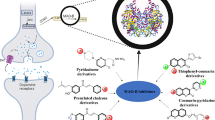
Abstract
In this work, a series of indoline derivatives as multifunctional neuroprotective agents for battling ischemic stroke were designed, synthesized, and biologically evaluated. In antioxidant assay, all compounds showed significant protective effects against H2O2-induced death of RAW 264.7 cells. In oxygen glucose deprivation/reperfusion (OGD/R)-induced neuronal damage, some compounds significantly elevated the cell survival rate. Among them, 7i, 7j and 7r exerted comparable neuroprotective effects to ifenprodil, and exhibited binding affinity to N-methyl-D-aspartic acid receptors 2B (NMDA-GluN2B). At the concentrations of 0.1, 1 and 10 μM, 7i, 7j and 7r dose-dependently lowered the LPS-induced secretion of inflammatory cytokines, including TNF-α, IL-6 and NO, by BV-2 cells. Importantly, 7i and 7j can dramatically reduce the cerebral infarction rate and improve neurological deficit scores in middle cerebral artery occlusion (MCAO) rat model. As demonstrated by the above results, 7i and 7j are potential neuroprotective agents for the treatment of ischemic stroke.

Graphical abstract







Similar content being viewed by others
References
Wu J, Wang B, Li M, Shi YH, Wang C, Kang YG. Network pharmacology identification of mechanisms of cerebral ischemia injury amelioration by Baicalin and Geniposide. Eur J Pharmacol 2019;859:172484 https://doi.org/10.1016/j.ejphar.2019.172484.
Feigin VL, Forouzanfar MH, Krishnamurthi R, Mensah GA, Connor M, Bennett DA, et al. Global and regional burden of stroke during 1990–2010: findings from the Global Burden of Disease Study 2010. Lancet 2014;383:245–55. https://doi.org/10.1016/S0140-6736(13)61953-4
Sommer, Clemens J. Ischemic stroke: experimental models and reality. Acta Neuropathol. 2017;133:245–61. https://doi.org/10.1007/s00401-017-1667-0
Roth GA, Forouzanfar MH, Moran AE, Barber R, Nguyen G, Feigin VL, et al. Demographic and epidemiologic drivers of global cardiovascular mortality. N. Engl J Med. 2015;372:1333–41. https://doi.org/10.1056/NEJMoa1406656
Deb P, Sharma S, Hassan KM. Pathophysiologic mechanisms of acute ischemic stroke: an overview with emphasis on therapeutic significance beyond thrombolysis. Pathophysiology. 2010;17:197–18. https://doi.org/10.1016/j.pathophys.2009.12.001
Jung S, Gralla J, Fischer U, Mono ML, Weck A, Ludi R, et al. Safety of endovascular treatment beyond the 6-h time window in 205 patients. Eur J Neurol. 2013;20:865–71. https://doi.org/10.1111/ene.12069
Ovbiagele B, Kidwell CS, Starkman S, Saver JL. Potential role of neuroprotective agents for the treatment of patients with acute ischemic stroke. Curr Treat Option Ne. 2003;3:9–20. https://doi.org/10.1007/s11940-003-0027-7
Luo Y, Tang H, Li H, Zhao R, Huang QH, Liu JM. Recent advances in the development of neuroprotective agents and therapeutic targets in the treatment of cerebral ischemia. Eur J Med Chem. 2019;162:132–46. https://doi.org/10.1016/j.ejmech.2018.11.014
Uzdensky AB. Photothrombotic stroke as a model of ischemic stroke. Transl Stroke Res. 2018;9:437–51. https://doi.org/10.1007/s12975-017-0593-8
Tewes B, Frehland B, Schepmann D, Schmidtke KU, Winckler T, Wunsch B. Conformationally constrained NR2B selective NMDA receptor antagonists derived from ifenprodil: Synthesis and biological evaluation of tetrahydro-3-benzazepine-1,7-diols. Bioorg Med Chem. 2010;18:8005–15. https://doi.org/10.1016/j.bmc.2010.09.026
Nakase T, Yamazaki T, Ogura N, Suzuki A, Nagata K. The impact of inflammation on the pathogenesis and prognosis of ischemic stroke. J Neurol Sci. 2008;271:104–9. https://doi.org/10.1016/j.jns.2008.03.020
