Abstract
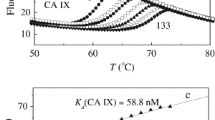
Inhibition of carbonic anhydrases is a potential avenue for treatment of various pathological conditions such as cancer, glaucoma, epileptic seizures, obesity, and altitude sickness. In this study, a series of 4-benzamidophenyl sulfamate derivatives were synthesized and tested against two carbonic anhydrase isoforms namely, carbonic anhydrase II (CA II) and carbonic anhydrase IX (CA IX). All the synthesized compounds inhibited the activity of both isoforms CA II and CA IX at micromolar IC50 concentrations. Compound 1n was found to be the most potent against CA II with an IC50 value of 0.78 μM, whereas compound 1f was the most potent inhibitor against CA IX with an IC50 value of 0.34 μM. Molecular modeling studies of the most potent compounds revealed a network of interactions with important amino acids residues in the active sites of CA II and CA IX.
Graphical Abstract

















Similar content being viewed by others
References
Supuran CT. Emerging role of carbonic anhydrase inhibitors. Clin Sci. 2021;135:1233–49.
Supuran CT. Carbonic anhydrase inhibitors. Bioorg Med. Chem Lett. 2010;20:3467–74.
Abdel-Mohsen HT, et al. New thiopyrimidine-benzenesulfonamide conjugates as selective carbonic anhydrase II inhibitors: synthesis, in vitro biological evaluation, and molecular docking studies. Bioorg Med Chem. 2020;28:115329.
Supuran CT. Structure and function of carbonic anhydrases. Biochem J. 2016;473:2023–32.
Li Y, et al. Catalysis and pH control by membrane-associated carbonic anhydrase IX in MDA-MB-231 breast cancer cells. J Biol Chem. 2011;286:15789–96.
Mahon BP, et al. The structure of carbonic anhydrase IX is adapted for low-pH catalysis. Biochemistry. 2016;55:4642–53.
Becker HM, Deitmer JW. Carbonic anhydrase II increases the activity of the human electrogenic Na+/HCO 3-cotransporter. J Biol Chem. 2007;282:13508–21.
Kirkpatrick JP, et al. Elevated CAIX expression is associated with an increased risk of distant failure in early-stage cervical cancer. Biomark Insights. 2008;3:BMI. S570.
Becker HM, Deitmer JW. Proton transport in cancer cells: the role of carbonic anhydrases. Int J Mol Sci. 2021;22:3171.
Gatenby RA, Gillies RJ. A microenvironmental model of carcinogenesis. Nat Rev Cancer. 2008;8:56–61.
Parks SK, Chiche J, Pouyssegur J. pH control mechanisms of tumor survival and growth. J Cell Physiol. 2011;226:299–308.
Supuran CT. Carbonic anhydrases: novel therapeutic applications for inhibitors and activators. Nat Rev Drug Discov. 2008;7:168–81.
Thiry A, et al. Carbonic anhydrase inhibitors as anticonvulsant agents. Curr Top Med Chem. 2007;7:855–64.
Kivelä AJ, et al. Expression of transmembrane carbonic anhydrase isoenzymes IX and XII in normal human pancreas and pancreatic tumours. Histochem Cell Biol. 2000;114:197–204.
Kummola L, et al. Expression of a novel carbonic anhydrase, CA XIII, in normal and neoplastic colorectal mucosa. BMC Cancer. 2005;5:1–7.
Barnett DH, et al. Estrogen receptor regulation of carbonic anhydrase XII through a distal enhancer in breast cancer. Cancer Res. 2008;68:3505–15.
Kaar JL, et al. Towards improved artificial lungs through biocatalysis. Biomaterials. 2007;28:3131–9.
Bond GM, et al. Development of integrated system for biomimetic CO2 sequestration using the enzyme carbonic anhydrase. Energy Fuels. 2001;15:309–16.
Jakubowski, M., E. Szahidewicz-Krupska, and A. Doroszko, The human carbonic anhydrase II in platelets: an underestimated field of its activity. BioMed Res Int. 2018; 2018:4548353.
George RF, et al. Synthesis and selective inhibitory effects of some 2-oxindole benzenesulfonamide conjugates on human carbonic anhydrase isoforms CA I, CA II, CA IX and CAXII. Bioorg Chem. 2020;95:103514.
Petrenko M, et al. Combined 3-O-acetylbetulin treatment and carbonic anhydrase IX inhibition results in additive effects on human breast cancer cells. Chem-Biol Interact. 2021;333:109326.
Ali M, et al. Benzylaminoethyureido-tailed benzenesulfonamides: Design, synthesis, kinetic and X-ray investigations on human carbonic anhydrases. Int J Mol Sci. 2020;21:2560.
Hou R, et al. Zinc enzymes in medicinal chemistry. Eur J Med Chem. 2021;226:113877.
Bozdag M, et al. N-aryl-N’-ureido-O-sulfamates: Potent and selective inhibitors of the human Carbonic Anhydrase VII isoform with neuropathic pain relieving properties. Bioorg Chem. 2019;89:103033.
