Abstract
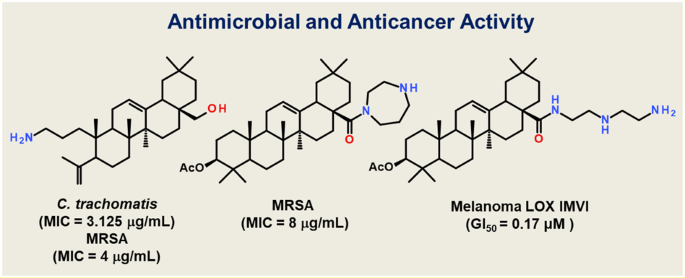
A series of nitrogen-containing modificants with amide, arylidene or heterocyclic fragments of oleanolic, oleanonic and 2,3-indolo-oleanolic acids have been synthesized and evaluated for activity against C. trachomatis and key ESKAPE pathogens. Oleanolic acid conjugates with homopyperazine 3, N-hydroxymethyl-homopyperazine 4, and diethylenetriamine 23 demonstrated a high inhibitory activity against C. trachomatis with chemotherapeutic index (CTI) 8 and >8, while 3-amino-3,4-seco-4(23)-en-erythrodiol 22 was found to be a leader compound with significant activity (MIC 3.125 μg/mL). Compounds 3 and 22 showed a moderate activity against MRSA with MICs of 8 and 4 μg/mL. Compounds 2, 3, and 23 exhibited remarkable activities against NCI-60 subpanel (GI50 ranges from 0.18 to 2.21 μM) exceeding the activity of sorafenib with compound 23 as a leader (GI50 0.17 μM for melanoma LOX IMVI).






Similar content being viewed by others
References
Pollier J, Goossens A. Oleanolic acid. Phytochem 2012;77:10–15. https://doi.org/10.1016/j.phytochem.2011.12.022
Sultana N, Ata A. Oleanolic acid and related derivatives as medicinally important compounds. J Enzym Inhib Med Chem. 2008;23:739–56. https://doi.org/10.1080/14756360701633187
Vyas N, Argal A. Isolation and characterization of oleanolic acid from roots of Lantana camara. Asian J Pharm Clin Res. 2014;7:189–91.
Xia EQ, Wang BW, Xu XR, Zhu L, Song Y, Li HB. Microwave-assisted extraction of oleanolic acid and ursolic acid from Ligustrum lucidum. Ait Int J Mol Sci. 2011;12:5319–29. https://doi.org/10.3390/ijms12085319
Verma SC, Jain CL, Nigam S, Padhi MM. Rapid extraction, isolation, and quantification of oleanolic acid from Lantana camara L. roots using microwave and HPLC-PDA techniques. Acta Chromatogr. 2013;25:181–99. https://doi.org/10.1556/AChrom.25.2013.1.12
Kim S, Lee H, Lee S, Yoon Y, Choi K-H. Antimicrobial action of oleanolic acid on listeria monocytogenes, enterococcus faecium, and enterococcus faecalis. PLoS ONE. 2015;10:e0118800. https://doi.org/10.1371/journal.pone.0118800
Wang J, Ren H, Xu QL, Zhou ZY, Wu P, Wei XY, et al. Antibacterial oleanane-type triterpenoids from pericarps of Akebia trifoliate. Food Chem. 2015;168:623–29. https://doi.org/10.1016/j.foodchem.2014.07.105
Ye M, Liao Y, Wu L, Qi W, Choudhry N, Liu Y, et al. An oleanolic acid derivative inhibits hemagglutinin-mediated entry of influenza A virus. Viruses. 2020;12:225. https://doi.org/10.3390/v12020225
Wang X, Ye X, Liu R, Chen HL, Bai H, Liang X. Antioxidant activities of oleanolic acid in vitro: possible role of Nrf2 and MAP kinases. Chem-Biol Interact. 2010;184:328–37. https://doi.org/10.1016/j.cbi.2010.01.034
Castellano JM, Guinda A, Macías L, Santos-Lozano JM, Lapetra J, Rada M. Free radical scavenging and glucosidase inhibition, two potential mechanisms involved in the anti-diabetic activity of oleanolic acid. Grasasy Aceites. 2016;67:e142. https://doi.org/10.3989/gya.1237153
