Abstract
Purpose
To investigate the impact of primary gross tumor volume (pGTV) and nodal gross tumor volume (nGTV) in oropharyngeal squamous cell carcinoma (OPSCC) and the difference in their role between human papillomavirus (HPV)-positive and HPV-negative patients.
Methods
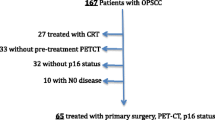
The patient cohort consists of 91 OPSCC patients treated with definitive radiochemotherapy or radiotherapy using intensity-modulated radiotherapy (IMRT). All patients had a minimum follow-up of 31 months. Volume measurements were made from computer tomography (CT) scans and HPV status was assessed by p16 immunohistochemistry. The end points were as follows: overall survival (OS), disease-free survival (DFS) and locoregional control (LRC).
Results
pGTV was a significant independent prognostic factor for overall survival (OS; p = 0.020) in p16-negative patients. nGTV of p16-negative tumors had significant prognostic value in all end points in multivariate analyses. High-stage (III–IVc) p16-negative tumors were only associated with significantly poorer OS (p = 0.046) but not with poorer LRC or DFS when compared with the low-stage (I–II) tumors. nGTV of p16-positive tumors was an independent prognostic factor for DFS (p = 0.005) and LRC (p = 0.007) in multivariate analyses.
Conclusion
pGTV may serve as an independent prognostic factor in p16-negative patients and nGTV may serve as an independent prognostic factor both in p16-positive and p16-negative patients treated with radiochemotherapy or radiotherapy using IMRT. Tumor volume may have an impact on selecting patients for de-escalation protocols in the future, both in p16-positive and p16-negative patients.
Zusammenfassung
Zielstellung
Ziel war es, die Bedeutung des makroskopischen Tumorvolumens des Primärtumors („primary gross tumor volume“, pGTV) und des makroskopischen Tumorvolumens der Lymphknoten („nodal gross tumor volume“, nGTV) bei Plattenepithelkarzinomen des Oropharynx („oropharyngeal squamous cell carcinoma“, OPSCC) und deren Unterschiede zwischen für humanes Papillomavirus (HPV) positiven und HPV-negativen Patienten zu untersuchten.
Methoden
In die Studie eingeschlossen wurden 91 OPSCC-Patienten, die mit definitiver intensitätsmodulierter Strahlentherapie („intensity-modulated radiotherapy“, IMRT) mit oder ohne gleichzeitige Chemotherapie behandelt wurden. Bei allen Patienten betrug der Nachbeobachtungszeitraum mindestens 31 Monate. Die Volumenmessungen erfolgten anhand von Computertomographie(CT)-Aufnahmen. Der HPV-Status wurde durch die p16-Immunhistochemie ermittelt. Die Endpunkte waren Gesamtüberleben („overall survival“, OS), krankheitsfreies Überleben („disease-free survival“, DFS) und lokoregionale Kontrolle („locoregional control“, LRC).
Ergebnisse
Das pGTV war ein signifikanter unabhängiger prognostischer Faktor für das OS (p = 0,20) bei p16-negativen Patienten. Das nGTV von p16-negativen Tumoren wies einen signifikanten prognostischen Wert für alle Endpunkte in multivariaten Analysen auf. p16-negative Tumoren in hohen Stadien (III-IVc) waren im Vergleich zu den Tumoren in niedrigeren Stadien (I‑II) nur mit einem signifikant schlechteren OS (p = 0,046), nicht aber mit schlechteren Werten für LRC oder DFS assoziiert. Das nGTV von p16-negativen Tumoren erwies sich in multivariaten Analysen als unabhängiger prognostischer Faktor für DFS (p = 0,005) und LRC (p = 0,007).
Schlussfolgerung
Das pGTV kann als unabhängiger prognostischer Faktor bei p16-negativen Patienten und das nGTV als unabhängiger prognostischer Faktor sowohl bei p16-positiven als auch bei p16-negativen Patienten dienen, bei denen eine Radiochemotherapie oder Strahlentherapie unter Verwendung der IMRT erfolgt. Das Tumorvolumen kann bei der Auswahl der Patienten für ein Deeskalationsprotokoll sowohl bei p16-positiven als auch bei p16-negativen Patienten eine Rolle spielen.





