Abstract
Introduction
Tissue injury (TI) and hemorrhagic shock (HS) are the major contributors to trauma-induced coagulopathy (TIC). However, the individual contributions of these insults are difficult to discern clinically because they typically coexist. TI has been reported to release procoagulants, while HS has been associated with bleeding. We developed a large animal model to isolate TI and HS and characterize their individual mechanistic pathways. We hypothesized that while TI and HS are both drivers of TIC, they provoke different pathways; specifically, TI reduces time to clotting, whereas, HS decreases clot strength stimulates hyperfibrinolysis.
Methods
After induction of general anesthesia, 50 kg male, Yorkshire swine underwent isolated TI (bilateral muscle cutdown of quadriceps, bilateral femur fractures) or isolated HS (controlled bleeding to a base excess target of − 5 mmol/l) and observed for 240 min. Thrombelastography (TEG), calcium levels, thrombin activatable fibrinolysis inhibitor (TAFI), protein C, plasminogen activator inhibitor 1 (PAI-1), and plasminogen activator inhibitor 1/tissue-type plasminogen activator complex (PAI-1-tPA) were analyzed at pre-selected timepoints. Linear mixed models for repeated measures were used to compare results throughout the model.
Results
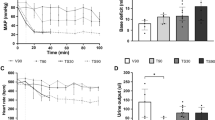
TI resulted in elevated histone release which peaked at 120 min (p = 0.02), and this was associated with reduced time to clot formation (R time) by 240 min (p = 0.006). HS decreased clot strength at time 30 min (p = 0.003), with a significant decline in calcium (p = 0.001). At study completion, HS animals had elevated PAI-1 (p = 0.01) and PAI-1-tPA (p = 0.04), showing a trend toward hyperfibrinolysis, while TI animals had suppressed fibrinolysis. Protein C, TAFI and skeletal myosin were not different among the groups.
Conclusion
Isolated injury in animal models can help elucidate the mechanistic pathways leading to TIC. Our results suggest that isolated TI leads to early histone release and a hypercoagulable state, with suppressed fibrinolysis. In contrast, HS promotes poor clot strength and hyperfibrinolysis resulting in hypocoagulability.




Similar content being viewed by others
References
Kilkenny C, Browne W, Cuthill IC, Emerson M, Altman DG, Group NCRRGW. Animal research: reporting in vivo experiments: the ARRIVE guidelines. Br J Pharmacol. 2010;160(7):1577–9. https://doi.org/10.1111/j.1476-5381.2010.00872.x.
World Health Organization. Fact Sheets: Injuries and violence. 2021. https://www.who.int/news-room/fact-sheets/detail/injuries-and-violence#:~:text=Injuries%20%E2%80%93%20both%20unintentional%20and%20violence,nearly%208%25%20of%20all%20deaths. Accessed 11 Jun 2022.
Kalkwarf KJ, Drake SA, Yang Y, et al. Bleeding to death in a big city: An analysis of all trauma deaths from hemorrhage in a metropolitan area during 1 year. J Trauma Acute Care Surg. 2020;89(4):716–22. https://doi.org/10.1097/TA.0000000000002833.
Moore HB, Moore EE, Chapman MP, et al. Plasma-first resuscitation to treat haemorrhagic shock during emergency ground transportation in an urban area: a randomised trial. Lancet. 2018;392(10144):283–91. https://doi.org/10.1016/S0140-6736(18)31553-8.
Brohi K, Singh J, Heron M, Coats T. Acute traumatic coagulopathy. J Trauma. 2003;54(6):1127–30. https://doi.org/10.1097/01.TA.0000069184.82147.06.
Cosgriff N, Moore EE, Sauaia A, Kenny-Moynihan M, Burch JM, Galloway B. Predicting life-threatening coagulopathy in the massively transfused trauma patient: hypothermia and acidoses revisited. J Trauma. 1997;42(5):857–61. https://doi.org/10.1097/00005373-199705000-00016 (Discussion 861-2).
Moore EE, Moore HB, Kornblith LZ, et al. Trauma-induced coagulopathy. Nat Rev Dis Primers. 2021;7(1):30. https://doi.org/10.1038/s41572-021-00264-3.
