Abstract
Purpose
Excessive daytime sleepiness (EDS) is a common non-motor symptom in Parkinson’s disease (PD), but its neuropathology remains elusive. Our goal is to explore the potential neural substrates of EDS in a large sample of individuals with PD.
Methods
We recruited 48 PD patients with and 87 PD patients without EDS. We used resting-state functional magnetic resonance imaging to compare amplitudes of low-frequency fluctuations (ALFF) between the two groups. We also explored functional connectivity (FC) between the entire brain and regions where ALFF differed between the two groups as well as FC between selected regions of interest. Age, Part III of the Movement Disorder Society Unified Parkinson’s Disease Rating Scale (MDS-UPDRS-III) score and use of dopamine receptor agonists were treated as covariates in the comparisons.
Results
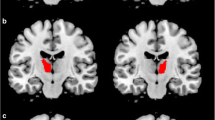
EDS was associated with significantly lower ALFF in the left angular gyrus, and ALFF in this region correlated negatively with score on the Epworth Sleepiness Scale in patients with PD. EDS was also associated with significantly lower FC between the left angular gyrus and right cerebellum, based on seed-to-voxel and inter-ROI analyses.
Conclusion
Our results suggest that EDS in PD patients is associated with reduced spontaneous neural activity in the left angular gyrus and with reduced FC between the left angular gyrus and cerebellum. These findings may help understand and treat EDS in PD.


Similar content being viewed by others
References
Knie B, Mitra MT, Logishetty K, Chaudhuri KR. Excessive daytime sleepiness in patients with Parkinson’s disease. CNS Drugs. 2011;25:203–12.
Frucht S, Rogers JD, Greene PE, Gordon MF, Fahn S. Falling asleep at the wheel: motor vehicle mishaps in persons taking pramipexole and ropinirole. Neurology. 1999;52:1908–10.
Yoo SW, Kim JS, Oh YS, Ryu DW, Lee KS. Excessive daytime sleepiness and its impact on quality of life in de novo Parkinson’s disease. Neurol Sci. 2019;40:1151–6.
Chahine LM, Amara AW, Videnovic A. A systematic review of the literature on disorders of sleep and wakefulness in Parkinson’s disease from 2005 to 2015. Sleep Med Rev. 2017;35:33–50.
Adler CH, Thorpy MJ. Sleep issues in Parkinson’s disease. Neurology. 2005;64:S12–20.
Happe S, Baier PC, Helmschmied K, Meller J, Tatsch K, Paulus W. Association of daytime sleepiness with nigrostriatal dopaminergic degeneration in early Parkinson’s disease. J Neurol. 2007;254:1037–43.
Pagano G, Molloy S, Bain PG, Rabiner EA, Chaudhuri KR, Brooks DJ, Pavese N. Sleep problems and hypothalamic dopamine D3 receptor availability in Parkinson disease. Neurology. 2016;87:2451–6.
Yoo SW, Oh YS, Ryu DW, Lee KS, Lyoo CH, Kim JS. Low thalamic monoamine transporter availability is related to excessive daytime sleepiness in early Parkinson’s disease. Neurol Sci. 2020;41:1081–7.
Wen MC, Ng SY, Heng HS, Chao YX, Chan LL, Tan EK, Tan LC. Neural substrates of excessive daytime sleepiness in early drug naive Parkinson’s disease: a resting state functional MRI study. Parkinsonism Relat Disord. 2016;24:63–8.
Wang X, Wang M, Yuan Y, Li J, Shen Y, Zhang K. Altered amplitude of low-frequency fluctuations and functional connectivity in excessive daytime sleepiness in Parkinson disease. Front Neurosci. 2020;14:29.
Zuo XN, Di Martino A, Kelly C, Shehzad ZE, Gee DG, Klein DF, Castellanos FX, Biswal BB, Milham MP. The oscillating brain: complex and reliable. Neuroimage. 2010;49:1432–45.
Gorges M, Müller HP, Lulé D, Ludolph AC, Pinkhardt EH, Kassubek J. Functional connectivity within the default mode network is associated with saccadic accuracy in Parkinson’s disease: a resting-state FMRI and videooculographic study. Brain Connect. 2013;3:265-72.
Postuma RB, Berg D, Stern M, Poewe W, Olanow CW, Oertel W, Obeso J, Marek K, Litvan I, Lang AE, Halliday G, Goetz CG, Gasser T, Dubois B, Chan P, Bloem BR, Adler CH, Deuschl G. MDS clinical diagnostic criteria for Parkinson’s disease. Mov Disord. 2015;30:1591–601.
Crum RM, Anthony JC, Bassett SS, Folstein MF. Population-based norms for the Mini-Mental State Examination by age and educational level. JAMA. 1993;269:2386–91.
Johns MW. A new method for measuring daytime sleepiness: the Epworth sleepiness scale. Sleep. 1991;14:540–5.