Terao S, Yilmaz G, Stokes KY, Russell J, Ishikawa M, Kawase T, et al. Blood cell-derived RANTES mediates cerebral microvascular dysfunction, inflammation and tissue injury after focal ischemia-reperfusion. Stoke. 2008;39:2560–70. https://doi.org/10.1161/strokeaha.107.513150
Pei J, You X, Fu Q. Inflammation in the pathogenesis of ischemic stroke. Front Biosci (Landmark Ed). 2015;20:772–83. https://doi.org/10.2741/4336
Zeeli S, Weill T, Finkin-Groner E, Bejar C, Melamed M, Furman S, et al. Synthesis and biological evaluation of derivatives of indoline as highly potent antioxidant and antiinflammatory agents. J Med Chem. 2018;61:4004–19. https://doi.org/10.1021/acs.jmedchem.8b00001
Doyle KP, Simon RP, Stenzel-Poore MP. Mechanisms of ischemic brain damage. Nueropharmacology. 2008;55:310–8. https://doi.org/10.1016/j.neuropharm.2008.01.005
Benveniste H, Drejer J, Schousboe A, Diemer NH. Elevation of the extracellular concentrations of glutamate and aspartate in rat hippocampus during transient cerebral ischemia monitored by intracerebral microdialysis. J Neurochem. 1984;43:1369–74. https://doi.org/10.1111/j.1471-4159.1984.tb05396.x
Lai TW, Zhang S, Wang YT. Excitotoxicity and stroke: idenfying novel targets for neuroprotection. Prog Neurobiol 2014;115:157–88. https://doi.org/10.1016/j.pneurobio.2013.11.006
Menniti F, Chenard B, Collins M, Ducat M, White F. CP-101,606, a potent neuroprotectant selective for forebrain neurons. Eur J Pharm. 1997;331:117–26. https://doi.org/10.1016/S0014-2999(97)10092-9
Henter ID, De Sousa RT, Zarate CA. Glutamatergic modulators in depression. Harv Rev Psychiatry. 2018;26:307–19. https://doi.org/10.1097/hrp.0000000000000183
Machado-Vieira R, Henter ID, Zarate CA. New targets for rapid antidepressant action. Prog Neurobiol. 2017;152:21–37. https://doi.org/10.1016/j.pneurobio.2015.12.001
Stroebel D, Buhl DL, Knafels JD, Chanda PK, Gree M, Sciabola S, et al. A novel binding mode reveals two distinct classes of NMDA receptor GluN2B-selective antagonists. Mol Pharm. 2016;89:541–51. https://doi.org/10.1124/mol.115.103036
Karakas E, Simorowski N, Furukawa H. Subunit arrangement and phenylethanolamine binding in GluN1/GluN2B NMDA receptors. Nature. 2011;475:249–53. https://doi.org/10.1038/nature10180
Li J, Nie CB, Qian Y, Hu J, Li QF, Wang Q, et al. Design, synthesis and biological evaluation of novel 2,3,4,9-tetrahydro-1H-pyrido[3,4-b]indole triazole derivatives as potent TRPV1 antagonists. Eur J Med Chem. 2019;178:433–45. https://doi.org/10.1016/j.ejmech.2019.06.007
Estopina-Duran S, Mclean EB, Donnelly LJ, Hockin BM, Taylor JE. Arylboronic acid catalyzed C-alkylation and allylation reactions using benzylic alcohols. Org Lett. 2020;22:7547–51. https://doi.org/10.1021/acs.orglett.0c02736
Chen ZW, Lin HX, Han J, Fang DM, Wang M, Liao J. Enantioselective copper-catalyzed electrophilic sulfenylation of cyclic imino esters. Org Lett 2021;23:1946–50. https://doi.org/10.1021/acs.orglett.1c03464
Seliger J, Oestreich M. Dynamic kinetic resolution of alcohols by enantioselective silylation enabled by two orthogonal transition‐metal catalysts. Angew Chem Int Ed. 2020;60:247–51. https://doi.org/10.1002/anie.202010484
Robert Z, Baumeister S, Dirk S, Wunsch B. Pyridine bioisosteres of potent GluN2B subunit containing NMDA receptor antagonists with benzo[7]annulene scaffold. Eur J Med Chem. 2018;157:397–04. https://doi.org/10.1016/j.ejmech.2018.08.003
Temme L, Bechthold E, Schreiber JA, Gawaskar S, Schepmann D, Robaa D, et al. Negative allosteric modulators of the GluN2B NMDA receptor with phenylethylamine structure embedded in ring-expanded and ring-contracted scaffolds. Eur J Med Chem. 2020;190:112138 https://doi.org/10.1016/j.ejmech.2020.112138
Chaffey H, Chazot PL. NMDA receptor subtypes: structure, function and therapeutics. Anaesth Crit Care. Pain Med. 2008;19:183–01. https://doi.org/10.1016/j.cacc.2008.05.004.
