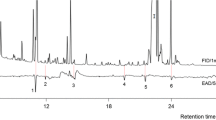
Abstract
The olive fly, Bactrocera oleae (Rossi) (Diptera: Tephritidae), is the major insect pest for olive production in the Mediterranean basin and worldwide. Monitoring of its populations is vital for efficient management to avoid yield losses. Olive fly adults are attracted to protein-based food lures. Various protein-based lures are used either for monitoring olive fly populations or for pest management in combination with insecticides. We used two techniques, i.e., dynamic headspace (DHS) and solid-phase microextraction, (SPME) for headspace collection of volatile organic compounds (VOCs) emitted from a commercially available protein-based food lure to identify specific VOCs that attract the olive flies. The collected VOCs were identified with GC–MS and electroantennographically tested with GC–EAD. Both sampling methods isolated a substantial number of VOCs but certain compounds were detected by only one of the two methods. In SPME, more alkyl-substituted pyrazines were detected rather than in DHS. VOCs from various chemical classes provoked electroantennographic responses. Μature mated females gave more responses compared to virgin ones. Both age groups of B. oleae female adults showed electrophysiological responses to 2,5-dimethyl-pyrazine which had lower abundance than the major compound 2,3,5-trimethyl pyrazine. Selected compounds were tested in field trials for attraction of olive fly adults. The highest number of olive fly adults was caught with sticky traps baited with the terpene nonanal. The importance of the collection method in identifying VOCs that might contribute to better monitoring and management of olive fly populations in the field is discussed.



Similar content being viewed by others
Data availability
All data are available to requests to the corresponding author.
References
Adams RP (2007) Identification of essential oil components by gas-chromatography/mass spectrometry, 4th edn. Allured Business Media, Illinois
Anastasaki E, Balayannis G, Papanikolaou NE, Michaelakis N, Milonas PG (2015) Oviposition induced volatiles in tomato plants. Phytochem Lett 13:262–266. https://doi.org/10.1016/j.phytol.2015.07.007
Anastasaki E, Drizou F, Milonas PG (2018) Electrophysiological and oviposition responses of Tuta absoluta females to herbivore-induced volatiles in tomato plants. J Chem Ecol 44:288–298. https://doi.org/10.1007/s10886-018-0929-1
Anastasaki E, Psoma A, Partsinevelos G, Papachristos DP, Milonas PG (2021) Electrophysiological responses of Philaenus spumarius and Neophilaenus campestris females to plant volatiles. Phtyochemistry 189:112848. https://doi.org/10.1016/j.phytochem.2021.112848
Biasazin TD, Chernet HT, Herrera SL, Bengtsson M, Karlsson MF, Lemmen-Lechelt JK, Dekker T (2018) Detection of volatile constituents from food lures by Tephritid fruit flies. InSects 9(3):119–133. https://doi.org/10.3390/insects9030119
Bohman B, Phillips RD, Menz MHM, Berntsson BW, Flematti GR, Barrow RA, Dixon KW, Peakall R (2014) Discovery of pyrazines as pollinator sex pheromones and orchids semiochemicals: implications for the evolution of sexual deception. New Phytol 203:939–952. https://doi.org/10.1111/nph.12800
Bortoli LC, Machota R Jr, Mello Garcia FR, Botton M (2016) Evaluation of food lures for fruit flies (Diptera: Tephritidae) captured in a citrus orchard of the Serra Gaúcha. Fla Entomol 99(3):381–384. https://doi.org/10.1653/024.099.0307
Calcagnile M, Tredici SM, Talà A, Alifano P (2019) Bacterial semiochemicals and transkingdom interactions with insects and plants. InSects 10:441. https://doi.org/10.3390/insects10120441
Canale A, Benelli G, Germinara GS, Fusini G, Romano D, Rapalini F, Desneux N, Rotundo G, Raspi A, Carpita A (2015) Behavioural and electrophysiological responses to overlooked female pheromone components in the olive fruit fly, Bactrocera oleae (Diptera: Tephritidae). Chemoecology 25:147–157. https://doi.org/10.1007/s00049-014-0183-0
García-García CR, Parrón T, Requena M, Alarcón R, Tsatsakis AM, Hernández AF (2016) Occupational pesticide exposure and adverse health effects at the clinical, hematological and biochemical level. Life Sci 145:274–283. https://doi.org/10.1016/j.lfs.2015.10.013
Gerofotis CD, Ioannou CS, Papadopoulos NT (2013) Aromatized to find mates: α-pinene aroma boosts the mating success of adult olive fruit flies. PLoS ONE 8(11):e81336. https://doi.org/10.1371/journal.pone.0081336
Hernandez MM, Sanz I, Adelantado M, Ballach S, Primo E (1996) Electroantennogram activity from antennae of Ceratitis capitata (WIED.) to fresh orange airborne volatiles. J Chem Ecol 22(9):1607–1619. https://doi.org/10.1007/BF02272401
Kokkari AI, Milonas PG, Anastasaki E, Floros GD, Kouloussis NA, Koveos DS (2021) Determination of volatile substances in olives and their effect on reproduction of the olive fruit fly. J Appl Entomol 145(9):841–855. https://doi.org/10.1111/jen.12929
