Abstract
Introduction
The clinical success of total knee arthroplasty (TKA) depends substantially on the quadriceps muscle function. A frequently applied thigh tourniquet during TKA may induce ischemia related injuries to quadriceps muscle cells. Animal limb muscles subjected to 2–5 h ischemia revealed dysfunctional mitochondria, which in turn compromised the cellular bioenergetics and increased the level of reactive oxygen species. The hypothesis of the present study was that tourniquet application during TKA for 60 min (min) affects the amount and function of mitochondria within musculus vastus medialis cells.
Materials and methods
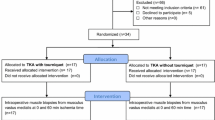
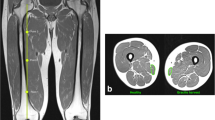
In a randomized clinical trial, 10 patients enrolled to undergo primary TKA. The patients were randomly assigned to the tourniquet (n = 5) or non-tourniquet group (n = 5) after obtaining a written informed consent. For each of the groups, the first muscle biopsy was harvested immediately after performing the surgical approach and the second biopsy exactly 60 min later. All biopsies (5 × 5 × 5 mm) 125 mm3 were harvested from musculus vastus medialis and snap-frozen in liquid nitrogen. The biochemical analysis of the prepared muscle tissues included the measurement of activities of mitochondrial respiratory chain enzyme complexes I–III and citrate synthase.
Results
Tourniquet-induced 60 min ischemia time did not significantly change the activities of the mitochondrial respiratory chain enzymes complexes I–III of the skeletal muscle cells. The citrate synthase activities found to be not significantly different between both groups.
Conclusions
The use of tourniquet during TKA within a limited time period of 60 min remained without substantial effects on the amount and function of mitochondria within human skeletal muscle cells.





Similar content being viewed by others
References
Hammers DW, Merritt EK, Matheny RW Jr, Adamo ML, Walters TJ, Estep JS, et al. Functional deficits and insulin-like growth factor-I gene expression following tourniquet-induced injury of skeletal muscle in young and old rats. J Appl Physiol. 2008;105(4):1274–81.
Tai TW, Chang CW, Lai KA, Lin CJ, Yang CY. Effects of tourniquet use on blood loss and soft-tissue damage in total knee arthroplasty: a randomized controlled trial. J Bone Joint Surg Am Vol. 2012;94(24):2209–15.
Finch E, Walsh M, Thomas SG, Woodhouse LJ. Functional ability perceived by individuals following total knee arthroplasty compared to age-matched individuals without knee disability. J Orthop Sports Phys Ther. 1998;27(4):255–63.
Mizner RL, Petterson SC, Snyder-Mackler L. Quadriceps strength and the time course of functional recovery after total knee arthroplasty. J Orthop Sports Phys Ther. 2005;35(7):424–36.
Mizner RL, Petterson SC, Stevens JE, Axe MJ, Snyder-Mackler L. Preoperative quadriceps strength predicts functional ability 1 year after total knee arthroplasty. J Rheumatol. 2005;32(8):1533–9.
Mizner RL, Petterson SC, Stevens JE, Vandenborne K, Snyder-Mackler L. Early quadriceps strength loss after total knee arthroplasty. The contributions of muscle atrophy and failure of voluntary muscle activation. J Bone Joint Surg Am Vol. 2005;87(5):1047–53.
Yoshida Y, Mizner RL, Ramsey DK, Snyder-Mackler L. Examining outcomes from total knee arthroplasty and the relationship between quadriceps strength and knee function over time. Clin Biomech. 2008;23(3):320–8.
Walsh M, Woodhouse LJ, Thomas SG, Finch E. Physical impairments and functional limitations: a comparison of individuals 1 year after total knee arthroplasty with control subjects. Phys Ther. 1998;78(3):248–58.
Westman B, Weidenhielm L, Rooyackers O, Fredriksson K, Wernerman J, Hammarqvist F. Knee replacement surgery as a human clinical model of the effects of ischaemia/reperfusion upon skeletal muscle. Clin Sci. 2007;113(7):313–8.
Vogt MT, Farber E. On the molecular pathology of ischemic renal cell death. Reversible and irreversible cellular and mitochondrial metabolic alterations. Am J Pathol. 1968;53(1):1–26.
Jennings RB. Relationship of acute ischemia to functional defects and irreversibility. Circulation. 1976;53(3 Suppl):I26–9.
Trump BF, Mergner WJ, Kahng MW, Salandino AJ. Studies on the subcellular pathophysiology of ischemia. Circulation. 1976;53(3):I17–26.
Mittnacht S Jr, Sherman SC, Farber JL. Reversal of ischemic mitochondrial dysfunction. J Biol Chem. 1979;254(19):9871–8.
Thaveau F, Zoll J, Bouitbir J, Ribera F, Di Marco P, Chakfe N, et al. Contralateral leg as a control during skeletal muscle ischemia-reperfusion. J Surg Res. 2009;155(1):65–9.
Brandao ML, Roselino JE, Piccinato CE, Cherri J. Mitochondrial alterations in skeletal muscle submitted to total ischemia. J Surg Res. 2003;110(1):235–40.
Pipinos II, Sharov VG, Shepard AD, Anagnostopoulos PV, Katsamouris A, Todor A, et al. Abnormal mitochondrial respiration in skeletal muscle in patients with peripheral arterial disease. J Vasc Surg. 2003;38(4):827–32.
Pipinos II, Judge AR, Zhu Z, Selsby JT, Swanson SA, Johanning JM, et al. Mitochondrial defects and oxidative damage in patients with peripheral arterial disease. Free Radic Biol Med. 2006;41(2):262–9.
Moses MA, Addison PD, Neligan PC, Ashrafpour H, Huang N, McAllister SE, et al. Inducing late phase of infarct protection in skeletal muscle by remote preconditioning: efficacy and mechanism. Am J Physiol Regul Integr Comp Physiol. 2005;289(6):R1609–17.
Pipinos II, Swanson SA, Zhu Z, Nella AA, Weiss DJ, Gutti TL, et al. Chronically ischemic mouse skeletal muscle exhibits myopathy in association with mitochondrial dysfunction and oxidative damage. Am J Physiol Regul Integr Comp Physiol. 2008;295(1):R290–6.
Gillani S, Cao J, Suzuki T, Hak DJ. The effect of ischemia reperfusion injury on skeletal muscle. Injury. 2012;43(6):670–5.
Chafin B, Belmont MJ, Quraishi H, Clovis N, Wax MK. Effect of clamp versus anastomotic-induced ischemia on critical ischemic time and survival of rat epigastric fasciocutaneous flap. Head Neck. 1999;21(3):198–203.
Schild L, Jaroscakova I, Lendeckel U, Wolf G, Keilhoff G. Neuronal nitric oxide synthase controls enzyme activity pattern of mitochondria and lipid metabolism. FASEB J. 2006;20(1):145–7.
Birch-Machin MA, Briggs HL, Saborido AA, Bindoff LA, Turnbull DM. An evaluation of the measurement of the activities of complexes I-IV in the respiratory chain of human skeletal muscle mitochondria. Biochem Med Metab Biol. 1994;51(1):35–42.
Appell HJ, Gloser S, Duarte JA, Zellner A, Soares JM. Skeletal muscle damage during tourniquet-induced ischaemia. The initial step towards atrophy after orthopaedic surgery? Eur J Appl Physiol. 1993;67(4):342–7.
Sapega AA, Heppenstall RB, Chance B, Park YS, Sokolow D. Optimizing tourniquet application and release times in extremity surgery. A biochemical and ultrastructural study. J Bone Joint Surg Am Vol. 1985;67(2):303–14.
Heffner RR, Barron SA. The early effects of ischemia upon skeletal muscle mitochondria. J Neurol Sci. 1978;38(3):295–315.
Schroll R, Smith A, McSwain NE, Jr Myers J, Rocchi K, Inaba K, et al. A multi-institutional analysis of prehospital tourniquet use. J Trauma Acute Care Surg. 2015;79(1):10–4 discussion 4.
Surgeons ACO. ATLS: advanced trauma life support for doctors. 8th ed. Chicago: American College of Surgeons; 2008.
Li B, Wen Y, Wu H, Qian Q, Lin X, Zhao H. The effect of tourniquet use on hidden blood loss in total knee arthroplasty. Int Orthop. 2009;33(5):1263–8.
Liu D, Graham D, Gillies K, Gillies RM. Effects of tourniquet use on quadriceps function and pain in total knee arthroplasty. Knee Surg Relat Res. 2014;26(4):207–13.
Harsten A, Bandholm T, Kehlet H, Toksvig-Larsen S. Tourniquet versus no tourniquet on knee-extension strength early after fast-track total knee arthroplasty; a randomized controlled trial. Knee. 2015;22(2):126–30.
Acknowledgments
We gratefully acknowledge excellent technical assistance by Mrs. Silke Niemann.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
The study was performed in accordance with the Declaration of Helsinki after receiving the institutional Ethics Committee approval (File reference 2012-334 N-MA).
Conflicts of interest
All authors (Ahmed Jawhar, Norbert Ponelies and Lorenz Schild) declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Jawhar, A., Ponelies, N. & Schild, L. Effect of limited ischemia time on the amount and function of mitochondria within human skeletal muscle cells. Eur J Trauma Emerg Surg 42, 767–773 (2016). https://doi.org/10.1007/s00068-015-0600-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00068-015-0600-2