Goetz CG, Tilley BC, Shaftman SR, Stebbins GT, Fahn S, Martinez-Martin P, Poewe W, Sampaio C, Stern MB, Dodel R, Dubois B, Holloway R, Jankovic J, Kulisevsky J, Lang AE, Lees A, Leurgans S, LeWitt PA, Nyenhuis D, Olanow CW, Rascol O, Schrag A, Teresi JA, van Hilten JJ, LaPelle N; Movement Disorder Society UPDRS Revision Task Force. Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): scale presentation and clinimetric testing results. Mov Disord. 2008;23:2129–70.
Jankovic J, McDermott M, Carter J, Gauthier S, Goetz C, Golbe L, Huber S, Koller W, Olanow C, Shoulson I, et al. Variable expression of Parkinson’s disease: a base-line analysis of the DATATOP cohort. The Parkinson Study Group. Neurology. 1990;40:1529–34.
Tomlinson CL, Stowe R, Patel S, Rick C, Gray R, Clarke CE. Systematic review of levodopa dose equivalency reporting in Parkinson’s disease. Mov Disord. 2010;25:2649–53.
Whitfield-Gabrieli S, Nieto-Castanon A. Conn: a functional connectivity toolbox for correlated and anticorrelated brain networks. Brain Connect. 2012;2:125–41.
Yan CG, Wang XD, Zuo XN, Zang YF. DPABI: Data Processing & Analysis for (Resting-State) Brain Imaging. Neuroinformatics. 2016;14:339–51.
Raichle ME, MacLeod AM, Snyder AZ, Powers WJ, Gusnard DA, Shulman GL. A default mode of brain function. Proc Natl Acad Sci USA. 2001;98:676–82.
Qin P, Wu X, Huang Z, Duncan NW, Tang W, Wolff A, Hu J, Gao L, Jin Y, Wu X, Zhang J, Lu L, Wu C, Qu X, Mao Y, Weng X, Zhang J, Northoff G. How are different neural networks related to consciousness? Ann Neurol. 2015;78:594–605.
Seghier ML. The angular gyrus: multiple functions and multiple subdivisions. Neuroscientist. 2013;19:43–61.
Binder JR, Desai RH. The neurobiology of semantic memory. Trends Cogn Sci. 2011;15:527–36.
Bonner MF, Peelle JE, Cook PA, Grossman M. Heteromodal conceptual processing in the angular gyrus. Neuroimage. 2013;71:175–86.
Guo Z, Jiang Z, Jiang B, McClure MA, Mu Q. High-Frequency Repetitive Transcranial Magnetic Stimulation Could Improve Impaired Working Memory Induced by Sleep Deprivation. Neural Plast. 2019;2019:7030286.
Ji T, Li X, Chen J, Ren X, Mei L, Qiu Y, Zhang J, Wang S, Xu Z, Li H, Zheng L, Peng Y, Liu Y, Ni X, Tai J, Liu J. Brain function in children with obstructive sleep apnea: a resting-state fMRI study. Sleep. 2021;44:zsab047.
Peng DC, Dai XJ, Gong HH, Li HJ, Nie X, Zhang W. Altered intrinsic regional brain activity in male patients with severe obstructive sleep apnea: a resting-state functional magnetic resonance imaging study. Neuropsychiatr Dis Treat. 2014;10:1819–26.
Song X, Roy B, Kang DW, Aysola RS, Macey PM, Woo MA, Yan-Go FL, Harper RM, Kumar R. Altered resting-state hippocampal and caudate functional networks in patients with obstructive sleep apnea. Brain Behav. 2018;8:e00994.
van Geest Q, Westerik B, van der Werf YD, Geurts JJ, Hulst HE. The role of sleep on cognition and functional connectivity in patients with multiple sclerosis. J Neurol. 2017;264:72–80.
Legostaeva L, Poydasheva A, Iazeva E, Sinitsyn D, Sergeev D, Bakulin I, Lagoda D, Kremneva E, Morozova S, Ryabinkina Y, Suponeva N, Piradov M. Stimulation of the Angular Gyrus Improves the Level of Consciousness. Brain Sci. 2019;9:103.
Huang YS, Hsiao IT, Liu FY, Hwang FM, Lin KL, Huang WC, Guilleminault C. Neurocognition, sleep, and PET findings in type 2 vs type 1 narcolepsy. Neurology. 2018;90:e1478–87.
Huang YS, Liu FY, Lin CY, Hsiao IT, Guilleminault C. Brain imaging and cognition in young narcoleptic patients. Sleep Med. 2016;24:137–44.
DelRosso LM, Hoque R. The cerebellum and sleep. Neurol Clin. 2014;32:893–900.
Pedroso JL, Braga-Neto P, Escorcio-Bezerra ML, Abrahão A, de Albuquerque MV, Filho FM, de Souza PV, de Rezende Pinto WB, Borges FR Jr, Saraiva-Pereira ML, Jardim LB, Barsottini OG. Non-motor and Extracerebellar Features in Spinocerebellar Ataxia Type 2. Cerebellum. 2017;16:34–9.
