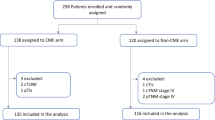
Abstract
One of the big controversies in colorectal cancer research is whether complete mesocolic excision (CME) offers a benefit for patients with carcinoma of the right colon or whether it might even harm these patients. A randomized trial to clarify this question has often been advocated. However, setting up such a trial is hampered by a major problem, which is mainly related to the difficulty of defining a standardized control group. First, there is no consensus of a standard non-CME hemicolectomy. Second, a surgeon who has been trained for CME will perform a “standard” right hemicolectomy differently than before. It won’t be the traditional hemicolectomy but a 5/6 CME-hemicolectomy, thus making it very difficult to detect a difference. Our approach to answering the question was a multicenter, non-randomized, registry-based trial with centralized assessment of the entire specimen. This trial has just closed recruitment after enrolling more than 1000 patients, so that long-term results can be expected in a few years.
Zusammenfassung
Eine der großen Kontroversen in der kolorektalen Chirurgie besteht in der Frage, ob eine komplette mesokolische Exzision (CME) zu einer Prognoseverbesserung führt. Zur Klärung dieser Frage wurde schon vielfach eine randomisierte Studie gefordert. Allerdings wäre eine solche Studie hauptsächlich aufgrund einer schwer zu definierenden Kontrollgruppe problematisch. Zum einen gibt es keine anerkannte Definition einer Standardhemikolektomie rechts, zum anderen würde ein Training der Chirurgen für die CME die Durchführung der Standardhemikolektomie stark beeinflussen. Das Ergebnis wäre ähnlich einer 5/6-CME, deren Unterlegenheit kaum nachweisbar wäre. Die Autoren haben sich daher entschlossen, einen Ansatz mit einer multizentrischen, nichtrandomisierten, registerbasierten Studie mit zentraler Aufarbeitung der Präparate zu wählen. Für diese Studie wurde gerade die Rekrutierung von über 1000 Patienten abgeschlossen. Langzeitergebnisse sind in einigen Jahren zu erwarten.

Similar content being viewed by others
References
Killeen S, Mannion M, Devaney A, Winter DC (2014) Complete mesocolic resection and extended lymphadenectomy for colon cancer: a systematic review. Colorectal Dis 16:577–594
Benz S, Tam Y, Tannapfel A, Stricker I (2015) The uncinate process first approach: a novel technique for laparoscopic right hemicolectomy with complete mesocolic excision. Surg Endosc 30(5):1930–1937. doi:10.1007/s00464-015-4417-1
Lu JY, Xu L, Xue HD, Zhou WX, Xu T, Qiu HZ, Wu B, Lin GL, Xiao Y (2016) The Radical Extent of lymphadenectomy – D2 dissection versus complete mesocolic excision of LAparoscopic Right Colectomy for right-sided colon cancer (RELARC) trial: study protocol for a randomized controlled trial. Trials 17:582
Hohenberger W, Weber K, Matzel K, Papadopoulos T, Merkel S (2009) Standardized surgery for colonic cancer: complete mesocolic excision and central ligation – technical notes and outcome. Colorectal Dis 11:354–364 (discussion 364–355)
Merkel S, Weber K, Matzel KE, Agaimy A, Gohl J, Hohenberger W (2016) Prognosis of patients with colonic carcinoma before, during and after implementation of complete mesocolic excision. Br J Surg 103:1220–1229
Bertelsen CA, Neuenschwander AU, Jansen JE, Wilhelmsen M, Kirkegaard-Klitbo A, Tenma JR, Bols B, Ingeholm P, Rasmussen LA, Jepsen LV, Iversen ER, Kristensen B, Gogenur I, Danish Colorectal Cancer G (2015) Disease-free survival after complete mesocolic excision compared with conventional colon cancer surgery: a retrospective, population-based study. Lancet Oncol 16:161–168
West NP, Morris EJ, Rotimi O, Cairns A, Finan PJ, Quirke P (2008) Pathology grading of colon cancer surgical resection and its association with survival: a retrospective observational study. Lancet Oncol 9:857–865
Lagerqvist B, Frobert O, Olivecrona GK, Gudnason T, Maeng M, Alstrom P, Andersson J, Calais F, Carlsson J, Collste O, Gotberg M, Hardhammar P, Ioanes D, Kallryd A, Linder R, Lundin A, Odenstedt J, Omerovic E, Puskar V, Todt T, Zelleroth E, Ostlund O, James SK (2014) Outcomes 1 year after thrombus aspiration for myocardial infarction. N Engl J Med 371:1111–1120
Munkedal DL, Laurberg S, Hagemann-Madsen R, Stribolt KJ, Krag SR, Quirke P, West NP (2016) Significant individual variation between pathologists in the evaluation of colon cancer specimens after complete mesocolic excision. Dis Colon Rectum 59:953–961
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
S. Benz, I. Stricker, Y. Tam, and A. Tannapfel declare that they have no competing interests.
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Informed consent was obtained from all individual participants included in the study. The protocol was approved by the ethics board of the “Landesärztekammer Baden-Württemberg” (2009-118-f).
Rights and permissions
About this article
Cite this article
Benz, S., Stricker, I., Tam, Y. et al. CME or traditional surgery for right-sided colon cancer?. coloproctology 39, 184–189 (2017). https://doi.org/10.1007/s00053-017-0158-z
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00053-017-0158-z