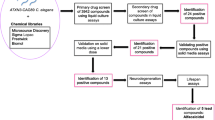
Abstract
Parkinson’s disease (PD) is an age-associated neurodegenerative movement disorder that leads to loss of dopaminergic neurons and motor deficits. Approaches to neuroprotection and symptom management in PD include use of monoamine oxidase B (MAO-B) inhibitors. Many patients with PD also exhibit memory loss in the later stages of disease progression, which is treated with acetylcholine esterase (AChE) inhibitors. We sought to identify a dual-mechanism compound that would inhibit both MAO-B and AChE enzymes. Our screen identified a promising compound (7) with balanced MAO-B (IC50 of 16.83 µM) and AChE inhibition activity (AChE IC50 of 22.04 µM). Application of this compound 7 increased short-term associative memory and significantly prevented 6-hydroxy-dopamine toxicity in dopaminergic neurons in the Caenorhabditis elegans nematode. These findings present a platform for future development of dual-mechanism drugs to treat neurodegenerative diseases such as PD.









Similar content being viewed by others
References
Galindo MF, Solesio ME, Atienzar-Aroca S, Zamora MJ, Jordan Bueso J. Mitochondrial dynamics and mitophagy in the 6-hydroxydopamine preclinical model of Parkinson’s disease. Parkinson’s Dis. 2012;2012:131058. https://doi.org/10.1155/2012/131058.
Bernheimer H, Birkmayer W, Hornykiewicz O, Jellinger K, Seitelberger F. Brain dopamine and the syndromes of Parkinson and Huntington. Clinical, morphological and neurochemical correlations. J Neurol Sci. 1973;20:415–55.
Dardiotis E, Xiromerisiou G, Hadjichristodoulou C, Tsatsakis AM, Wilks MF, Hadjigeorgiou GM. The interplay between environmental and genetic factors in Parkinson’s disease susceptibility: the evidence for pesticides. Toxicology. 2013;307:17–23. https://doi.org/10.1016/j.tox.2012.12.016.
Singleton AB, Farrer MJ, Bonifati V. The genetics of Parkinson’s disease: progress and therapeutic implications. Mov Disord : Off J Mov Disord Soc. 2013;28:14–23. https://doi.org/10.1002/mds.25249.
Surmeier DJ, Guzman JN, Sanchez-Padilla J, Goldberg JA. What causes the death of dopaminergic neurons in Parkinson’s disease?. Prog brain Res. 2010;183:59–77. https://doi.org/10.1016/S0079-6123(10)83004-3.
Tolleson CM, Fang JY. Advances in the mechanisms of Parkinson’s disease. Discov Med. 2013;15:61–6.
Chung SJ, Yoo HS, Lee YH, Sohn YH, Ye BS, Cha J. et al. Minimal parkinsonism in the elderly is associated with striatal dopamine loss and pontine structural damage. Parkinsonism Relat Disord. 2020;81:140–3. https://doi.org/10.1016/j.parkreldis.2020.10.038.
Edmondson DE, Binda C, Mattevi A. Structural insights into the mechanism of amine oxidation by monoamine oxidases A and B. Arch Biochem Biophys. 2007;464:269–76. https://doi.org/10.1016/j.abb.2007.05.006.
Weinreb O, Amit T, Bar-Am O, Youdim MB. Ladostigil: a novel multimodal neuroprotective drug with cholinesterase and brain-selective monoamine oxidase inhibitory activities for Alzheimer’s disease treatment. Curr Drug Targets. 2012;13:483–94.
Weinreb O, Amit T, Bar-Am O, Youdim MB. Rasagiline: a novel anti-Parkinsonian monoamine oxidase-B inhibitor with neuroprotective activity. Prog Neurobiol. 2010;92:330–44. https://doi.org/10.1016/j.pneurobio.2010.06.008.
