Abstract
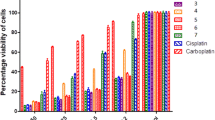
Three new platinum complexes with chemical formulas cis-[PtL2Cl2], cis-[PtL2I2] and cis-[PtL(NH3)Cl2], where L is 3’-methyl-4-thio-1H-tetrahydropyranspiro-5’-hydantoin were synthesized and studied. The spectral and theoretical investigations proved that ligand coordinates through sulfur atom from tetrahydrothiopyrane fragment by monodentate manner with platinum ion. The newly synthesized compounds were tested for cytotoxic activity in vitro against four human tumor cell lines using MTT dye-reduction assay for cell viability. The tested compounds displayed cytotoxic effects in a concentration-dependent manner. Complex 2 as well as complex 3 have similar cytotoxicity to that of the prototype drug cisplatin on colon adenocarcinoma model HT-29.



Similar content being viewed by others
References
Johnstone T, Suntharalingam K, Lippard ST. The next generation of platinum drugs: targeted Pt(II) agents, nanoparticle delivery, and Pt(IV) prodrugs. Chem Rev. 2016;116:3436–86.
Freire R, Marques M, Fagundes E, Oliveira R, Alves R. Synthesis and evaluation of platinum complexes with potential antitumor activity. Braz J Pharm Sci. 2017;53:e15235.
Ahmad S, Isab A, Ali S. Structural and mechanistic aspects of platinum anticancer agents. Transit Met Chem. 2006;31:1003–16.
Graf N, Lippard J. Redox activation of metal-based prodrugs as a strategy for drug delivery. Adv Drug Deliv Rev. 2012;64:993–1004.
Cho S, Kimb S, Shin D. Recent applications of hydantoin and thiohydantoin in medicinal chemistry. Eur J Med Chem. 2019;164:517–45.
Jung M, Ouk S, Yoo D, Sawyers C, Chen C, Tran C, et al. Structure-activity relationship for thiohydantoin androgen receptor antagonists for castration-resistant prostate cancer (CRPC). J Med Chem. 2010;53:2779–96.
Kavitha C, Nambiar M, Kumar C, Choudhary B, Muniyappa K, Rangappa K, et al. Novel derivatives of spirohydantoin induce growth inhibition followed by apoptosis in leukemia cells. Biochem Pharm. 2009;77:348–63.
Malancona S, Altamura S, Filocamo G, Kinzel O, Hernando J, Rowley M, et al. Identification of MK-5710 ((8aS)-8amethyl- 1,3-dioxo-2-[(1S,2R)-2-phenylcyclopropyl]-N-(1-phenyl-1H-pyrazol-yl)hexahydroimidazo[1,5a]pyrazine-7(1H)-carboxamide), a potent smoothened antagonist for use in Hedgehog pathway dependent malignancies. Part 1. Bioorg Med Chem Lett. 2011;21:4422–8.
Basappa C, Kumar A, Swamy S, Sugahara K, Rangappa K. Antitumor and anti-angiogenic activity of novel hydantoin derivatives: inhibition of VEGF secretion in liver metastatic osteosarcoma cells. Bioorg Med Chem. 2009;17:4928–34.
Carmi C, Cavazzoni A, Zuliani V, Lodola A, Bordi F, Plazzi P, et al. 5-Benzylidene-hydantoins as new EGFR inhibitors with antiproliferative activity. Bioorg Med Chem Lett. 2006;16:4021–5.
Sergent D, Wang Q, Sasaki N, Ouazzani J. Synthesis of hydantoin analogues of (2S,3R,4S)-4-hydroxyisoleucine with insulinotropic properties. Bioorg Med Chem Lett. 2008;18:4332–5.
Fujisaki F, Shoji K, Shimodouzono M, Kashige N, Miake F, Sumoto K. Antibacterial activity of 5-dialkylaminomethylhydantoins and related compounds. Chem Pharm Bull. 2010;58:1123–6.
Zhu Q, Pan Y, Xu Z, Li R, Qiu G, Xu W, et al. Synthesis and potential anticonvulsant activity of new N-3-substituted 5,5-cyclopropanespirohydantoins. Eur J Med Chem. 2009;44:296–302.