Shanmugam MK, Dai X, Kumar AP, Tan BK, Sethi G, Bishayee A. Oleanolic acid and its synthetic derivatives for the prevention and therapy of cancer: Preclinical and clinical evidence. Cancer Lett. 2014;346:206–16. https://doi.org/10.1016/j.canlet.2014.01.016
Amara S, Zheng M, Tiriveedhi V. Oleanolic acid inhibits high salt-induced exaggeration of warburg-like metabolism in breast cancer cells. Cell Biochem Biophys 2016;74:427–34. https://doi.org/10.1007/s12013-016-0736-7
Nyakudya T, Mukwevho E, Nkomozepi P, Swanepoel E, Erlwanger KH. Early postnatal administration of oleanolic acid attenuates the development of non-alcoholic fatty liver disease in fructose fed adult female rats. FASEB J. 2017;31:2. https://doi.org/10.1017/S2040174420000124
Jesus JJ, Lago JHG, Laurenti MD, Yamamoto ES, Passero LFD. Antimicrobial activity of oleanolic acids. evid based complement alternat med. 2015. https://doi.org/10.1155/2015/620472.
Liu Q, Niu H, Zhang W, Mu H, Sun C, Duan J. Synergy among thymol, eugenol, berberine, cinnamaldehyde and streptomycin against planktonic and biofilm-associated food-borne pathogens. Lett Appl Microbiol. 2015;60:21–30. https://doi.org/10.1111/lam.12401
Feng A, Yang S, Sun Y, Zhang L, Bo F, Li L. Development and evaluation of oleanolic acid dosage forms and its derivatives. BioMed Res. 2020. https://doi.org/10.1155/2020/1308749. [Online first article]
Chouaïb K, Hichri F, Nguir A, Daami-Remadi M, Elie N, Touboul D, et al. Semi-synthesis of new antimicrobial esters from the natural oleanolic and maslinic acids. Food Chem. 2015;183:8–17. https://doi.org/10.1016/j.foodchem.2015.03.018
Kazakova OB, Brunel JM, Khusnutdinova EF, Negrel S, Giniyatullina GV, Lopatina TV et al. A-ring modified triterpenoids and their spermidine-aldimines with strong antibacterial activity. Molbank. 2019. https://doi.org/10.3390/M1078.
Medvedeva NI, Kazakova OB, Lopatina TV, Smirnova IE, Giniyatullina GV, Baikova IP, et al. Synthesis and antimycobacterial activity of triterpenic A-ring azepanes. Eur J Med Chem. 2018;143:464–72. https://doi.org/10.1016/j.ejmech.2017.11.035
Porritt RA, Crother TR. Chlamydia pneumoniae. Infection and inflammatory diseases. Immunopathol Dis Ther. 2016;7:237–54. https://doi.org/10.1615/ForumImmunDisTher.2017020161
Alakurtti S, Mäkelä T, Koskimies S, Yli-Kauhaluoma J. Pharmacological properties of the ubiquitous natural product betulin. Eur J Pharm Sci. 2006;29:1–13. https://doi.org/10.1016/j.ejps.2006.04.006
Salin O, Alakurtti S, Pohjala L, Siiskonen A, Maass V, Maass M, et al. Inhibitory effect of the natural product betulin and its derivatives against the intracellular bacterium Chlamydia pneumoniae. Biochem Pharm. 2010;80:1141–51. https://doi.org/10.1016/j.bcp.2010.06.051
Tazoo D, Krohn K, Hussain H, Kouam SF, Dongo E. Laportoside A and laportomide A: a new cerebroside and a new ceramide from leaves of Laportea ovalifolia. Z Naturforsch. 2007;62B:12081212.