Youdim MB, Bakhle YS. Monoamine oxidase: isoforms and inhibitors in Parkinson’s disease and depressive illness. Br J Pharmacol. 2006;147 Suppl 1:S287–96. https://doi.org/10.1038/sj.bjp.0706464.
Gerlach M, Riederer P, Przuntek H, Youdim MB. MPTP mechanisms of neurotoxicity and their implications for Parkinson’s disease. Eur J Pharmacol. 1991;208:273–86.
Saura J, Andres N, Andrade C, Ojuel J, Eriksson K, Mahy N. Biphasic and region-specific MAO-B response to aging in normal human brain. Neurobiol Aging. 1997;18:497–507.
Mahy N, Andres N, Andrade C, Saura J. Age-related changes of MAO-A and -B distribution in human and mouse brain. Neurobiology. 2000;8:47–54.
Geldenhuys WJ, Youdim MB, Carroll RT, Van der Schyf CJ. The emergence of designed multiple ligands for neurodegenerative disorders. Prog Neurobiol. 2011;94:347–59. https://doi.org/10.1016/j.pneurobio.2011.04.010.
Allahtavakoli M, Shabanzadeh AP, Sadr SS, Parviz M, Djahanguiri B. Rosiglitazone, a peroxisome proliferator-activated receptor-gamma ligand, reduces infarction volume and neurological deficits in an embolic model of stroke. Clin Exp Pharmacol Physiol. 2006;33:1052–8. https://doi.org/10.1111/j.1440-1681.2006.04486.x.
Weinstock M, Bejar C, Wang RH, Poltyrev T, Gross A, Finberg JP. et al. TV3326, a novel neuroprotective drug with cholinesterase and monoamine oxidase inhibitory activities for the treatment of Alzheimer’s disease. J Neural Transm Suppl. 2000;60:157–69. https://doi.org/10.1007/978-3-7091-6301-6_10.
Zhang Q, Aldridge GM, Narayanan NS, Anderson SW, Uc EY. Approach to cognitive impairment in Parkinson’s disease. Neurotherapeutics. 2020. https://doi.org/10.1007/s13311-020-00963-x.
Savelieff MG, Nam G, Kang J, Lee HJ, Lee M, Lim MH. Development of multifunctional molecules as potential therapeutic candidates for Alzheimer’s disease, Parkinson’s disease, and amyotrophic lateral sclerosis in the last decade. Chem Rev. 2019;119:1221–322. https://doi.org/10.1021/acs.chemrev.8b00138.
Geldenhuys WJ, Van der Schyf CJ. Rationally designed multi-targeted agents against neurodegenerative diseases. Curr Med Chem. 2013;20:1662–72. https://doi.org/10.2174/09298673113209990112.
Van der Schyf CJ, Geldenhuys WJ. Multimodal drugs and their future for Alzheimer’s and Parkinson’s disease. Int Rev Neurobiol. 2011;100:107–25. https://doi.org/10.1016/B978-0-12-386467-3.00006-6.
Santin Y, Resta J, Parini A, Mialet-Perez J. Monoamine oxidases in age-associated diseases: new perspectives for old enzymes. Ageing Res Rev. 2021;66:101256. https://doi.org/10.1016/j.arr.2021.101256.
Jellinger KA. Neurobiology of cognitive impairment in Parkinson’s disease. Expert Rev Neurother. 2012;12:1451–66. https://doi.org/10.1586/ern.12.131
Kerns EH, Di L. Pharmaceutical profiling in drug discovery. Drug Discov Today. 2003;8:316–23.
Ploch-Jankowska A, Pentak D. A comprehensive spectroscopic analysis of the ibuprofen binding with human serum albumin, part I. Pharmaceuticals. 2020;13. https://doi.org/10.3390/ph13090205.
Czub MP, Handing KB, Venkataramany BS, Cooper DR, Shabalin IG, Minor W. Albumin-based transport of nonsteroidal anti-inflammatory drugs in mammalian blood plasma. J Med Chem. 2020;63:6847–62. https://doi.org/10.1021/acs.jmedchem.0c00225.
Wishart DS. Improving early drug discovery through ADME modelling: an overview. Drugs R D. 2007;8:349–62. https://doi.org/10.2165/00126839-200708060-00003.
Martinez BA, Caldwell KA, Caldwell GA. C. elegans as a model system to accelerate discovery for Parkinson disease. Curr Opin Genet Dev. 2017;44:102–9. https://doi.org/10.1016/j.gde.2017.02.011.
Geldenhuys WJ, Kochi A, Lin L, Sutariya V, Dluzen DE, Van der Schyf CJ. et al. Methyl yellow: a potential drug scaffold for Parkinson’s disease. Chembiochem. 2014;15:1591–8. https://doi.org/10.1002/cbic.201300770.
Binda C, Aldeco M, Geldenhuys WJ, Tortorici M, Mattevi A, Edmondson DE. Molecular insights into human monoamine oxidase B inhibition by the glitazone anti-diabetes drugs. ACS Med Chem Lett. 2011;3:39–42. https://doi.org/10.1021/ml200196p.
Binda C, Aldeco M, Mattevi A, Edmondson DE. Interactions of monoamine oxidases with the antiepileptic drug zonisamide: specificity of inhibition and structure of the human monoamine oxidase B complex. J Med Chem. 2011;54:909–12. https://doi.org/10.1021/jm101359c.
Joubert J, Foka GB, Repsold BP, Oliver DW, Kapp E, Malan SF. Synthesis and evaluation of 7-substituted coumarin derivatives as multimodal monoamine oxidase-B and cholinesterase inhibitors for the treatment of Alzheimer’s disease. Eur J Med Chem. 2017;125:853–64. https://doi.org/10.1016/j.ejmech.2016.09.041.
Daina A, Michielin O, Zoete V. SwissADME: a free web tool to evaluate pharmacokinetics, drug-likeness and medicinal chemistry friendliness of small molecules. Sci Rep. 2017;7:42717. https://doi.org/10.1038/srep42717.
Acharya R, Chacko S, Bose P, Lapenna A, Pattanayak SP. Structure based multitargeted molecular docking analysis of selected furanocoumarins against breast cancer. Sci Rep. 2019;9:15743. https://doi.org/10.1038/s41598-019-52162-0.
Greeff J, Joubert J, Malan SF, van Dyk S. Antioxidant properties of 4-quinolones and structurally related flavones. Bioorg Med Chem. 2012;20:809–18. https://doi.org/10.1016/j.bmc.2011.11.068.
Al-Baghdadi OB, Prater NI, Van der Schyf CJ, Geldenhuys WJ. Inhibition of monoamine oxidase by derivatives of piperine, an alkaloid from the pepper plant Piper nigrum, for possible use in Parkinson’s disease. Bioorg Med Chem Lett. 2012;22:7183–8. https://doi.org/10.1016/j.bmcl.2012.09.056.
Kauffman A, Parsons L, Stein G, Wills A, Kaletsky R, Murphy C. C. elegans positive butanone learning, short-term, and long-term associative memory assays. J Vis Exp : JoVE. 2011. https://doi.org/10.3791/2490.
Acknowledgements
This study was funded in part by the Richard Nicely and Glenn and Karen Leppo Parkinson’s Disease Research Funds, the Stark Community Foundation Canton, Ohio, USA, and a grant from the Michael J. Fox Foundation. Additional funding for this work was provided in part by NIH/NIGMS Award Number U54GM104942, WV INBRE P20GM103434 and the WVU Stroke CoBRE NIH grant P20GM109098.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Boos, J.R., Shubbar, A. & Geldenhuys, W.J. Dual monoamine oxidase B and acetylcholine esterase inhibitors for treating movement and cognition deficits in a C. elegans model of Parkinson’s disease. Med Chem Res 30, 1166–1174 (2021). https://doi.org/10.1007/s00044-021-02720-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-021-02720-x