Abstract
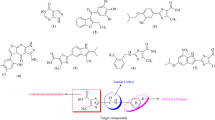
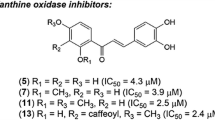
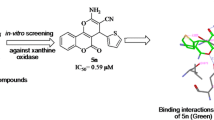
A library of indolinedione–coumarin hybrid molecules was rationally designed and synthesized against hyperuricemia. All of the synthesized hybrid molecules were tested to check their inhibitory activity against xanthine oxidase enzyme by using a spectrophotometric assay. The results revealed that the compound showed IC50 values within the range of 6.5–24.5 µM amongst which compound K-7 was found to be endowed with the most potent IC50 value against xanthine oxidase enzyme. Kinetic studies were also performed to check the mode of inhibition of most potent compound K-7, which revealed its mixed-type inhibition behavior. Structure-activity relationships revealed that electron-donating groups and small alkyl chains between the two active scaffolds might be beneficial in inhibiting xanthine oxidase enzyme. It was also shown that various electrostatic interactions stabilized the compound K-7 within the active site of xanthine oxidase enzyme, which confirmed that it can completely block its catalytic active site. Thus, K-7 is regarded as a potent xanthine oxidase inhibitor and can be served as a promising molecular architectural unit for anti-hyperuricemic drug design.





Similar content being viewed by others
References
Ali HI, Fujita T, Akaho E, Nagamatsu T (2010) A comparative study of AutoDock and PMF scoring performances, and SAR of 2-substituted pyrazolotriazolopyrimidines and 4-substituted pyrazolopyrimidines as potent xanthine oxidase inhibitors. J Comput Aided Mol Des 24:57–75
Becker MA, Kisicki J, Khosravan R, Wu J, Mulford D, Hunt B, MacDonald P, Joseph-Ridge N (2004) Febuxostat (TMX‐67), a novel, non‐purine, selective inhibitor of xanthine oxidase, is safe and decreases serum urate in healthy volunteers. Nucl Nucl Nucl 23:35–40
Becker M, Schumacher H, MacDonald PA (2007) Febuxostat: a guide to its use in chronic hyperuricaemia. Abstr no 757 Arthritis Rheum 56(9 Suppl):S322
Biagi G, Giorgi I, Pacchini F, Livi O, Scartoni V (2001) 2-Alkyloxyalkylthiohypoxanthines as new potent inhibitors of xanthine oxidase. Farmaco 56:809–813
Borges F, Fernandes E, Roleira F (2002) Progress towards the discovery of xanthine oxidase inhibitors. Curr Med Chem 9:195–217
Brien DE, Springer RH, Albert TNA, Senga K, Miller JP, Streeter DG (1985) Purine analog inhibitors of xanthine oxidase ‐ structure activity relationships and proposed binding of the molybdenum cofactor. J Heterocycl Chem 22:601–634
ChemDraw Ultra 6.0 and Chem3D Ultra (2013); Cambridge Soft Corporation: Cambridge, USA
Chen C, Huang C, Tsai K, Huang W, Huang W, Hsu Y, Hsu F (2014) Evaluation of the antihyperuricemic activity of phytochemicals from davallia formosana by enzyme assay and hyperuricemic mice model. Evid Based Complement Alternat Med 2014:1–8
Copeland RA (2005) Evaluation of Enzyme Inhibitors in Drug Discovery. Wiley, Hoboken
Dassault Systemes BIOVIA (2016) Discovery studio modeling environment, release 2017. Dassault Systemes, San Diego
Dhiman R, Sharma S, Singh G, Nepali K, Bedi PMS (2012) Design and synthesis of aza‐flavones as a new class of xanthine oxidase inhibitors. Arch Pharm Chem Life Sci 346:7–16
Escribano J, Gracia-Canovas F, Garcia-Carmona F (1988) A kinetic study of hypoxanthine oxidation by milk xanthine oxidase. Bio Chem J 254:829–833
