Abstract
Background
This study evaluated whether subclinical myocardial dysfunction occurs in first-degree relatives of patients with idiopathic dilated cardiomyopathy (IDCM), using strain echocardiographic imaging, before apparent left ventricular (LV) failure is observed.
Patients and methods
The study comprised 77 subjects aged 16–63 years who had first-degree relatives with a previous or new diagnosis of IDCM. LV myocardial deformation parameters of the first-degree relatives with normal LVEF (≥55%) values, as assessed using 2D echocardiography, were evaluated. The findings of the first-degree relatives were compared with an age- and sex-matched control group (n = 86).
Results
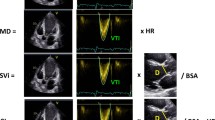
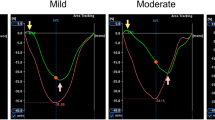
No difference in terms of age, gender, and body surface area was detected between first-degree relatives and controls. First-degree relatives of IDCM patients had significantly lower LVEF (62.04 ± 5.8% vs. 65.65 ± 6.3%, p < 0.001) and FS values (39.4 ± 6.6 vs. 41.45 ± 5.5, p = 0.03) compared with the controls. Assessment of LV deformation parameters revealed that LV global longitudinal strain (−17.34 ± 2.19% vs. −19.21 ± 2.16%, p < 0.001) and strain rate (0.94 ± 0.14 s−1 vs. 1.03 ± 0.14 s−1, p < 0.001), radial strain (34.47 ± 9.14% vs 42.79 ± 11.91%, p < 0.001) and strain rate (1.6 ± 0.38 s−1 vs. 1.75 ± 0.29 s−1, p = 0.006), circumferential strain (−6.07 ± 2.83% vs. −18.29 ± 3.39%, p < 0.001) and strain rate (1.09 ± 0.24 s−1 vs. 1.2 ± 0.25 s−1, p = 0.004), and torsion (10.07 ± 5.18o/cm vs. 12.42 ± 5.78o/cm, p = 0.009) were significantly reduced in first-degree relatives compared with controls.
Conclusion
LV deformation parameters are impaired in first-degree relatives of patients with IDCM. Screening of this population using standard 2D echocardiography and strain imaging may provide early detection of those with subclinical myocardial dysfunction.
Zusammenfassung
Hintergrund
In der vorliegenden Studie wurde untersucht, ob bei Verwandten ersten Grades von Patienten mit idiopathischer dilatativer Kardiomyopathie (IDCM) eine subklinische Myokardfunktionsstörung auftritt. Dazu wurde die echokardiographische Deformationsbildgebung („strain imaging“) eingesetzt, bevor eine offensichtliche linkventrikuläre (LV-)Insuffizienz beobachtet wurde.
Patienten und Methoden
An der Studie nahmen 77 Probanden im Alter von 16 bis 63 Jahren teil, die Verwandte ersten Grades mit der früher oder aktuell gestellten Diagnose einer IDCM hatten. Es wurden LV-Myokarddeformationsparameter der Verwandten ersten Grades mit normaler linksventrikulärer Ejektionsfraktion in der 2‑D-Echokardiographie (LVEF ≥55%) untersucht. Die Befunde der Verwandten ersten Grades wurden mit einer nach Alter und Geschlecht entsprechend ausgesuchten Kontrollgruppe verglichen (n = 86).
Ergebnisse
Es wurden keine Unterschiede hinsichtlich Alter, Geschlecht und Körperoberfläche zwischen Verwandten ersten Grades und Kontrollen festgestellt. Verwandte ersten Grades von IDCM-Patienten wiesen signifikant niedrigere Werte für die LVEF (62,04 ± 5,8% vs. 65,65 ± 6,3%; p < 0,001) und die Verkürzungsfraktion (FS, „fractional shortening“; 39,4 ± 6,6 vs. 41,45 ± 5,5; p = 0,03) als die Kontrollen auf. Die Beurteilung der LV-Deformationsparameter ergab, dass die linksventrikuläre longitudinale globale Deformation (−17,34 ± 2,19% vs. −19,21 ± 2,16%; p < 0,001) und Deformationsrate (0,94 ± 0,14 s−1 vs. 1,03 ± 0,14 s−1; p < 0,001), die radiale Deformation (34,47 ± 9,14% vs. 42,79 ± 11,91%; p < 0,001) und Deformationsrate (1,6 ± 0,38 s−1 vs. 1,75 ± 0,29 s−1; p = 0,006), die zirkumferenzielle Deformation (−6,07 ± 2,83% vs. −18,29 ± 3,39%; p < 0,001) und Deformationsrate (1,09 ± 0,24 s−1 vs. 1,2 ± 0,25 s−1; p = 0,004) sowie die Torsion (10,07 ± 5,18o/cm vs. 12,42 ± 5,78o/cm; p = 0,009) bei den Verwandten ersten Grades im Vergleich zu den Kontrollen signifikant vermindert waren.
Schlussfolgerung
Die LV-Deformationsparameter sind bei Verwandten ersten Grades von IDCM-Patienten beeinträchtigt. Das Screening dieser Bevölkerungsgruppe unter Verwendung der Standard-2-D-Echokardiographie und der Deformationsbildgebung könnte die Früherkennung von Personen mit subklinischer Myokardfunktionsstörung ermöglichen.