Dang D, Cunnington D. Excessive daytime somnolence in spinocerebellar ataxia type 1. J Neurol Sci. 2010;290:146–7.
Yuan X, Ou R, Hou Y, Chen X, Cao B, Hu X, Shang H. Extra-Cerebellar Signs and Non-motor Features in Chinese Patients With Spinocerebellar Ataxia Type 3. Front Neurol. 2019;10:110.
Joo EY, Noh HJ, Kim JS, Koo DL, Kim D, Hwang KJ, Kim JY, Kim ST, Kim MR, Hong SB. Brain Gray Matter Deficits in Patients with Chronic Primary Insomnia. Sleep. 2013;36:999–1007
Desseilles M, Dang-Vu T, Schabus M, Sterpenich V, Maquet P, Schwartz S. Neuroimaging insights into the pathophysiology of sleep disorders. Sleep. 2008;31:777–94.
Zhang LB, Zhang J, Sun MJ, Chen H, Yan J, Luo FL, Yao ZX, Wu YM, Hu B. Neuronal Activity in the Cerebellum During the Sleep-Wakefulness Transition in Mice. Neurosci Bull. 2020;36:919–31.
Canto CB, Onuki Y, Bruinsma B, van der Werf YD, De Zeeuw CI. The Sleeping Cerebellum. Trends Neurosci. 2017;40:309–23.
Zhang Y, Yang Y, Yang Y, Li J, Xin W, Huang Y, Shao Y, Zhang X. Alterations in Cerebellar Functional Connectivity Are Correlated With Decreased Psychomotor Vigilance Following Total Sleep Deprivation. Front Neurosci. 2019;13:134.
Yang L, Lei Y, Wang L, Chen P, Cheng S, Chen S, Sun J, Li Y, Wang Y, Hu W, Yang Z. Abnormal functional connectivity density in sleep-deprived subjects. Brain Imaging Behav. 2018;12:1650–7.
Desjardins MÈ, Baril AA, Soucy JP, Dang-Vu TT, Desautels A, Petit D, Montplaisir J, Zadra A. Altered brain perfusion patterns in wakefulness and slow-wave sleep in sleepwalkers. Sleep. 2018;41:zsy039.
Lim JS, Shin SA, Lee JY, Nam H, Lee JY, Kim YK. Neural substrates of rapid eye movement sleep behavior disorder in Parkinson’s disease. Parkinsonism Relat Disord. 2016;23:31–6.
Ayalon L, Ancoli-Israel S, Drummond SP. Altered brain activation during response inhibition in obstructive sleep apnea. J Sleep Res. 2009;18:204–8.
Zhou F, Huang S, Zhuang Y, Gao L, Gong H. Frequency-dependent changes in local intrinsic oscillations in chronic primary insomnia: A study of the amplitude of low-frequency fluctuations in the resting state. Neuroimage Clin. 2016;15:458–65.
Han XH, Li XM, Tang WJ, Yu H, Wu P, Ge JJ, Wang J, Zuo CT, Shi KY. Assessing gray matter volume in patients with idiopathic rapid eye movement sleep behavior disorder. Neural Regen Res. 2019;14:868–75.
Tholfsen LK, Larsen JP, Schulz J, Tysnes OB, Gjerstad MD. Development of excessive daytime sleepiness in early Parkinson disease. Neurology. 2015;85:162–8.
Johns MW. Sensitivity and specificity of the multiple sleep latency test (MSLT), the maintenance of wakefulness test and the epworth sleepiness scale: failure of the MSLT as a gold standard. J Sleep Res. 2000;9:5–11.
Tandberg E, Larsen JP, Karlsen K. A community-based study of sleep disorders in patients with Parkinson’s disease. Mov Disord. 1998;13:895–9.
Videnovic A, Golombek D. Circadian and sleep disorders in Parkinson’s disease. Exp Neurol. 2013;243:45–56.
Acknowledgements
We would like to thank all the patients in this study. This study was supported by the Henan Province Science and Technology Development Plan (192102310085) and the Henan Province Medical Science and Technology Research Program (201701018).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
J.H. Zheng, J.J. Ma, W.H. Sun, Z.D. Wang, Q.Q. Chang, L.R. Dong, X.X. Shi and M.J. Li declare that they have no competing interests.
Ethical standards
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1975 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study.
Additional information
Data availability
The data that support the findings of the present study are available from the corresponding authors upon reasonable request.
Rights and permissions
About this article
Cite this article
Zheng, J.H., Ma, J.J., Sun, W.H. et al. Excessive Daytime Sleepiness in Parkinson’s Disease is Related to Functional Abnormalities in the Left Angular Gyrus. Clin Neuroradiol 33, 121–127 (2023). https://doi.org/10.1007/s00062-022-01190-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00062-022-01190-x