Similar content being viewed by others
References
Chao KS, Ozyigit G, Blanco AI, Thorstad WL, Deasy JO, Haughey BH, Spector GJ, Sessions DG (2004) Intensity-modulated radiation therapy for oropharyngeal carcinoma: impact of tumor volume. Int J Radiat Oncol Biol Phys 59(1):43–50. https://doi.org/10.1016/j.ijrobp.2003.08.004
Chen SW, Yang SN, Liang JA, Lin FJ, Tsai MH (2009) Prognostic impact of tumor volume in patients with stage III–IVA hypopharyngeal cancer without bulky lymph nodes treated with definitive concurrent chemoradiotherapy. Head Neck 31(6):709–716. https://doi.org/10.1002/hed.21011
Lok BH, Setton J, Caria N, Romanyshyn J, Wolden SL, Zelefsky MJ, Park J, Rowan N, Sherman EJ, Fury MG, Ho A, Pfister DG, Wong RJ, Shah JP, Kraus DH, Zhang Z, Schupak KD, Gelblum DY, Rao SD, Lee NY (2012) Intensity-modulated radiation therapy in oropharyngeal carcinoma: effect of tumor volume on clinical outcomes. Int J Radiat Oncol Biol Phys 82(5):1851–1857. https://doi.org/10.1016/j.ijrobp.2011.03.029
He YX, Wang Y, Cao PF, Shen L, Zhao YJ, Zhang ZJ, Chen DM, Yang TB, Huang XQ, Qin Z, Dai YY, Shen LF (2016) Prognostic value and predictive threshold of tumor volume for patients with locally advanced nasopharyngeal carcinoma receiving intensity-modulated radiotherapy. Chin J Cancer 35(1):96. https://doi.org/10.1186/s40880-016-0159-2
Linge A, Lohaus F, Lock S, Nowak A, Gudziol V, Valentini C, von Neubeck C, Jutz M, Tinhofer I, Budach V, Sak A, Stuschke M, Balermpas P, Rodel C, Grosu AL, Abdollahi A, Debus J, Ganswindt U, Belka C, Pigorsch S, Combs SE, Monnich D, Zips D, Buchholz F, Aust DE, Baretton GB, Thames HD, Dubrovska A, Alsner J, Overgaard J, Krause M, Baumann M, DKTK-ROG (2016) HPV status, cancer stem cell marker expression, hypoxia gene signatures and tumour volume identify good prognosis subgroups in patients with HNSCC after primary radiochemotherapy: A multicentre retrospective study of the German Cancer Consortium Radiation Oncology Group (DKTK-ROG). Radiother Oncol 121(3):364–373. https://doi.org/10.1016/j.radonc.2016.11.008
Studer G, Lutolf UM, El-Bassiouni M, Rousson V, Glanzmann C (2007) Volumetric staging (VS) is superior to TNM and AJCC staging in predicting outcome of head and neck cancer treated with IMRT. Acta Oncol 46(3):386–394. https://doi.org/10.1080/02841860600815407
Doweck I, Denys D, Robbins KT (2002) Tumor volume predicts outcome for advanced head and neck cancer treated with targeted chemoradiotherapy. Laryngoscope 112(10):1742–1749. https://doi.org/10.1097/00005537-200210000-00006
Rutkowski T (2014) The role of tumor volume in radiotherapy of patients with head and neck cancer. Radiat Oncol 9:23. https://doi.org/10.1186/1748-717x-9-23
Strongin A, Yovino S, Taylor R, Wolf J, Cullen K, Zimrin A, Strome S, Regine W, Suntharalingam M (2012) Primary tumor volume is an important predictor of clinical outcomes among patients with locally advanced squamous cell cancer of the head and neck treated with definitive chemoradiotherapy. Int J Radiat Oncol Biol Phys 82(5):1823–1830. https://doi.org/10.1016/j.ijrobp.2010.10.053
Knegjens JL, Hauptmann M, Pameijer FA, Balm AJ, Hoebers FJ, de Bois JA, Kaanders JH, van Herpen CM, Verhoef CG, Wijers OB, Wiggenraad RG, Buter J, Rasch CR (2011) Tumor volume as prognostic factor in chemoradiation for advanced head and neck cancer. Head Neck 33(3):375–382. https://doi.org/10.1002/hed.21459
Brenner DJ (1993) Dose, volume, and tumor-control predictions in radiotherapy. Int J Radiat Oncol Biol Phys 26(1):171–179
Dubben HH, Thames HD, Beck-Bornholdt HP (1998) Tumor volume: a basic and specific response predictor in radiotherapy. Radiother Oncol 47(2):167–174
Johnson CR, Thames HD, Huang DT, Schmidt-Ullrich RK (1995) The tumor volume and clonogen number relationship: tumor control predictions based upon tumor volume estimates derived from computed tomography. Int J Radiat Oncol Biol Phys 33(2):281–287
Yang SN, Chiou YR, Zhang GG, Chou KT, Huang TC (2017) The clinical outcome correlations between radiation dose and pretreatment metabolic tumor volume for radiotherapy in head and neck cancer: a retrospective analysis. Medicine (Baltimore) 96(26):e7186. https://doi.org/10.1097/MD.0000000000007186
Edge SBBD, Compton CC, Fritz AG, Greene FL, Trotti A (2010) AJCC cancer staging manual, 7th edn. Springer, New York
Edge SB, Compton CC (2010) The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol 17(6):1471–1474. https://doi.org/10.1245/s10434-010-0985-4
Davis KS, Lim CM, Clump DA, Heron DE, Ohr JP, Kim S, Duvvuri U, Johnson JT, Ferris RL (2016) Tumor volume as a predictor of survival in human papillomavirus-positive oropharyngeal cancer. Head Neck 38(Suppl 1):E1613–1617. https://doi.org/10.1002/hed.24287
Hermans R, Op de beeck K, Van den Bogaert W, Rijnders A, Staelens L, Feron M, Bellon E (2001) The relation of CT-determined tumor parameters and local and regional outcome of tonsillar cancer after definitive radiation treatment. Int J Radiat Oncol Biol Phys 50(1):37–45
Lydiatt WM, Patel SG, O’Sullivan B, Brandwein MS, Ridge JA, Migliacci JC, Loomis AM, Shah JP (2017) Head and Neck cancers-major changes in the American Joint Committee on cancer eighth edition cancer staging manual. CA Cancer J Clin 67(2):122–137. https://doi.org/10.3322/caac.21389
Amin MB, Edge S, Greene F, Byrd DR, Brookland RK, Washington MK, Gershenwald JE, Compton CC, Hess KR, Sullivan DC, Jessup JM, Brierley JD, Gaspar LE, Schilsky RL, Balch CM, Winchester DP, Asare EA, Madera M, Gress DM, Meyer LR (2017) AJCC cancer staging manual, 8th edn. Springer, New York
Ang KK, Harris J, Wheeler R, Weber R, Rosenthal DI, Nguyen-Tan PF, Westra WH, Chung CH, Jordan RC, Lu C, Kim H, Axelrod R, Silverman CC, Redmond KP, Gillison ML (2010) Human papillomavirus and survival of patients with oropharyngeal cancer. N Engl J Med 363(1):24–35. https://doi.org/10.1056/NEJMoa0912217
Gillison ML, D’Souza G, Westra W, Sugar E, Xiao W, Begum S, Viscidi R (2008) Distinct risk factor profiles for human papillomavirus type 16-positive and human papillomavirus type 16-negative head and neck cancers. J Natl Cancer Inst 100(6):407–420. https://doi.org/10.1093/jnci/djn025
Mehanna H, Beech T, Nicholson T, El-Hariry I, McConkey C, Paleri V, Roberts S (2013) Prevalence of human papillomavirus in oropharyngeal and nonoropharyngeal head and neck cancer—systematic review and meta-analysis of trends by time and region. Head Neck 35(5):747–755. https://doi.org/10.1002/hed.22015
D’Souza G, Kreimer AR, Viscidi R, Pawlita M, Fakhry C, Koch WM, Westra WH, Gillison ML (2007) Case-control study of human papillomavirus and oropharyngeal cancer. N Engl J Med 356(19):1944–1956. https://doi.org/10.1056/NEJMoa065497
Fakhry C, Westra WH, Li S, Cmelak A, Ridge JA, Pinto H, Forastiere A, Gillison ML (2008) Improved survival of patients with human papillomavirus-positive head and neck squamous cell carcinoma in a prospective clinical trial. J Natl Cancer Inst 100(4):261–269. https://doi.org/10.1093/jnci/djn011
Bird D, Scarsbrook AF, Sykes J, Ramasamy S, Subesinghe M, Carey B, Wilson DJ, Roberts N, McDermott G, Karakaya E, Bayman E, Sen M, Speight R, Prestwich RJ (2015) Multimodality imaging with CT, MR and FDG-PET for radiotherapy target volume delineation in oropharyngeal squamous cell carcinoma. BMC Cancer 15:844. https://doi.org/10.1186/s12885-015-1867-8
Emami B, Sethi A, Petruzzelli GJ (2003) Influence of MRI on target volume delineation and IMRT planning in nasopharyngeal carcinoma. Int J Radiat Oncol Biol Phys 57(2):481–488. https://doi.org/10.1016/s0360-3016(03)00570-4
van den Brekel MW, Stel HV, Castelijns JA, Nauta JJ, van der Waal I, Valk J, Meyer CJ, Snow GB (1990) Cervical lymph node metastasis: assessment of radiologic criteria. Radiology 177(2):379–384. https://doi.org/10.1148/radiology.177.2.2217772
Cantrell SC, Peck BW, Li G, Wei Q, Sturgis EM, Ginsberg LE (2013) Differences in imaging characteristics of HPV-positive and HPV-Negative oropharyngeal cancers: a blinded matched-pair analysis. AJNR Am J Neuroradiol 34(10):2005–2009. https://doi.org/10.3174/ajnr.A3524
McIlwain WR, Sood AJ, Nguyen SA, Day TA (2014) Initial symptoms in patients with HPV-positive and HPV-negative oropharyngeal cancer. JAMA Otolaryngol Head Neck Surg 140(5):441–447. https://doi.org/10.1001/jamaoto.2014.141
Martinez I, Wang J, Hobson KF, Ferris RL, Khan SA (2007) Identification of differentially expressed genes in HPV-positive and HPV-negative oropharyngeal squamous cell carcinomas. Eur J Cancer 43(2):415–432. https://doi.org/10.1016/j.ejca .2006.09.001
Huang SH, O’Sullivan B (2017) Overview of the 8th edition TNM classification for head and neck cancer. Curr Treat Options Oncol 18(7):40. https://doi.org/10.1007/s11864-017-0484-y
van den Broek GB, Rasch CR, Pameijer FA, Peter E, van den Brekel MW, Tan IB, Schornagel JH, de Bois JA, Zijp LJ, Balm AJ (2004) Pretreatment probability model for predicting outcome after intraarterial chemoradiation for advanced head and neck carcinoma. Cancer 101(8):1809–1817. https://doi.org/10.1002/cncr.20556
Acknowledgements
This study was supported by grants from the Helsinki University Hospital Research Fund and the Finska Läkaresällskapet.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
T. Carpén, K. Saarilahti, C. Haglund, A. Markkola, J. Tarkkanen, J. Hagström, P. Mattila, and A. Mäkitie declare that they have no competing interests.
Ethical standards
This article does not contain any studies with human participants or animals performed by any of the authors. Consent was obtained from all patients identifiable from images or other information within the manuscript. In the case of underage patients, consent was obtained from a parent or legal guardian.
Rights and permissions
About this article
Cite this article
Carpén, T., Saarilahti, K., Haglund, C. et al. Tumor volume as a prognostic marker in p16-positive and p16-negative oropharyngeal cancer patients treated with definitive intensity-modulated radiotherapy. Strahlenther Onkol 194, 759–770 (2018). https://doi.org/10.1007/s00066-018-1309-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00066-018-1309-z