Abstract
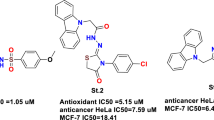
This article reports the diastereoselective synthesis of some novel naphthalimido and bis-naphthalimido β-lactam derivatives and a preliminary evaluation of their anticancer properties. The reactions were completely diastereoselective, leading exclusively to the formation of cis-β-lactams 11a–l and trans-bis-β-lactams 16a–g. All of these compounds were obtained in good to excellent yields and their structures were established based on IR, 1H NMR, 13C NMR spectral data, and elemental analysis. Each of the β-lactams was screened for antioxidant and anticancer activities. Our results showed that all the compounds lacked cytotoxicity against HepG2 cells, whereas 16a and 16b exhibited excellent anticancer activity with IC50 values below 191.57 µM on MCF-7 cell line and also, bis-β-lactams 16a–g showed excellent antitumor activity against the TC-1 cell line. Antioxidant experiments of 16a–d by the diphenylpicrylhydrazyl (DPPH) assay showed IC50 values ranging from 7 to 32.3 µg/ml. Interaction of 16a, 16b, 16d–g with calf-thymus DNA (CT-DNA) was also supported by absorption titration studies. The compounds exhibit good binding propensity to CT-DNA and the DNA binding affinity (Kb) of the compounds varies as 16a; 16b; 16e; 16g > 16d; 16f. Interaction of 16d with CT-DNA was also investigated by fluorescence spectroscopy. The results support an intercalative interaction of 16d and 16f and non-intercalation mechanism for 16a, 16b, 16e, and 16g.














Similar content being viewed by others
Abbreviations
- MCF-7 :
-
Breast cancer cells
- TC-1 :
-
Mouse lung epithelial cells
- HepG2 :
-
Liver hepatocellular carcinoma
- DPPH:
-
Diphenylpicrylhydrazyl
- ORTEP:
-
Oak Ridge Thermal Ellipsoid Plot
- CT-DNA:
-
Calf thymus-deoxyribonucleic acid
- K b :
-
binding affinity
- IC50:
-
Half maximal inhibitory concentration
- NI:
-
Naphthalimide
- NDI:
-
1,4,5,8-Naphthalenetetracarboxylicdiimide
- MTT:
-
Methyl thiazol tetrazolium bromide
- OD:
-
Optical density
References
Abdel-Aziz AA-M, ElTahir KEH, Asiri YA (2011) Synthesis, anti-inflammatory activity and COX-1/COX-2 inhibition of novel substituted cyclic imides. Part 1: molecular docking study. Eur J Med Chem 46:1648–1655
Alami N, Paterson J, Belanger S, Juste S, Grieshaber CK, Leyland-Jones B (2007) Comparative analysis of xanafide cytotoxicity in breast cancer cell lines. Br J Cancer 97:58–64
Alborz M, Jarrahpour AA, Pournejati R, Karbalaei-Heidari HR, Sinou V, Latour C, Brunel JM, Sharghi H, Aberi M, Turos E, Wojtas L (2018) Synthesis and biological evaluation of some novel diastereoselective benzothiazole β-lactam conjugates. Eur J Med Chem 143:283–291
Ameri RJ, Jarrahpour A, Latour CH, Sinou V, Brunel JM, Zgou H, Mabkhot Y, Ben Hadda T, Turos E (2017) Synthesis and antimicrobial/antimalarial activities of novel naphthalimido trans-β-lactam derivatives. Med Chem Res 10:2235–2242
Amr AEGE, Sabry NM, Abdulla MM (2007) Synthesis, reactions, and anti-inflammatory activity of heterocyclic systems fused to a thiophene moiety using citrazinic acid as synthon. Monatshefte für Chem Chem Monthly 138:699–707
Anizon F, Belin L, Moreau P, Sancelme M, Voldoire A, Prudhomme M, Ollier M, Severe D, Riou JF, Bailly C, Fabbro D, Thomas M (1997) Syntheses and biological activities (topoisomerase inhibition and antitumor and antimicrobial properties) of rebeccamycin analogues bearing modified sugar moieties and substituted on the imide nitrogen with a methyl group. J Med Chem 21:3456–3465