Frith D, Goslings JC, Gaarder C, et al. Definition and drivers of acute traumatic coagulopathy: clinical and experimental investigations. J Thromb Haemost. 2010;8(9):1919–25. https://doi.org/10.1111/j.1538-7836.2010.03945.x.
Kutcher ME, Howard BM, Sperry JL, et al. Evolving beyond the vicious triad: differential mediation of traumatic coagulopathy by injury, shock, and resuscitation. J Trauma Acute Care Surg. 2015;78(3):516–23. https://doi.org/10.1097/TA.0000000000000545.
Martini WZ, Pusateri AE, Uscilowicz JM, Delgado AV, Holcomb JB. Independent contributions of hypothermia and acidosis to coagulopathy in swine. J Trauma. 2005;58(5):1002–9. https://doi.org/10.1097/01.ta.0000156246.53383.9f (Discussion 1009-10).
Johansson PI, Stensballe J, Rasmussen LS, Ostrowski SR. A high admission syndecan-1 level, a marker of endothelial glycocalyx degradation, is associated with inflammation, protein C depletion, fibrinolysis, and increased mortality in trauma patients. Ann Surg. 2011;254(2):194–200. https://doi.org/10.1097/SLA.0b013e318226113d.
Johansson PI, Windelov NA, Rasmussen LS, Sorensen AM, Ostrowski SR. Blood levels of histone-complexed DNA fragments are associated with coagulopathy, inflammation and endothelial damage early after trauma. J Emerg Trauma Shock. 2013;6(3):171–5. https://doi.org/10.4103/0974-2700.115327.
Moore HB, Moore EE, Gonzalez E, et al. Hyperfibrinolysis, physiologic fibrinolysis, and fibrinolysis shutdown: the spectrum of postinjury fibrinolysis and relevance to antifibrinolytic therapy. J Trauma Acute Care Surg. 2014;77(6):811–7. https://doi.org/10.1097/TA.0000000000000341 (Discussion 817).
Moore HB, Moore EE. Temporal Changes in Fibrinolysis following Injury. Semin Thromb Hemost. 2020;46(2):189–98. https://doi.org/10.1055/s-0039-1701016.
Moore HB, Moore EE, Huebner BR, et al. Fibrinolysis shutdown is associated with a fivefold increase in mortality in trauma patients lacking hypersensitivity to tissue plasminogen activator. J Trauma Acute Care Surg. 2017;83(6):1014–22. https://doi.org/10.1097/TA.0000000000001718.
Davenport RA, Guerreiro M, Frith D, et al. Activated protein C drives the hyperfibrinolysis of acute traumatic coagulopathy. Anesthesiology. 2017;126(1):115–27. https://doi.org/10.1097/ALN.0000000000001428.
Moore HB, Moore EE, Lawson PJ, et al. Fibrinolysis shutdown phenotype masks changes in rodent coagulation in tissue injury versus hemorrhagic shock. Surgery. 2015;158(2):386–92. https://doi.org/10.1016/j.surg.2015.04.008.
Seok J, Warren HS, Cuenca AG, et al. Genomic responses in mouse models poorly mimic human inflammatory diseases. Proc Natl Acad Sci USA. 2013;110(9):3507–12. https://doi.org/10.1073/pnas.1222878110.
Cralley AL, Moore EE, Kissau D, Coleman JR, Vigneshwa N, Debot M, Schaid TR Jr, Moore HB, Cohen MJ, Hansen K, Silliman CC. A combat casualty relevant dismounted complex blast injury model in swine. J Trauma Acute Care Surg. 2022;93(2S Suppl 1):S110-S118. https://doi.org/10.1097/TA.0000000000003674
Stettler GR, Moore EE, Moore HB, et al. Thrombelastography indicates limitations of animal models of trauma-induced coagulopathy. J Surg Res. 2017;217:207–12. https://doi.org/10.1016/j.jss.2017.05.027.
D’Alessandro A, Dzieciatkowska M, Hill RC, Hansen KC. Supernatant protein biomarkers of red blood cell storage hemolysis as determined through an absolute quantification proteomics technology. Transfusion. 2016;56(6):1329–39. https://doi.org/10.1111/trf.13483.