Antoine KZ, Hussain H, Dongo E, Kouam SF, Schulz B, Krohn K, et al. A new ceramide from Helichrysum cameroonensei. J Asian Nat Prod Res. 2010;12:629–33. https://doi.org/10.1080/10286020.2010.485933
Eyong KO, Krohn K, Hussain H, Folefoc GN, Nkengfack AE, Schulz B, et al. Newbouldiaquinone and Newbouldiamide: a new naphthoquinone-anthraquinone coupled pigment and a new ceramide from Newbouldia laevis. Chem Pharm Bull. 2005;53:616–9. https://doi.org/10.1248/cpb.53.616
Kazakova OB, Rubanik LV, Smirnova IE, Savinova OV, Petrova AV, Poleschuk NN, et al. Synthesis and in vitro activity of oleanane type derivatives against Chlamydia trachomatis. Org Commun. 2019;12:169–75. https://doi.org/10.25135/acg.oc.66.19.07.1352
Wu P, Tu B, Liang J, Guo S, Cao N, Chen S, et al. Synthesis and biological evaluation of pentacyclic triterpenoid derivatives as potential novel antibacterial agents. Bioorg Chem. 2021;109:104692. https://doi.org/10.1016/j.bioorg.2021.104692
Bildziukevich U, Vida N, Rárová L, Kolář M, Šaman D, Havlíček L, et al. Polyamine derivatives of betulinic acid and β-sitosterol: a comparative investigation. Steroids. 2015;100:27–35. https://doi.org/10.1016/j.steroids.2015.04.005
Bildziukevich U, Malik M, Özdemir Z, Rárová L, Janovska L, Slouf M et al. Spermine amides of selected triterpenoid acids: dynamic supramolecular systems formation influences cytotoxicity of the drugs. J Mat Chem B. 2019. https://doi.org/10.1039/c9tb01957j.
Blanchet M, Borselli D, Brunel JM. Polyamine derivatives: a revival of an old neglected scaffold to fight resistant Gram-negative bacteria? Future Med Chem. 2016;8:963–73. https://doi.org/10.4155/fmc-2016-0011
Spivak AY, Khalitova RR, Nedopekina DA, Gubaidullin RR. Antimicrobial properties of amine- and guanidine-functionalized derivativesof betulinic, ursolic and oleanolic acids: Synthesis and structure/activity evaluation. Steroids 2020. 2020;154:108530. https://doi.org/10.1016/j.steroids.2019.108530
Kazakova OB, Giniyatullina GV, Mustafin AG, Babkov DA, Sokolova EV, Spasov AA. Evaluation of cytotoxicity and α-glucosidase inhibitory activity of amide and polyamino-derivatives of lupane triterpenoids. Molecules. 2020;25:4833. https://doi.org/10.3390/molecules25204833
la Hoz ADE, Cortés JA. Bacterial and atypical infections in critically ill cancer patients. Oncologic Crit Care. 2019;379–1400. https://doi.org/10.1007/978-3-319-74588-6_123.
Boyd MR, Paul KD. Some practical considerations and applications of the National Cancer Institute in vitro anticancer drug discovery screen. Drug Res Rep. 1995;34:91–109.
Grever MR, Schepartz SA, Chabner BA. The National Cancer Institute: cancer drug discovery and development program. Semin Oncol. 1992;19:622–38.
Monks A, Scudiero D, Skehan P, Shoemaker R, Paull KD, Vistica D. Feasibility of a highflux anticancer drug screen using a diverse panel of cultured human tumor cell lines. Nat Cancer Inst. 1991;183:757–66.
Monks A, Scudiero DA, Johnson GS, Paull KD, Sausville EA. The NCI anti-cancer drug screen: a smart screen to identify effectors of novel targets. Anti-Cancer Drug Des. 1997;12:533–41.