Arif R, Rana M, Yasmeen S, Khan MS, Abid M, Khan MS (2020) Facile synthesis of chalcone derivatives as antibacterial agents: synthesis, DNA binding, molecular docking, DFT and antioxidant studies. J Mol Struct 1208:127905
Arunadevi A, Porkodi J, Ramgeetha L, Raman N (2019) Biological evaluation, molecular docking and DNA interaction studies of coordination compounds gleaned from a pyrazolone incorporated ligand. Nucleosides Nucleotides Nucleic Acids 38:656–679
Arya N, Jagdale AY, Patil TA, Yeramwar SS, Holikatti SS, Dwivedi J, Shishoo CJ, Jain KS (2014) The chemistry and biological potential of azetidin-2-ones. Eur J Med Chem 74:619–656
Arya S, Kumar S, Rani R, Kumar N, Roy P, Sondhi S (2013) Synthesis, anti-inflammatory, and cytotoxicity evaluation of 9,10-dihydroanthracene-9,10-α,β-succinimide and bis-succinimide derivatives. Med Chem Res 22:4278–4285
Ayati A, Bakhshaiesh TO, Moghimi S, Esmaeili R, Majidzadeh-A K, Safavi M, Firoozpour L, Emami S, Foroumadi A (2018) Synthesis and biological evaluation of new coumarins bearing 2, 4-diaminothiazole-5-carbonyl moiety. Eur J Med Chem 155:483–491
Banerjee S, Kitchen JA, Gunnlaugsson T, Kelly JM (2013) The effect of the 4-amino functionality on the photophysical and DNA binding properties of alkyl-pyridinium derived 1,8-naphthalimides. Org Biomol Chem 11:5642–5655
Baraldi PG, Preti D, Fruttarolo F, Tabrizi MA, Romagnoli R (2007) Hybrid molecules between distamycin A and active moieties of antitumor agents. Bioorg Med Chem 15:17–35
Baraldi PG, Romagnoli R, Guadix AE, Pineda de las Infantas MJ, Gallo MA, Espinosa A, Martinez A, Bingham JP, Hartley JA (2002) Design, synthesis, and biological activity of hybrid compounds between uramustine and DNA minor groove binder distamycin A. J Med Chem 45:3630–3638
Benzie IFF, Strain JJ (1999) Ferric reducing/antioxidant power assay: direct measure of total antioxidant activity of biological fluids and modified version for simultaneous measurement of total antioxidant power and ascorbic acid concentration. Meth Enzymol 299:15–27
Berlinck RG, Britton R, Piers E, Lim L, Roberge M, Moreira da Rocha R, Andersen RJ (1998) Granulatimide and isogranulatimide, aromatic alkaloids with G2 checkpoint inhibition activity isolated from the Brazilian ascidian Didemnum granulatum: structure elucidation and synthesis. J Org Chem 63:9850–9856
Bhat SS, Kumbhar AA, Heptullah H, Khan AA, Gobre VV, Gejji SP, Puranik VG (2010) Synthesis, electronic structure, DNA and protein binding, DNA cleavage, and anticancer activity of fluorophore-labeled copper (II) complexes. Inorg Chem 50:545–558
Borazjani N, Jarrahpour A, Ameri Rad J, Mohkam M, Behzadi M, Ghasemi Y, Mirzaeinia S, Karbalaei-Heidari HR, Ghanbari MM, Batta G, Turos E (2019a) Design, synthesis and biological evaluation of some novel diastereoselective β-lactams bearing 2-mercaptobenzothiazole and benzoquinoline. Med Chem Res 28:329–339
Borazjani N, Sepehri S, Behzadi M, Jarrahpour A, Ameri Rad J, Sasanipour M, Mohkam M, Ghasemi Y, Akbarizadeh AR, Digiorgio C, Brunel JM, Ghanbari MM, Batta G, Turos E (2019b) Three-component synthesis of chromeno β-lactam, Eur. J Med Chem 179:389–403
Cossio FP, Ugalde JM, Lopez X, Lecea B, Palomo C (1993) A semiempirical theoretical study on the formation of β-lactams from ketenes and imines. J Am Chem Soc 115:995–1004