Byrtus H, Obniska J, Czopek A, Kaminski K. Synthesis and anticonvulsant activity of new N-Mannich bases derived from 5-Cyclopropyl-5-phenylhydantoins. Arch Pharm. 2011;344:231–41.
Kim D, Wang L, Caldwell C, Chen P, Finke P, Oates B, et al. Discovery of human CCR5 antagonists containing hydantoins for the treatment of HIV-1 infection. Bioorg Med Chem Lett. 2001;11:3099–102.
Kim D, Wang L, Caldwell C, Chen P, Finke P, Oates B, et al. Design, synthesis, and SAR of heterocycle-containing antagonists of the human CCR5 receptor for the treatment of HIV-1 infection. Bioorg Med Chem Lett. 2001;11:3103–6.
Romine J, Laurent D, Leet J, Martin W, Serrano-Wu M, Yang F, et al. Inhibitors of HCV NS5A: from iminothiazolidinones to symmetrical stilbenes. ACS Med Chem Lett. 2011;2:224–9.
Bakalova A, Buyukliev R, Momekov G. Synthesis, DFT calculations and cytotoxic investigation of platinum complexes with 3-thiolanespiro-5’-hydantoin and 4-thio-1H-tetrahydropyranspiro-5’-hydantoin. J Mol Struct. 2015;1091:118–24.
Cherneva E, Atanasova M, Buyukliev R, Tomovic K, Smelcerovic Z, Bakalova A et al. 3’-Methyl-4-thio-1H-tetrahydropyranspiro-5’-hydantoin platinum complex as novel potent anticancer agent and xanthine oxidase inhibitor, Archiv der Pharmazie. 2020. https://doi.org/10.1002/ardp.202000039 (in press).
Bakalova A, Buyukliev R, Varbanov H. Design, synthesis and comparative cytotoxic investigation of platinum(II) complexes with some derivatives of 5-methyl-5-(4-pyridyl)hydantoin. Inorg Chim Acta. 2014;423:46–51.
Cherneva E, Buyukliev R, Bakalova A. Pt(II) complex with 3’-amino-2-indanespiro-5’-hydantoin – synthesis, spectral investigation and quantum chemical study. Compt rend Acad bulg Scien. 2017;70:941–6.
Cherneva E, Buyukliev R, Bakalova A. Palladium complexes with 3-methyl-8-thia-1,3-diaza-spiro[4.5]decane-2,4-dione: Experimental and theoretical studies. Arkivoc. 2018;part vii:398–400.
Štarha P, Vančo J, Trávníček Z. Platinum iodido complexes: a comprehensive overview of anticancer activity and mechanisms of action. Coord Chem Revs. 2019;380:103–35.
Frisch M, Trucks G, Schlegel H, Scuseria G, Robb M, Cheeseman J, et al. Gaussian 03, Revision C.02. Wallingford CT: Gaussian, Inc.; 2004.
Dhara S. Rapid colorimetric assay for cellular growth and survival: Application to proliferation and cytotoxicity assays. Ind J Chem. 1970;8:193.
Stephens P, Devlin F, Chabalowski C, Frisch M. Ab initio calculation of vibrational absorption and circular dichroism spectra using density functional force fields. J Phys Chem. 1994;98:11623–7.
Lee C, Yang W, Parr R. Development of the Cole–Salvetti correlation-energy formula into a functional of the electron density. Phys Rev B. 1988;37:785–9.
Mosmann T. Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods. 1983;65:55–63.
Konstantinov S, Eibl H, Berger M. BCR-ABL influences the antileukaemic efficacy of alkylphos-phocholines. Br J Haematol. 1999;107:365–74.
Acknowledgements
The financial support of this work by the Medical Science Council at the Medical University – Sofia within the Grant no. 84/23.04.2019 is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Bakalova, A.G., Buyukliev, R.T. & Cherneva, E.D. New Pt(II) complexes with 3’-methyl tetrahydro-4H-thiopyranspiro-5’-hydantoin: synthesis, theoretical and cytotoxic investigation. Med Chem Res 29, 2218–2223 (2020). https://doi.org/10.1007/s00044-020-02639-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02639-9