Weinstein JN, Myers TG, O’Connor PM, Friend SH Jr, Fornace AJ, Kohn KW. An information-intensive approach to the molecular pharmacology of cancer. Science. 1997;275:343–9. https://doi.org/10.1126/science.275.5298.343
Sahu NP, Mahato SB, Banerji N, Chakravarti RN. Cadambagenic acid. New triterpenic acid from Anthocephalus cadamba. Ind J Chem. 1974;12:284–6.
Jurstrand M, Falk L, Fredlund H, Lindberg M, Olcen P, Andersson S, et al. Characterization of Chlamydia trachomatis omp1 genotypes among sexually transmitted disease patients in Sweden. J Сlin Microbiol. 2001;39:3915–9. https://doi.org/10.1128/JCM.39.11.3915-3919.2001
Bednarczyk-Cwynar B, Ruszkowski P, Bobkiewicz-Kozlowska T, Zaprutko L. Oleanolic acid A-lactams inhibit the growth of HeLa, KB, MCF-7 and Hep-G2 cancer cell lines at micromolar concentrations. Anti-Cancer Agents Med Chem. 2016;16:579–92. https://doi.org/10.2174/1871520615666150907095756
Nie W, Luo JG, Wang XB, Yin H, Sun HB, Yao HQ, et al. Synthesis of new α-glucosidase inhibitors based on oleanolic acid incorporating cinnamic amides. Chem Pharm Bull. 2011;59:1051–6. https://doi.org/10.1248/cpb.59.1051
Petrova AV, Lopatina TV, Mustafin AG, Kazakova OB. Modification of azepanobetulin at the isopropenyl group. Russ J Org Chem. 2020;56:1582–7. https://doi.org/10.1134/S1070428020090134
Zaprutko L. Triterpenoids. Part 9. Structure elucidation of a new sodium dichromate oxidation product of methyl oleanolate and some of its derivatives. Pol J Chem. 1995;69:1003–12.
Kazakova OB, Medvedeva NI, Samoilova IA, Baikova IP, Tolstikov GA, Kataev VE, et al. Conjugates of several lupane, oleanane, and ursane triterpenoids with the antituberculosis drug isoniazid and pyridinecarboxaldehydes. Chem Nat Comp. 2011;47:752–8.
Khusnutdinova EF, Petrova AV, Apryshko GN, Kukovinets OS, Kazakova OB. Synthesis and cytotoxicity of indole derivatives of betulin, erythrodiol, and uvaol. Russ J Bioorg Chem. 2018a;44:322–9. https://doi.org/10.1134/S1068162018030081
Khusnutdinova EF, Petrova AV, Kukovinets OS, Kazakova OB. Synthesis and cytotoxicity of 28-N-propargylaminoalkylated 2,3-indolotriterpenic acids. Nat Prod Comm. 2018b;13:665–8. https://doi.org/10.1177/1934578X1801300603
Kazakova O, Smirnova I, Lopatina T, Giniyatullina G, Petrova A, Khusnutdinova E, et al. Synthesis and cholinesterase inhibiting potential of A-ring azepano- and 3-amino-3,4-seco-triterpenoids. Bioorg Chem. 2020;101:104001. https://doi.org/10.1016/j.bioorg.2020.104001
Shen Y, Shi D, Tang J, Sui M. Oleanolic acid derivatives, preparation method and application; CN. 2012-10045425.
Acknowledgements
The reported study was funded by RFBR and BRFBR (project no. 20-53-00014) and the Belarusian Republican Foundation for Fundamental Research (project no. M20P-275). We thank National Cancer Institute for the screening of cytotoxicity for compounds 2, 3, and 23. The antimicrobial screening was performed by CO-ADD (The Community for Antimicrobial Drug Discovery), funded by the Wellcome Trust (UK) and The University of Queensland (Australia).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
About this article
Cite this article
Kazakova, O., Rubanik, L., Smirnova, I. et al. Synthesis and in vitro activity of oleanolic acid derivatives against Chlamydia trachomatis and Staphylococcus aureus. Med Chem Res 30, 1408–1418 (2021). https://doi.org/10.1007/s00044-021-02741-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-021-02741-6