Supuran CT. Experimental carbonic anhydrase inhibitors for the treatment of hypoxic tumors. J Exp Pharmacol. 2020;12:603.
Moi D, et al. Structure-activity relationship with pyrazoline-based aromatic sulfamates as carbonic anhydrase isoforms I, II, IX and XII inhibitors: Synthesis and biological evaluation. Eur J Med Chem. 2019;182:111638.
Daryadel S, et al. Novel sulfamate derivatives of menthol: Synthesis, characterization, and cholinesterases and carbonic anhydrase enzymes inhibition properties. Arch Pharm. 2018;351:1800209.
Nocentini A, et al. Discovery of thiazolin-4-one-based aromatic sulfamates as a new class of carbonic anhydrase isoforms I, II, IV, and IX inhibitors. Bioorg Chem. 2018;77:293–9.
Williams KJ, Gieling RG. Preclinical evaluation of ureidosulfamate carbonic anhydrase IX/XII inhibitors in the treatment of cancers. Int J Mol Sci. 2019;20:6080.
Othman IMM, et al. Toward a treatment of antibacterial and antifungal infections: Design, synthesis and in vitro activity of novel arylhydrazothiazolylsulfonamides analogues and their insight of DFT, docking and molecular dynamic simulations. J Mol Struct. 2021;1243:130862.
Ezugwu JA, et al. Design, Synthesis, Molecular Docking, Molecular Dynamics and In Vivo Antimalarial Activity of New Dipeptide-Sulfonamides. ChemistrySelect. 2022;7:e202103908.
Rudrapal, M, et al., Analgesic and Anti-Inflammatory Potential of Indole Derivatives. Polycyclic Aromatic Compounds. https://www.tandfonline.com/doi/full/10.1080/10406638.2022.2139733?scroll=top&needAccess=true&role=tab.
El-Gamal MI, et al. A new series of aryl sulfamate derivatives: design, synthesis, and biological evaluation. Bioorg Med Chem. 2020;28:115406.
Zaib S, et al. New aminobenzenesulfonamide–thiourea conjugates: Synthesis and carbonic anhydrase inhibition and docking studies. Eur J Med Chem. 2014;78:140–50.
MOE (Molecular Operating Environment) Version 2019.0201. Chemical Computing Group, (CCG).
LeadIT version 2.3.2; BioSolveIT GmbH, Sankt Augustin, Germany, 2017.
Dassault Syst’emes BIOVIA, Discovery Studio Modeling Environment, Release 2017, Dassault Syst’emes, San Diego, 2016.
Bowers, K.J, et al. Scalable algorithms for molecular dynamics simulations on commodity clusters. In SC'06: Proceedings of the 2006 ACM/IEEE Conference on Supercomputing. 2006. IEEE.
Shivakumar D, et al. Prediction of absolute solvation free energies using molecular dynamics free energy perturbation and the OPLS force field. J Chem Theory Comput. 2010;6:1509–19.
Barclay PL, Zhang DZ. Periodic boundary conditions for arbitrary deformations in molecular dynamics simulations. J Comput. Phys. 2021;435:110238.
Martyna GJ, Tobias DJ, Klein ML. Constant pressure molecular dynamics algorithms. J Chem Phys. 1994;101:4177–89.
Luty BA, et al. A comparison of particle-particle, particle-mesh and Ewald methods for calculating electrostatic interactions in periodic molecular systems. Mol Simul. 1994;14:11–20.
Humphreys DD, Friesner RA, Berne BJ. A multiple-time-step molecular dynamics algorithm for macromolecules. J Phys Chem A. 1994;98:6885–92.
Acknowledgements
Authors acknowledge University of Sharjah, United Arab Emirates, for financially supporting this project (grant No. 2201110159 & Drug Design and Discovery Research Group operational fund). The authors gratefully acknowledge the financial support for this research provided by the Higher Education Commission of Pakistan (HEC) via NRPU project No. 20-15846/NRPU/R&D/HEC/2021, German-Pakistani Research Collaboration Programme and Equipment Grant funded by DAAD, Germany.
Funding
This study was funded by University of Sharjah, United Arab Emirates (grant No. 2201110159 & Drug Design and Discovery Research Group operational fund). The authors gratefully acknowledge the financial support for this research provided by the Higher Education Commission of Pakistan (HEC) via NRPU project No. 20-15846/NRPU/R&D/HEC/2021, German-Pakistani Research Collaboration Programme and Equipment Grant funded by DAAD, Germany.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Informed consent
All the authors have read the article content and agree on it. They also agree on submitting it to Medicinal Chemistry Research.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jalil, S., Ullah, S., Zaraei, SO. et al. Synthesis and biological evaluation of sulfamate derivatives as inhibitors of carbonic anhydrases II and IX. Med Chem Res 32, 869–883 (2023). https://doi.org/10.1007/s00044-023-03043-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-023-03043-9