile rheumatoid arthritis. Pediatric Rheumatology Collaborative Study Group. N Engl J Med 342:763–769
Bonnet S, Michelakis ED, Porter CJ, Andrade-Navarro MA, Thebaud B, Bonnet S, Haromy A, Harry G, Moudgil R, McMurtry MS et al (2006) An abnormal mitochondrial-hypoxia inducible factor-1alpha-Kv channel pathway disrupts oxygen sensing and triggers pulmonary arterial hypertension in fawn hooded rats: similarities to human pulmonary arterial hypertension. Circulation 113:2630–2641
Papandreou I, Cairns RA, Fontana L, Lim AL, Denko NC (2006) HIF-1 mediates adaptation to hypoxia by actively downregulating mitochondrial oxygen consumption. Cell Metab 3:187–197
Daley E, Emson C, Guignabert C, de Waal MR, Louten J, Kurup VP, Hogaboam C, Taraseviciene-Stewart L, Voelkel NF, Rabinovitch M et al (2008) Pulmonary arterial remodeling induced by a Th2 immune response. J Exp Med 205:361–372
Henriques-Coelho T, Brandao-Nogueira A, Moreira-Goncalves D, Correia-Pinto J, Leite-Moreira AF (2008) Effects of TNF-alpha blockade in monocrotaline-induced pulmonary hypertension. Rev Port Cardiol 27:341–348
Bargagli E, Galeazzi M, Bellisai F, Volterrani L, Rottoli P (2008) Infliximab treatment in a patient with systemic sclerosis associated with lung fibrosis and pulmonary hypertension. Respiration 75:346–349
Fujita M, Shannon JM, Irvin CG, Fagan KA, Cool C, Augustin A, Mason RJ (2001) Overexpression of tumor necrosis factor-alpha produces an increase in lung volumes and pulmonary hypertension. Am J Physiol Lung Cell Mol Physiol 280:L39–L49
Voelkel NF, Tuder RM, Bridges J, Arend WP (1994) Interleukin-1 receptor antagonist treatment reduces pulmonary hypertension generated in rats by monocrotaline. Am J Respir Cell Mol Biol 11:664–675
Clausell N, Molossi S, Sett S, Rabinovitch M (1994) In vivo blockade of tumor necrosis factor-alpha in cholesterol-fed rabbits after cardiac transplant inhibits acute coronary artery neointimal formation. Circulation 89:2768–2779
Stevens T, Phan S, Frid MG, Alvarez D, Herzog E, Stenmark KR (2008) Lung vascular cell heterogeneity: endothelium, smooth muscle, and fibroblasts. Proc Am Thorac Soc 5:783–791
Xu W, Koeck T, Lara AR, Neumann D, DiFilippo FP, Koo M, Janocha AJ, Masri FA, Arroliga AC, Jennings C et al (2007) Alterations of cellular bioenergetics in pulmonary artery endothelial cells. Proc Natl Acad Sci USA 104:1342–1347
Disclosures
The authors have no conflicts to disclose.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(PDF 9.22 MB)
Rights and permissions
About this article
Cite this article
Sutendra, G., Dromparis, P., Bonnet, S. et al. Pyruvate dehydrogenase inhibition by the inflammatory cytokine TNFα contributes to the pathogenesis of pulmonary arterial hypertension. J Mol Med 89, 771–783 (2011). https://doi.org/10.1007/s00109-011-0762-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00109-011-0762-2