De Oliveira KN, Chiaradia LD, Martins PGA, Mascarello A, Cordeiro MNS, Guido RVC, Andricopulo AD, Yunes RA, Nunes RJ, Vernal J, Terenzi H (2011) Sulfonyl-hydrazones of cyclic imides derivatives as potent inhibitors of the Mycobacterium tuberculosis protein tyrosine phosphatase B (PtpB). MedChemComm 2:500–504
El-Azab AS, Alanazi AM, Abdel-Aziz NI, Al-Suwaidan IA, El-Sayed MAA, El-Sherbeny MA, Abdel-Aziz AAM (2013) Synthesis, molecular modeling study, preliminary antibacterial, and antitumor evaluation of N-substituted naphthalimides and their structural analogues. Med Chem Res 22:2360–2375
Galletti P, Soldati R, Pori M, Durso M, Tolomelli A, Gentilucci L, Dattoli SD, Baiula M, Spampinato S, Giacomini D (2014) Targeting integrins αvβ3 and α5β1 with new β-lactam derivatives. Eur J Med Chem 83:284–293
Ge C, Chang L, Zhao Y, Chang C, Xu X, He H, Wang Y, Dai F, Xie S, Wang C (2017) Design, synthesis and evaluation of naphthalimide derivatives as potential anticancer agents for hepatocellular carcinoma. Molecules 22:342
Geesala R, Gangasani JK, Budde M, Balasubramanian S, Vaidya JR, Das A (2016) 2-Azetidinones: Synthesis and biological evaluation as potential anti-breast cancer agents. Eur J Med Chem 124:544–558
Gudeika D, Lygaitis R, Gražulevičius JV, Kublickas RH, Rubežienė V, Vedegytė J (2012) Synthesis and properties of dimeric naphthalene diimides. Chemija 23:233–238
Hénon H, Messaoudi S, Anizon F, Aboab B, Kucharczyk N, Léonce S, Golsteyn R, Pfeiffer MB, Prudhomme M (2007) Bis-imide granulatimide analogues as potent checkpoint 1 kinase inhibitors. Eur J Pharm 554:106–112
Iraji A, Firuzi O, Khoshneviszadeh M, Nadri H, Edraki N, Miri R (2018) Synthesis and structure-activity relationship study of multi-target triazine derivatives as innovative candidates for treatment of Alzheimer’s disease. Bioorg Chem 77:223–235
Kamal A, Reddy BSN, Reddy GSK, Ramesh G (2002) Design and synthesis of C-8 linked pyrrolobenzodiazepine–naphthalimide hybrids as anti-tumour agents. Bioorg Med Chem Lett 12:1933–1935
Kamboj VK, Kapoor A, Jain S (2019) Synthesis, antimicrobial, and antioxidant screening of aryl acetic acid incorporated 1,2,4‐triazolo‐1,3,4‐thiadiazole derivatives. J Heterocycl Chem 56:1376–1382
Kianpour S, Ebrahiminezhad A, Mohkam M, Tamaddon AM, Dehshahri A, Heidari R, Ghasemi Y (2017) Physicochemical and biological characteristics of the nanostructured polysaccharide-iron hydrogel produced by microorganism Klebsiella oxytoca. J Basic Microbiol 57:132–140
Kianpour S, Ebrahiminezhad A, Negahdaripour M, Mohkam M, Mohammadi F, Niknezhad SV (2018) Y. Ghasemi, Characterization of biogenic Fe (III)-binding exopolysaccharide nanoparticles produced by Ralstonia sp. SK03. Biotech Prog 34:1167–76
Kostova I, Bhatia S, Grigorov P, Balkansky S, Parmar VS, Prasad AK, Saso L (2011) Coumarins as antioxidants. Curr Med Chem 18:3929–3951
Kumar Gupta R, Pandey R, Sharma G, Prasad R, Koch B, Srikrishna S, Li PZ, Xu Q, Pandey DS (2013) DNA binding and anti-cancer activity of redox-active heteroleptic piano-stool Ru(II), Rh(III), and Ir(III) complexes containing 4‑(2-methoxypyridyl)phenyldipyrromethene. Inorg Chem 52:3687–3698
Kumar A, Banerjee S, Roy P, Sondhi SM, Sharma A (2017) Solvent free, catalyst free, microwave or grinding assisted synthesis of bis-cyclic imide derivatives and their evaluation for anticancer activity. Bioorg Med Chem Lett 27:501–504
Landa A, Mielgo A, Oiarbide M, Palomo C (2018) Asymmetric synthesis of β‐lactams by the Staudinger reaction. Organic reactions. John Wiley & Sons, Hobokenpp, p 1–123