Dzieciatkowska M, D’Alessandro A, Hill RC, Hansen KC. Plasma QconCATs reveal a gender-specific proteomic signature in apheresis platelet plasma supernatants. J Proteomics. 2015;120:1–6. https://doi.org/10.1016/j.jprot.2015.02.010.
The UniProt Consortium. UniProt: the universal protein knowledgebase. Nucleic Acids Res. 2018;46(5):2699. https://doi.org/10.1093/nar/gky092.
Coleman JR, Deguchi H, Deguchi TK, Cohen MJ, Moore EE, Griffin JH. Full-length plasma skeletal muscle myosin isoform deficiency is associated with coagulopathy in acutely injured patients. J Thromb Haemost. 2022. https://doi.org/10.1111/jth.15695.
Chapman MP, Moore EE, Ramos CR, et al. Fibrinolysis greater than 3% is the critical value for initiation of antifibrinolytic therapy. J Trauma Acute Care Surg. 2013;75(6):961–7. https://doi.org/10.1097/TA.0b013e3182aa9c9f (Discussion 967).
Esmon CT. The protein C pathway. Chest. 2003;124(3 Suppl):26S-32S. https://doi.org/10.1378/chest.124.3_suppl.26s.
de Fouw NJ, de Jong YF, Haverkate F, Bertina RM. Activated protein C increases fibrin clot lysis by neutralization of plasminogen activator inhibitor–no evidence for a cofactor role of protein S. Thromb Haemost. 1988;60(2):328–33.
Brohi K, Cohen MJ, Ganter MT, Matthay MA, Mackersie RC, Pittet JF. Acute traumatic coagulopathy: initiated by hypoperfusion: modulated through the protein C pathway? Ann Surg. 2007;245(5):812–8. https://doi.org/10.1097/01.sla.0000256862.79374.31.
Chesebro BB, Rahn P, Carles M, et al. Increase in activated protein C mediates acute traumatic coagulopathy in mice. Shock. 2009;32(6):659–65. https://doi.org/10.1097/SHK.0b013e3181a5a632.
Moore HB, Tessmer MT, Moore EE, et al. Forgot calcium? Admission ionized-calcium in two civilian randomized controlled trials of prehospital plasma for traumatic hemorrhagic shock. J Trauma Acute Care Surg. 2020;88(5):588–96. https://doi.org/10.1097/TA.0000000000002614.
Matthay ZA, Fields AT, Nunez-Garcia B, et al. Dynamic effects of calcium on in vivo and ex vivo platelet behavior after trauma. J Trauma Acute Care Surg. 2020;89(5):871–9. https://doi.org/10.1097/TA.0000000000002820.
Singh S, Dodt J, Volkers P, et al. Structure functional insights into calcium binding during the activation of coagulation factor XIII A. Sci Rep. 2019;9(1):11324. https://doi.org/10.1038/s41598-019-47815-z.
Brannstrom A, von Oelreich E, Degerstedt LE, et al. The swine as a vehicle for research in trauma-induced coagulopathy: Introducing principal component analysis for viscoelastic coagulation tests. J Trauma Acute Care Surg. 2021;90(2):360–8. https://doi.org/10.1097/TA.0000000000002997.
Schochl H, Solomon C, Schulz A, et al. Thromboelastometry (TEM) findings in disseminated intravascular coagulation in a pig model of endotoxinemia. Mol Med. 2011;17(3–4):266–72. https://doi.org/10.2119/molmed.2010.00159.
Saetre T, Lindgaard AK, Lyberg T. Systemic activation of coagulation and fibrynolysis in a porcine model of serogroup A streptococcal shock. Blood Coagul Fibrinolysis. 2000;11(5):433–8. https://doi.org/10.1097/00001721-200007000-00006.
Chandler WL, Trimble SL, Loo SC, Mornin D. Effect of PAI-1 levels on the molar concentrations of active tissue plasminogen activator (t-PA) and t-PA/PAI-1 complex in plasma. Blood. 1990;76(5):930–7.