Liscia A, Angioni P, Sacchetti P, Poddighe S, Granchietti A, Setzu MD, Belcari A (2013) Characterization of olfactory sensilla of the olive fly: behavioral and electrophysiological responses to volatile organic compounds from the host plant and bacterial filtrate. J Insect Physiol 59:705–716. https://doi.org/10.1016/j.jinsphys.2013.04.008
Malheiro R, Casal S, Cunha SC, Baptista P, Pereira JA (2015a) Olive volatiles from Portuguese cultivars Cobrançosa, Madural and Verdeal Transmontana: role in oviposition preference of Bactrocera oleae (Rossi) (Diptera: Tephritidae). PLoS ONE 10(5):e0125070. https://doi.org/10.1371/journal.pone.0125070
Malheiro R, Ortiz A, Casal S, Baptista P, Pereira JA (2015b) Electrophysiological response of Bactrocera oleae (Rossi) (Diptera:Tephritidae) adults to olive leaves essential oils from different cultivars and olive tree volatiles. Ind Crops Prod 77:81–88. https://doi.org/10.1016/j.indcrop.2015.08.046
Manrakhan A, Lux SA (2006) Contribution of natural food sources to reproductive behaviour, fecundity and longevity of Ceratitis cosyra, C. fasciventris and C. capitata (Diptera: Tephritidae). Bull Entomol Res 96(3):259–268. https://doi.org/10.1079/ber2006421
Mazomenos BE, Haniotakis GE (1981) A multicomponent female sex pheromone of dacus oleae gmelin: isolation and bioassay. J Chem Ecol 7(2):437–444. https://doi.org/10.1007/BF00995766
Njihia TN, Torto B, Murungi LK, Irungu J, Mwenda DM, Babin R (2018) Ripe coffee berry volatiles repel second instar nymphs of Antestia bugs (Heteroptera: Pentatomidae: Antestiopsis thunbergii). Chemoecology 28:91–100. https://doi.org/10.1007/s00049-018-0259-3
NIST standard reference data. http://webbook.nist.gov/chemistry/name-ser.html
Pinto-Zevallos DM, Strapasson P, Zarbin PHG (2016) Herbivore-induced volatile organic compounds emitted by maize: electrophysiological responses in Spodoptera frugiperda females. Phytochem Lett 16:70–74. https://doi.org/10.1016/j.phytol.2016.03.00
Rasgado MA, Malo EA, Cruz-Lόpez L, Rojas JC, Toledo J (2009) Olfactory response of the Mexican fruit fly (Diptera: Tephritidae) to Citrus aurantium volatiles. J Econ Entomol 102(2):585–594. https://doi.org/10.1603/029.102.0216
Robacker DC, Bartelt RJ (1997) Chemicals attractive to Mexican fruit fly from Klebsiella pneumoniae and Citrobacter freundii cultures sampled by solid-phase microextraction. J Chem Ecol 23(12):2897–2915. https://doi.org/10.1023/A:1022579414233
Robacker DC, Flath RA (1995) Attractants from Staphylococcus aureus cultures for Mexican fruit fly, Anastrepha ludens. J Chem Ecol 21(11):1861–1874. https://doi.org/10.1007/BF02033682
Scarpati ML, Lo Scalzo R, Vita G (1993) Olea europaea volatiles attractive and repellent to the olive fruit fly (Dacus oleae, Gmelin). J Chem Ecol 19(4):881–891. https://doi.org/10.1007/BF00985017
Scarpati ML, Lo Scalzo R, Vita G, Gambacorta A (1996) Chemiotropic behavior of female olive fly (Bactrocera oleae GMEL.) on Olea europaea L. J Chem Ecol 22(5):1027–1036. https://doi.org/10.1007/BF02029952
Varikou K, Alexandrakis V, Gika V, Birouraki A, Marnelakis C, Sergentani C (2013) Estimation of fly population density of Bactrocera oleae in olive groves of Crete. Phytoparasitica 41:105–111. https://doi.org/10.1007/s12600-012-0270-0
Varikou K, Garantonakis N, Birouraki A (2014a) Response of olive fruit fly Bactrocera oleae to various attractants combinations, in orchards of Crete. Bull Insectol 67(1):109–114
Varikou K, Garantonakis N, Birouraki A (2014b) Comparative field studies of Bactrocera oleae baits in olive orchards in Crete. J Crop Prot 65:238–243. https://doi.org/10.1016/j.cropro.2014.08.005
Acknowledgements
We would like to thank two anonymous reviewers for their constructive comments.
Funding
This research has been partially funded by the Public Investments Program of General Secretariat for Research & Technology (GSRT), under the Emblematic Action “The Olive Road” (project code: 2018ΣE01300000).
Author information
Authors and Affiliations
Contributions
PM contributed to the study conception and design. Material preparation, data collection and analysis were performed by EA, AP and GP. The manuscript was written by PM, EA and AP and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no competing interests to declare that are relevant to the content of this article.
Additional information
Communicated by Günther Raspotnig.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Psoma, A., Anastasaki, E., Partsinevelos, G. et al. Isolation and identification of volatile compounds from a protein-based food lure: electrophysiological and behavioral responses of Bactrocera oleae adults. Chemoecology 33, 99–112 (2023). https://doi.org/10.1007/s00049-023-00388-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-023-00388-w