Tang N, Wu JH, Zhu H, Yan HL, Guo Y, Cai Y, et al. Genetic mutation of GluN2B protects brain cells against stroke damages. Mol Neurobiol. 2018;55:2979–90. https://doi.org/10.1007/s12035-017-0562-y
Zhang BY, Wang GR, Ning WH, Liu J, Yang S, Shen Y, et al. Electroacupuncture pretreatment elicits tolerance to cerebral ischemia/reperfusion through inhibition of the GluN2B/m-Calpain/p38 MAPK Proapoptotic Pathway. Neural Plast. 2020;2020:1–14. https://doi.org/10.1155/2020/8840675
Huang L, Wang J, Chen L, Zhu C, Wu SB, Chu SH, et al. Design, synthesis, and evaluation of NDGA analogues as potential anti-ischemic stroke agents. Eur J Med Chem. 2018;143:1165–73. https://doi.org/10.1016/j.ejmech.2017.09.028
Abdel-Salam OM, Baiuomy AR, EI-Shenawy SM, Aribid MS. The anti-inflammatory effects of the phosphodiesterase inhibitor pentoxifylline in the rat. Pharmacol Res 2003;47:331–40. https://doi.org/10.1016/S1043-6618(03)00002-1
Gitto R, Luca LD, Ferro S, Buemi MR, Russo E, Sarro GD, et al. Synthesis and biological characterization of 3-substituted-1H-indoles as ligands of GluN2B-containing N-methyl-D-aspartate receptors. Bioorgan Med Chem. 2014;22:1040–8. https://doi.org/10.1016/j.bmc.2013.12.040
Hu H, Li Z, Zhu X, Li RH, Lin JM, Peng J, et al. Gua Lou Gui Zhi decoction suppresses LPS-induced activation of the TLR4/NF-κB pathway in BV-2 murine microglial cells. Int J Mol Med. 2013;31:1327–32. https://doi.org/10.3892/ijmm.2013.1331
Durukan A, Tatlisumak T. Acute ischemic stroke: overview of major experimental rodent models, pathophysiology, and therapy of focal cerebral ischemia. Pharm Blochem Be. 2007;87:179–97. https://doi.org/10.1016/j.pbb.2007.04.015
Ballabh P, Braun A, Nedergaard M. The blood–brain barrier: an overview Structure, regµlation, and clinical imPlications. Neurobiol Dis. 2004;16:1–13. https://doi.org/10.1016/j.nbd.2003.12.016
Acknowledgements
This work was Supported by the Key Project of Natural Science Research in Universities of Anhui Province (No. KJ2020A0412), and the University-Enterprise Cooperative Project (No. 2020HZ062).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Du, S., Jin, F., Li, J. et al. Design, synthesis and biological evaluation of indoline derivatives as multifunctional agents for the treatment of ischemic stroke. Med Chem Res 31, 805–818 (2022). https://doi.org/10.1007/s00044-022-02875-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-022-02875-1