Bjorquist P, Brohlin M, Ehnebom J, et al. Plasminogen activator inhibitor type-1 interacts exclusively with the proteinase domain of tissue plasminogen activator. Biochim Biophys Acta. 1994;1209(2):191–202. https://doi.org/10.1016/0167-4838(94)90184-8.
Cardenas JC, Matijevic N, Baer LA, Holcomb JB, Cotton BA, Wade CE. Elevated tissue plasminogen activator and reduced plasminogen activator inhibitor promote hyperfibrinolysis in trauma patients. Shock. 2014;41(6):514–21. https://doi.org/10.1097/SHK.0000000000000161.
Chapman MP, Moore EE, Moore HB, et al. Overwhelming tPA release, not PAI-1 degradation, is responsible for hyperfibrinolysis in severely injured trauma patients. J Trauma Acute Care Surg. 2016;80(1):16–23. https://doi.org/10.1097/TA.0000000000000885 (Discussion 23-5).
Chandler WL, Alessi MC, Aillaud MF, Henderson P, Vague P, Juhan-Vague I. Clearance of tissue plasminogen activator (TPA) and TPA/plasminogen activator inhibitor type 1 (PAI-1) complex: relationship to elevated TPA antigen in patients with high PAI-1 activity levels. Circulation. 1997;96(3):761–8. https://doi.org/10.1161/01.cir.96.3.761.
Coleman JR, Moore EE, Zilberman-Rudenko J, et al. Cardiac and skeletal muscle myosin exert procoagulant effects. Shock. 2019;52(5):554–5. https://doi.org/10.1097/SHK.0000000000001426.
Deguchi H, Sinha RK, Marchese P, et al. Prothrombotic skeletal muscle myosin directly enhances prothrombin activation by binding factors Xa and Va. Blood. 2016;128(14):1870–8. https://doi.org/10.1182/blood-2016-03-707679.
Vlasakova K, Lane P, Michna L, Muniappa N, Sistare FD, Glaab WE. Response of novel skeletal muscle biomarkers in dogs to drug-induced skeletal muscle injury or sustained endurance exercise. Toxicol Sci. 2017;156(2):422–7. https://doi.org/10.1093/toxsci/kfw262.
Burch PM, Greg Hall D, Walker EG, et al. Evaluation of the relative performance of drug-induced skeletal muscle injury biomarkers in rats. Toxicol Sci. 2016;150(1):247–56. https://doi.org/10.1093/toxsci/kfv328.
Guerrero M, Guiu-Comadevall M, Cadefau JA, et al. Fast and slow myosins as markers of muscle injury. Br J Sports Med. 2008;42(7):581–4. https://doi.org/10.1136/bjsm.2007.037945 (Discussion 584).
Kutcher ME, Xu J, Vilardi RF, Ho C, Esmon CT, Cohen MJ. Extracellular histone release in response to traumatic injury: implications for a compensatory role of activated protein C. J Trauma Acute Care Surg. 2012;73(6):1389–94. https://doi.org/10.1097/TA.0b013e318270d595.
Ammollo CT, Semeraro F, Xu J, Esmon NL, Esmon CT. Extracellular histones increase plasma thrombin generation by impairing thrombomodulin-dependent protein C activation. J Thromb Haemost. 2011;9(9):1795–803. https://doi.org/10.1111/j.1538-7836.2011.04422.x.
Zeerleder S, Zwart B, Wuillemin WA, et al. Elevated nucleosome levels in systemic inflammation and sepsis. Crit Care Med. 2003;31(7):1947–51. https://doi.org/10.1097/01.CCM.0000074719.40109.95.
Brinkmann V, Reichard U, Goosmann C, et al. Neutrophil extracellular traps kill bacteria. Science. 2004;303(5663):1532–5. https://doi.org/10.1126/science.1092385.
Xu J, Zhang X, Pelayo R, et al. Extracellular histones are major mediators of death in sepsis. Nat Med. 2009;15(11):1318–21. https://doi.org/10.1038/nm.2053.
Kleine TJ, Lewis PN, Lewis SA. Histone-induced damage of a mammalian epithelium: the role of protein and membrane structure. Am J Physiol. 1997;273(6):C1925–36. https://doi.org/10.1152/ajpcell.1997.273.6.C1925.