Laronze M, Boisbrun M, Leonce S, Pfeiffer B, Renard P, Lozach O, Meijer L, Lansiaux A, Bailly C, Sapi J, Laronzea JY (2005) Synthesis and anticancer activity of new pyrrolocarbazoles and pyrrolo-β-carbolines. Bioorg Med Chem 13:2263–2283
Li Q, Fang H, Wang X, Xu W (2010) Novel cyclic-imide peptidomimetics as aminopeptidase N inhibitors. Structure-based design, chemistry and activity evaluation. Eur J Med Chem 45:1618–1626
Li Y, Li Y, Liu X, Yang Y, Lin D, Gao Q (2020) The synthesis, characterization, DNA/protein interaction, molecular docking and catecholase activity of two Co (II) complexes constructed from the aroylhydrazone ligand. J Mol Struct 1202:127229
Li S, Xu S, Tang Y, Ding S, Zhang J, Wang S, Zhou G, Zhou C, Li X (2014). Synthesis, anticancer activity and DNA-binding properties of novel 4-pyrazolyl-1,8-naphthalimide derivatives. Bioorg Med Chem Lett 24:586–590
Machado KE, de Oliveira KN, Santos-Bubniak L, Licinio MA, Nunes RJ, Santos-Silva MC (2011) Evaluation of apoptotic effect of cyclic imide derivatives on murine B16F10 melanoma cells. Bioorg Med Chem 19:6285–6291
Malterud KE, Farbrot TL, Huse AE, Sund RB (1993) Antioxidant and radical scavenging effects of anthraquinones and anthrones. Pharmacol 47:77–85
Mandegani Z, Asadi Z, Asadi M, Karbalaei-Heidari HR, Rastegari B (2016) Synthesis, characterization, DNA binding, cleavage activity, cytotoxicity and molecular docking of new nano water-soluble [M(5-CH2PPh3-3,4-salpyr)](ClO4)2 (M = Ni, Zn) complexes. Dalton Trans 15:6592–6611
Marmur J (1961) A procedure for the isolation of deoxyribonucleic acid from micro-organisms. J Mol Biol 3:208-IN1
Milelli A, Tumiatti V, Micco M, Rosini M, Zuccari G, Raffaghello L, Bianchi G, Pistoia V, Díaz JF, Pera B, Trigili C (2012) Structure–activity relationships of novel substituted naphthalene diimides as anticancer agents. Eur J Med Chem 57:417–428
Miller KD, Siegel RL, Lin CC, Mariotto AB, Kramer JL, Rowland JH, Stein KD, Alteri R, Jemal A (2016) Cancer treatment and survivorship statistics. CA Cancer J Clin 66:271–289
Mondal S, Samajdar RN, Mukherjee S, Bhattacharyya AJ, Bagchi B (2018) Unique eatures of metformin: A combined experimental, theoretical, and simulation study of its structure, dynamics, and interaction energetics with DNA grooves. J Phys Chem B 122:227–2242
Mosmann T (1983) Rapid colorimetric assay for cellular growth and survival: application to proliferation and cytotoxicity assays. J Immunol Methods 65:55–63
Palomo C, Aizpurua JM, Ganboa I, Oiarbide M (2004) Asymmetric synthesis of β-lactams through the Staudinger reaction and their use as building blocks of natural and nonnatural products. Curr Med Chem 11:1837–1872
Parul DM, Sengar NPS, Pathak AK (2010) 2-Azetidinone A new profile of pharmacological activities. Eur J Med Chem 45:5541–5560
Riss TL, Moravec RA, Niles AL, Benink HA, Worzella TJ, Minor L (2013) Cell viability assays, assay guidance manual. Eli Lilly & Company and the National Center for Advancing Translational Sciences, Bethesda, MD, USA, p 1–23
Rowan NJ, Deans K, Anderson JG, Gemmell CG, Hunter IS, Chaithong T (2001) Putative virulence factor expression by clinical and food isolates of Bacillus spp. after growth in reconstituted infant milk formulae. Appl Environ Microbiol 67:3873–3881
Said SA, Amr AEGE, Sabry NM, Abdalla MM (2009) Analgesic, anticonvulsant and anti-inflammatory activities of some synthesized benzodiazipine, triazolopyrimidine and bis-imide derivatives. Eur J Med Chem 44:4787–4792
Shaikh SKJ, Kamble RR, Somagond SM, Devarajegowda HC, Dixit SR, