Abstract
Background
Preoperative radiotherapy (PRT) or radiochemotherapy (PRCT) is used in different tumor sites. The aim of the study was to examine the long-term quality of life (QoL) of localized / locally advanced breast cancer patients treated with PRT/PRCT followed by breast-conserving surgery (BCS) or mastectomy (ME).
Methods
Assessment of QoL was done using EORTC QLQ-C30 questionnaires for overall QoL and EORTC QLQ-BR23 for breast-specific QoL. The summary scores were categorized into 4 distinct groups to classify the results. Furthermore, a comparative analysis was performed between the study cohort and a previously published reference cohort of healthy adults. We assessed the impact of different clinical, prognostic, and treatment-related factors on selected items from C30 and BR23 using a dependence analysis.
Results
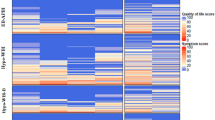
Out of 315 patients treated with PRT/PCRT in the years 1991 to 1999, 203 patients were alive at long-term follow-up after a mean of 17.7 years (range 14–21). 37 patients were lost to follow-up and 61 patients refused to be contacted, leading to 105 patients (64 patients after BCS and 41 after ME) being willing to undergo further clinical assessment regarding QoL outcome. Overall, QoL (QLQ-C30) was rated “excellent” or “good” in 85% (mean value) of all patients (BCS 83%, ME 88%). Comparative analysis between the study cohort and a published healthy control group revealed significantly better global health status and physical and role functioning scores in the PRT/PRCT group. The analysis demonstrates no differences in nausea/vomiting, dyspnea, insomnia, constipation, or financial difficulties. According to the dependence analysis, global QoL was associated with age, operation type and ME reconstruction.
Conclusion
We did not detect any inferiority of PRT/PRCT compared to a healthy reference group with no hints of a detrimental long-term effect on general and breast-specific quality of life.


Similar content being viewed by others
Abbreviations
- BCS:
-
Breast-conserving surgery
- BCT:
-
Breast-conserving treatment
- BMI:
-
Body mass index
- CI:
-
Confidence interval
- DFS:
-
Disease-free survival
- FU:
-
Follow-up
- HR:
-
Hazard ratio
- ME:
-
Mastectomy
- n.v.:
-
No value
- n:
-
Number
- OS:
-
Overall survival
- pCR:
-
Pathological complete response
- PCRT:
-
Preoperative radiochemotherapy
- QLQ:
-
Quality of life questionnaire
- QoL:
-
Quality of life
- RR:
-
Risk ratio
- RT:
-
Radiotherapy
- SAS:
-
Statistical analysis system
- w:
-
With
- w/o:
-
Without
References
Budach W, Bolke E, Kammers K, Gerber PA, Nestle-Kramling C, Matuschek C (2015) Adjuvant radiation therapy of regional lymph nodes in breast cancer—a meta-analysis of randomized trials—an update. Radiat Oncol 10:258
Budach W, Kammers K, Boelke E, Matuschek C (2013) Adjuvant radiotherapy of regional lymph nodes in breast cancer—A meta-analysis of randomized trials. Radiat Oncol 8:267
Budach W, Bolke E, Matuschek C (2015) Hypofractionated radiotherapy as adjuvant treatment in early breast cancer. A review and meta-analysis of randomized controlled trials. Breast Care 10:240–245
Fokas E, Conradi L, Weiss C et al (2013) Preoperative chemoradiation therapy with capecitabine/oxaliplatin and cetuximab in rectal cancer: long-term results of a prospective phase 1/2 study. Int J Radiat Oncol Biol Phys 87:992–999
Nestle-Kramling C, Bolke E, Budach W, Andree C (2016) Breast reconstruction after neoadjuvant radio chemotherapy: Review and personal technique IDEAL concept REV-EJMR-D-15-00268. Eur J Med Res 21:24
Cocquyt VF, Blondeel PN, Depypere HT et al (2003) Better cosmetic results and comparable quality of life after skin-sparing mastectomy and immediate autologous breast reconstruction compared to breast conservative treatment. Br J Plast Surg 56:462–470
Skinner KA, Dunnington G, Silberman H, Florentine B, Spicer D, Formenti SC (1997) Preoperative 5‑fluorouracil and radiation therapy for locally advanced breast cancer. Am J Surg 174:705–707 (discussion 7–8)
Bollet MA, Belin L, Reyal F et al (2012) Preoperative radio-chemotherapy in early breast cancer patients: long-term results of a phase II trial. Radiother Oncol 102:82–88
Calitchi E, Kirova YM, Otmezguine Y, Feuilhade F, Piedbois Y, Le Bourgeois JP (2001) Long-term results of neoadjuvant radiation therapy for breast cancer. Int J Cancer 96:253–259
Riet FG, Fayard F, Arriagada R et al (2017) Preoperative radiotherapy in breast cancer patients: 32 years of follow-up. Eur J Cancer 76:45–51
Woodward WA, Fang P, Arriaga L et al (2017) A phase 2 study of capecitabine and concomitant radiation in women with advanced breast cancer. Int J Radiat Oncol Biol Phys 99:777–783
Brackstone M, Palma D, Tuck AB et al (2017) Concurrent neoadjuvant chemotherapy and radiation therapy in locally advanced breast cancer. Int J Radiat Oncol Biol Phys 99:769–776
Coelho RC, Da Silva FML, Do Carmo IML, Bonaccorsi BV, Hahn SM, Faroni LD (2017) Is there a role for salvage radiotherapy in locally advanced breast cancer refractory to neoadjuvant chemotherapy? Breast 31:192–196
Rutqvist LE, Pettersson D, Johansson H (1993) Adjuvant radiation therapy versus surgery alone in operable breast cancer: long-term follow-up of a randomized clinical trial. Radiother Oncol 26:104–110
Wallgren A, Arner O, Bergström J et al (1986) Radiation therapy in operable breast cancer: results from the Stockholm trial on adjuvant radiotherapy. Int J Radiat Oncol Biol Phys 12:533–537
Masuda N, Lee SJ, Ohtani S et al (2017) Adjuvant capecitabine for breast cancer after preoperative chemotherapy. N Engl J Med 376:2147–2159
von Minckwitz G, Huang CS, Mano MS et al (2019) Trastuzumab emtansine for residual invasive HER2-positive breast cancer. N Engl J Med 380:617–628
Krug D, Baumann R, Budach W et al (2018) Individualization of post-mastectomy radiotherapy and regional nodal irradiation based on treatment response after neoadjuvant chemotherapy for breast cancer. Strahlenther Onkol 194:607–618
Krug D, Baumann R, Budach W et al (2018) Neoadjuvant chemotherapy for breast cancer—background for the indication of locoregional treatment. Strahlenther Onkol 194:797–805
Corradini S, Krug D, Meattini I et al (2019) Preoperative radiotherapy: a paradigm shift in the treatment of breast cancer? A review of literature. Crit Rev Oncol Hematol 141:102–111
Pazos M, Corradini S, Dian D et al (2017) Neoadjuvant radiotherapy followed by mastectomy and immediate breast reconstruction: an alternative treatment option for locally advanced breast cancer. Strahlenther Onkol 193:324–331
Jagsi R, Li Y, Morrow M et al (2015) Patient-reported quality of life and satisfaction with cosmetic outcomes after breast conservation and mastectomy with and without reconstruction: results of a survey of breast cancer survivors. Ann Surg 261:1198–1206
Ruvalcaba-Limon E, Robles-Vidal C, Poitevin-Chacon A, Chavez-Macgregor M, Gamboa-Vignolle C, Vilar-Compte D (2006) Complications after breast cancer surgery in patients treated with concomitant preoperative chemoradiation: a case-control analysis. Breast Cancer Res Treat 95:147–152
Matuschek C, Nestle-Kraemling C, Haussmann J, Bölke E, Wollandt S, Speer V, Djiepmo Njanang FJ, Tamaskovics B, Gerber PA, Orth K, Ruckhaeberle E, Fehm T, Corradini S, Lammering G, Mohrmann S, Audretsch W, Roth S, Kammers K, Budach W (2019) Long-term cosmetic outcome after preoperative radio-/chemotherapy in locally advanced breast cancer patients. Strahlenther Onkol 195(7):615–628. https://doi.org/10.1007/s00066-019-01473-2
Musoro ZJ, Hamel JF, Ediebah DE et al (2018) Establishing anchor-based minimally important differences (MID) with the EORTC quality-of-life measures: a meta-analysis protocol. Bmj Open 8:e19117
Roth SL, Audretsch W, Bojar H, Lang I, Willers R, Budach W (2010) Retrospective study of neoadjuvant versus adjuvant radiochemotherapy in locally advanced noninflammatory breast cancer: survival advantage in cT2 category by neoadjuvant radiochemotherapy. Strahlenther Onkol 186:299–306
Matuschek C, Bolke E, Roth SL et al (2012) Long-term outcome after neoadjuvant radiochemotherapy in locally advanced noninflammatory breast cancer and predictive factors for a pathologic complete remission: results of a multivariate analysis. Strahlenther Onkol 188:777–781
Schwarz R, Hinz A (2001) Reference data for the quality of life questionnaire EORTC QLQ-C30 in the general German population. Eur J Cancer 37:1345–1351
Early Breast Cancer Trialists’ Collaborative G. (2015) Aromatase inhibitors versus tamoxifen in early breast cancer: patient-level meta-analysis of the randomised trials. Lancet 386:1341–1352
Poortmans PM, Struikmans H, Bartelink H (2015) Regional Nodal Irradiation in Early-Stage Breast Cancer. N Engl J Med 373:1879–1880
Whelan TJ, Olivotto IA, Levine MN (2015) Regional Nodal Irradiation in Early-Stage Breast Cancer. N Engl J Med 373:1878–1879
Engel J, Kerr J, Schlesinger-Raab A, Sauer H, Holzel D (2004) Quality of life following breast-conserving therapy or mastectomy: results of a 5-year prospective study. Breast J 10:223–231
Munshi A, Dutta D, Budrukkar A et al (2012) Impact of adjuvant radiation therapy photon energy on quality of life after breast conservation therapy: linear accelerator versus the cobalt machine. J Cancer Res Ther 8:361–366
Dujmovic A, Marcinko D, Bulic K, Kisic H, Dudukovic M, Mijatovic D (2017) Quality of life and depression among female patients undergoing surgical treatment for breast cancer: A prospective study. Psychiatr Danub 29:345–350
Vartanian JG, Kowalski LP (2009) Acceptance of major surgical procedures and quality of life among long-term survivors of advanced head and neck cancer. Arch Otolaryngol Head Neck Surg 135:376–379
Balk EM, Gazula A, Markozannes G et al (2019) Psychometric Properties of Functional, Ambulatory, and Quality of Life Instruments in Lower Limb Amputees: a Systematic Review. Arch Phys Med Rehabil 100(12):2354–2370
Bantema-Joppe EJ, de Bock GH, Woltman-van Iersel M et al (2015) The impact of age on changes in quality of life among breast cancer survivors treated with breast-conserving surgery and radiotherapy. Br J Cancer 112:636–643
Hsu T, Ennis M, Hood N, Graham M, Goodwin PJ (2013) Quality of life in long-term breast cancer survivors. J Clin Oncol 31:3540–3548
Author information
Authors and Affiliations
Contributions
CM, CNK, EB, and WB had the idea, coordinated the work, and wrote parts of the manuscript. JH did the literature research, prepared the data for analysis, and wrote parts of the manuscript. KK and JH did the statistical analysis. SW, VS, FJN, BZ, PAG, KO, ER, TF, SC, GL, SM, and SR wrote parts of the manuscript. WA operated all patients. JH contributed significantly to the discussion on the interpretation of the results. CM, JH, and PAG prepared the figures and tables. All authors read and approved the final manuscript. All authors gave consent for the publication.
Corresponding author
Ethics declarations
Conflict of interest
J. Haussmann, C. Nestle-Kraemling, E. Bölke, S. Wollandt, V. Speer, F.‑J. Djiepmo Njanang, B. Tamaskovics, P.A. Gerber, K. Orth, E. Ruckhaeberle, T. Fehm, S. Corradini, G. Lammering, S. Mohrmann, W. Audretsch, K. Maas, S. Roth, K. Kammers, W. Budach, and C. Matuschek declare that they have no competing interests.
Ethical standards
There was no ethics approval necessary, because in this meta-analysis, we were pulling numbers from the published manuscripts and pooling results. The study was approved by the local ethics committee (no.: 4049 and 4070).
Additional information
The authors Jan Haussmann and Carolin Nestle-Kraemling contributed equally to the manuscript.
The work is a part of the thesis of SW and VS.
Caption Electronic Supplementary Material
66_2019_1557_MOESM1_ESM.doc
Table 5a: Patient characteristics of breast-conserving surgery. Median follow-up is 16.6 years. The comparison between the original neoadjuvant group and the current cohort is presented.
66_2019_1557_MOESM2_ESM.doc
Table 5b: Patient characteristics of mastectomy. Median follow-up is 17.2 years. The comparison between the original neoadjuvant group and the current cohort is shown.
66_2019_1557_MOESM3_ESM.pptx
Table 6: EORTC QLQ-C30 functional scales between different subgroups. Presented are the mean values of each scale and their corresponding reference group value with confidence intervals. P-values are calculated from two-sided paired t‑tests. Additionally, the rate of excellent and good quality of life summary scales is given for all patients, breast-conserved women, and mastectomy patients.
66_2019_1557_MOESM4_ESM.pptx
Table 7: EORTC QLQ-C30 symptom scales between different subgroups. Presented are the mean values of each scale and their corresponding reference group value with confidence intervals. P-values are calculated from two-sided paired t‑tests. Additionally, the rate of excellent and good quality of life summary scales is given for all patients, breast-conserved women, and mastectomy patients.
Rights and permissions
About this article
Cite this article
Haussmann, J., Nestle-Kraemling, C., Bölke, E. et al. Long-term quality of life after preoperative radiochemotherapy in patients with localized and locally advanced breast cancer. Strahlenther Onkol 196, 386–397 (2020). https://doi.org/10.1007/s00066-019-01557-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-019-01557-z