Kleine TJ, Gladfelter A, Lewis PN, Lewis SA. Histone-induced damage of a mammalian epithelium: the conductive effect. Am J Physiol. 1995;268(5 Pt 1):C1114–25. https://doi.org/10.1152/ajpcell.1995.268.5.C1114.
Xu J, Zhang X, Monestier M, Esmon NL, Esmon CT. Extracellular histones are mediators of death through TLR2 and TLR4 in mouse fatal liver injury. J Immunol. 2011;187(5):2626–31. https://doi.org/10.4049/jimmunol.1003930.
Parr MJ, Bouillon B, Brohi K, et al. Traumatic coagulopathy: where are the good experimental models? J Trauma. 2008;65(4):766–71. https://doi.org/10.1097/TA.0b013e31818606d2.
Ask A, Eltringham-Smith L, Bhakta V, Donkor DA, Pryzdial ELG, Sheffield WP. Spotlight on animal models of acute traumatic coagulopathy: an update. Transfus Apher Sci. 2022;61(2): 103412. https://doi.org/10.1016/j.transci.2022.103412.
Reynolds PS, Fisher BJ, McCarter J, et al. Interventional vitamin C: a strategy for attenuation of coagulopathy and inflammation in a swine multiple injuries model. J Trauma Acute Care Surg. 2018;85(1S Suppl 2):S57–67. https://doi.org/10.1097/TA.0000000000001844.
Martini WZ, Rodriguez CM, Cap AP, Dubick MA. Efficacy of resuscitation with fibrinogen concentrate and platelets in traumatic hemorrhage swine model. J Trauma Acute Care Surg. 2020;89(2S Suppl 2):S137–45. https://doi.org/10.1097/TA.0000000000002736.
Spronk HM, Braunschweig T, Rossaint R, et al. Recombinant factor VIIa reduces bleeding after blunt liver injury in a pig model of dilutional coagulopathy under severe hypothermia. PLoS One. 2015;10(6): e0113979. https://doi.org/10.1371/journal.pone.0113979.
Hansson KM, Johansson KJ, Wingren C, Fries D, Nelander K, Lovgren A. Recombinant human prothrombin reduced blood loss in a porcine model of dilutional coagulopathy with uncontrolled bleeding. Blood Coagul Fibrinolysis. 2017;28(3):244–53. https://doi.org/10.1097/MBC.0000000000000590.
Kutcher ME, Kornblith LZ, Narayan R, et al. A paradigm shift in trauma resuscitation: evaluation of evolving massive transfusion practices. JAMA Surg. 2013;148(9):834–40. https://doi.org/10.1001/jamasurg.2013.2911.
Funding
This research is funded in part by the Trans-Agency Consortium for Trauma-Induced Coagulopathy (TACTIC) contract number UM1-HL120877. Research support is also provided by the National Institute of General Medical Sciences of the National Institutes of Health (T32 GM008315). The current major funding source is an RM1 grant (1RM1GM131968-01). The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
E.E.M. has patents pending related to coagulation and fibrinolysis diagnostics and therapeutic fibrinolytics and is a cofounder with stock options in ThromboTherepeutics. E.E.M. has received grant support from Haemonetics, Inc., Stago, Hemosonics, Instrumentation Laboratories, Inc, and Diapharma outside the submitted work.
Ethical approval
This animal study was conducted in compliance with the Animal Welfare Act, implementing Animal Welfare Regulations, and the principles of the Guide for the Care and Use of Laboratory Animals, National Research Council. Results are reported in accordance with the ARRIVE guidelines [1]. The University of Colorado Institutional Animal Care and Use Committee approved this animal study under protocol #0323, and the research was conducted in a fully accredited Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) facility.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cralley, A.L., Moore, E.E., Coleman, J.R. et al. Hemorrhagic shock and tissue injury provoke distinct components of trauma-induced coagulopathy in a swine model. Eur J Trauma Emerg Surg 49, 1079–1089 (2023). https://doi.org/10.1007/s00068-022-02148-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-022-02148-x