Fais A, Era B, Asthana S, Sogos V, Medda R, Santana L, Uriarte E, Mtos MJ, Delogu F, Kumar A (2018) Coumarin derivatives as promising xanthine oxidase inhibitors. Ijbiomac 120:1286–1293
Hille R (2006) Structure and function of xanthine oxidoreductase. Eur J Inorg Chem 10:1913–1926
Ishibuchi S, Morimoto H, Oe T, Ikebe T, Inoue H, Fukunari A, Kamezawa M, Yamada I, Naka Y (2001) Synthesis and structure–activity relationships of 1-Phenylpyrazoles as xanthine oxidase inhibitors. Bioorg Med Chem Lett 11:879–882
Kaur C, Dhiman S, Singh H, Kaur M, Bhagat S, Gupta M, Sharma S, Bedi PMS (2015) Synthesis, screening and docking studies of benzochromone derivatives as xanthine oxidase inhibitors. J Chem Phar Res 7:127–136
Kaur G, Singh JV, Gupta MK, Bhagat K, Gulati HK, Singh A, Bedi PMS, Singh H, Sharma S (2019) Thiazole-5-carboxylic acid derivatives as potent xanthine oxidase inhibitors: design, synthesis, in vitro evaluation, and molecular modeling studies. Medicinal Chem Res 29:83–93
Kaur M, Kaur A, Mankotia S, Singh H, Singh A, Singh JV, Gupta MK, Sharma S, Nepali K, Bedi PMS (2017) Synthesis, screening and docking of fused pyrano[3,2-d]pyrimidine derivatives as xanthine oxidase inhibitor. Eur J Med Chem 131:14–28
Kaur R, Naaz F, Sharma S, Mehndiratta S, Gupta MK, Bedi PMS, Nepali K (2015) Screening of a library of 4-aryl/heteroaryl-4H-fused pyrans for xanthine oxidase inhibition: synthesis, biological evaluation and docking studies. Med Chem Res 24:3334–3349
Komoriya K, Osada Y, Hasegawa M, Horiuchi H, Kondo S, Couch RC, Griffin TB (1993) Hypouricemic effect of allopurinol and the novel xanthine oxidase inhibitor TEI-6720 in chimpanzees. Eur J Pharm 250:455–460
Kumar R, Darpan, Sharma S, Singh R(2011) Xanthine oxidase inhibitors: a patent survey Expert Opin Ther Pat 21:1071–1108
Love BL, Barrons R, Veverka A, Snider KM (2010) Urate-lowering therapy for gout: focus on febuxostat. Pharmacother 30:594–608
Malik N, Dhiman P, Sobarzo-Sanchez E, Khatkar A (2018) Flavonoids and anthranquinones as xanthine oxidase and monoamine oxidase inhibitors: a new approach towards inflammation and oxidative stress. Curr Top Med Chem 18:2154–2164
Mehmood A, Ishaq M, Zhao L, Safdar B, Rehman AU, Munir M, Raza A, Nadeem M, Iqbal W, Wang C (2019) Natural compounds with xanthine oxidase inhibitory activity: a review. Chem Biol Drug Des 93:387–418
Nagamatsu T, Fujita T, Endo K (2000) Novel xanthine oxidase inhibitor studies. Part 2. Synthesis and xanthine oxidase inhibitory activities of 2-substituted 6-alkylidenehydrazino- or 6-arylmethylidenehydrazino-7H-purines and 3- and/or 5-substituted 9H-1,2,4-triazolo[3,4-i]purines. Perkin Trans 1:33–38
Nagamatsu T, Ukai M,F, Yoneda F, Brown DJ (1985) Syntheses of 4 Methyl-s-triazolo[4,3-a]purin-9(4H)-ones and tetrazolo-[1,5-a]purin-9(4H)-ones as Aza Analogs of “Y” Bases. Chem Pharm Bull 33:3113–3121
Nagamatsu T, Yamasaki H (1995) Facile, general and productive syntheses of the fluorescent wye (4,9-dihydro-4,6-dimethyl-9-oxo-1H-imidazo[1,2-a]purine) in phenylalanine tRNA, its 2-substituted derivatives and 7-aza analogues. J Chem Soc Chem Commun 19:2041–2043
Oettl K, Reibneggar G (1999) Pteridines as inhibitors of xanthine oxidase: structural requirements. Biochim Biophys Acta 1430:387–395
Ojha R, Singh J, Ojha A, Singh H, Sharma S, Nepali K (2017) An updated patent review: xanthine oxidase inhibitors for the treatment of hyperuricemia and gout (2011-2015). Expert Opin Ther Pat 27:311–345
Osada Y, Tsuchimoto M, Fukushima H, Takahashi K, Kondo S, Hasegawa M, Komoriya K (1993) Hypouricemic effect of the novel xanthine oxidase inhibitor, TEI-6720, in rodents. Eur J Pharm 241:183–188
Pacher P, Nivorozhkin A, Szabo C (2006) Therapeutic effects of xanthine oxidase inhibitors: renaissance half a century after the discovery of allopurinol. Pharm Rev 58:87–114
Pascart T, Richette P (2018) Investigational drugs for hyperuricemia, an update on recent developments. Expert Opin Inv Drug 27:437–444
Schumacher HRJ, Becker MA, Wortmann RL, Macdonald PA, Hunt B, Streit J, Lademacher C, Joseph-Ridge N (2008) Effects of febuxostat versus allopurinol and placebo in reducing serum urate in subjects with hyperuricemia and gout: a 28-week, phase III, randomized, double-blind, parallel-group trial. Arthritis Rheum 59:1540–1548
Sebastian E, Sattui, Gaffo AL (2016) Treatment of hyperuricemia in gout: current therapeutic options, latest developments and clinical implications. Ther Adv Musculoskel Dis 8:145–159
Singh JV, Mal G, Kaur G, Gupta MK, Singh A, Nepali K, Singh H, Sharma S, Bedi PMS (2019) Benzoflavone derivatives as potent antihyperuricemic agents. Med Chem Commun 10:128–147
Sharma S, Sharma K, Ojha R, Kumar D, Singh G, Nepali K, Bedi PMS (2014) Microwave assisted synthesis of naphthopyrans catalysed by silica supported fluoroboric acid as a new class of non purine xanthine oxidase inhibitors. Bioorg Med Chem Lett 24:495–500
Shukla S, Kumar D, Ojha R, Gupta MK, Nepali K, Bedi PMS (2014) 4,6‐Diaryl/heteroarylpyrimidin‐2(1H)‐ones as a new class of xanthine oxidase inhibitors. Arch Pharm Chem Life Sci 347:1–10
Strilchuk L, Fogacci F, Cicero AF (2019) Safety and tolerability of available urate-lowering drugs: a critical review. Expert Opin Drug Saf 18:261–271
Takano Y, Hase-Aoki K, Horiuchi H, Zhao L, Kasahara Y, Kondo S, Becker MA (2005) Selectivity of febuxostat, a novel non-purine inhibitor of xanthine oxidase/xanthine dehydrogenase. Life Sci 76:1835–1847
Virdi HS, Sharma S, Mehndiratta S, Bedi PMS, Nepali K (2014) Design, synthesis and evaluation of 2,4-diarylpyrano[3,2-c]chromen-5(4H)-one as a new class of non-purine xanthine oxidase inhibitors. J Enzym Inhib Med Chem 30:1–7
Wortmann RL (1998) Gout and other disorders of purine metabolism. In: Brunwald E ed. Harrison’s principles of internal medicine. McGraw-Hill, New York (NY), Vol. 14, p 2158–2166
Acknowledgements
Authors are grateful to the University Grants Commission for providing funds under Rajiv Gandhi National Fellowship (RGNF) and National Fellowships for Other Backword Classes (NFOBC), Department of Science & Technology (DST-PURSE), Council of Scientific and Industrial Research (CSIR): Project no. 02(0319)17/EMR-II, Women Scientists Scheme-A (WOS-A: DST) and DST-FIST. The authors are also thankful to Guru Nanak Dev University, Amritsar for providing various basic facilities to carry out the research work.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Gulati, H.K., Bhagat, K., Singh, A. et al. Design, synthesis and biological evaluation of novel indolinedione–coumarin hybrids as xanthine oxidase inhibitors. Med Chem Res 29, 1632–1642 (2020). https://doi.org/10.1007/s00044-020-02589-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02589-2