Similar content being viewed by others
References
Elliott P, Anderson B, Arbustini E et al (2008) Classification of the cardiomyopathies: a position statement from the European Society of Cardiology working group on myocardial and pericardial diseases. Eur Heart J 29:270–276
Fatkin D, Otway R, Richmond Z (2010) Genetics of dilated cardiomyopathy. Heart Fail Clin 6:129–140
Michels VV, Moll PP, Miller FA et al (1992) The frequency of familial dilated cardiomyopathy in a series of patients with idiopathic dilated cardiomyopathy. N Engl J Med 326:77–82
Hershberger RE, Siegfried JD (2011) Update 2011: clinical and genetic issues in familial dilated cardiomyopathy. J Am Coll Cardiol 57:1641–1649
Petretta M, Pirozzi F, Sasso L et al (2011) Review and metaanalysis of the frequency of familial dilated cardiomyopathy. Am J Cardiol 108:1171–1176
Fatkin D, members of the CSANZ Cardiac Genetic Diseases Council Writing Group (2011) Guidelines for the diagnosis and management of familial dilated cardiomyopathy. Heart Lung Circ 20:691–693
Lakdawala NK, Funke BH, Baxter S et al (2012) Genetic testing for dilated cardiomyopathy in clinical practice. J Card Fail 18:296–303
Elliott P, Charron P, Blanes JR et al (2015) European Cardiomyopathy Pilot Registry: EURObservational Research Programme of the European Society of Cardiology. Euro Heart J. doi:10.1093/eurheartj/ehv497
Ackerman MJ, Priori SG, Willems S et al (2011) HRS/EHRA expert consensus statement on the state of genetic testing for the channelopathies and cardiomyopathies: this document was developed as a partnership between the Heart Rhythm Society (HRS) and the European Heart Rhythm Association (EHRA). Europace 13:1077–1109
Campbell N, Sinagra G, Jones KL et al (2013) Whole Exome sequencing identifies a Troponin T mutation hot spot in familial dilated cardiomyopathy. PLOS ONE 8:e78104
Yuda S, Fang ZY, Marwick TH (2003) Association of severe coronary stenosis with subclinical left ventricular dysfunction in the absence of infarction. J Am Soc Echocardiogr 16:1163–1170
Wang Q, Sun QW, Wu D et al (2015) Early detection of regional and global left ventricular myocardial function using strain and strain-rate imaging in patients with metabolic syndrome. Chin Med J (Engl) 12:226–232
Richardson P, McKenna W, Bristow M et al (1996) Report of the 1995 World Health Organization/International Society and Federation of Cardiology Task Force on the definition and classification of cardiomyopathies. Circulation 93:841–842
Kushner JD, Nauman D, Burgess D et al (2006) Clinical characteristics of 304 kindreds evaluated for familial dilated cardiomyopathy. J Card Fail 12:422–429
Serio A, Narula N, Kodama T et al (2012) Familial dilated cardiomyopathy. Clinical and genetic characteristics. Herz 37:822–829
Mestroni L, Rocco C, Gregori D et al (1999) Familial dilated cardiomyopathy: evidence for genetic and phenotypic heterogeneity. Heart Muscle Disease Study Group. J Am Coll Cardiol 34:181–190
Lang RM, Bierig M, Devereux RB et al (2006) Recommendations for chamber quantification. Eur J Echocardiogr 7:79–108
Burkett EL, Hershberger RE (2005) Clinical and genetic issues in familial dilated cardiomyopathy. J Am Coll Cardiol 45:969–981
Hershberger RE (2008) Cardiovascular genetic medicine: evolving concepts, rationale, and implementation. J Cardiovasc Transl Res 1:137–143
Baig MK, Goldman JH, Caforio AL et al (1998) Familial dilated cardiomyopathy: cardiac abnormalities are common in asymptomatic relatives and may represent early disease. J Am Coll Cardiol 31:195–201
Michels VV, Olson TM, Miller FA et al (2013) Frequency of development of idiopathic dilated cardiomyopathy among relatives of patients with idiopathic dilated cardiomyopathy. Am J Cardiol 91:1389–1392
Lakdawala NK, Thune JJ, Colan SD et al (2012) Subtle abnormalities in contractile function are an early manifestation of sarcomere mutations in dilated cardiomyopathy. Circ Cardiovasc Genet 5:503–510
Hersberger RE, Hedges DJ, Morales A (2013) Dilated cardiomyopathy: the complexity of a diverse genetic architecture. Nat Rev Cardiol 10:531–547
Caforio AL, Keeling PJ, Zachara E et al (1994) Evidence from family studies for autoimmunity in dilated cardiomyopathy. Lancet 344:773–777
Yingchoncharoen T, Agarwal S, Popović ZB et al (2013) Normal ranges of left ventricular strain: a meta-analysis. J Am Soc Echocardiogr 26:185–191
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
M. Sefa Okten, K. Tuluce, S. Yakar Tuluce, S. Kilic, H. Soner Kemal, A. Sayin, O. Vuran, B. Yagmur, I. Mutlu, E. Simsek, C. Soydas Cinar, and C. Gurgun declare that they have no competing interests.
All studies on humans described in the present manuscript were carried out with the approval of the responsible ethics committee and in accordance with national law and the Helsinki Declaration of 1975 (in its current, revised form). Informed consent was obtained from all patients included in studies.
Additional information
The results of this study were presented in part at the American College of Cardiology Scientific Sessions, 2014, Washington, DC.
Rights and permissions
About this article
Cite this article
Sefa Okten, M., Tuluce, K., Yakar Tuluce, S. et al. Screening first-degree relatives of patients with idiopathic dilated cardiomyopathy. Herz 42, 669–676 (2017). https://doi.org/10.1007/s00059-016-4498-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00059-016-4498-1
Keywords
- Cardiomyopathy, familial idiopathic
- Dilated cardiomyopathy
- Cardiomyopathy, idiopathic dilated
- Echocardiography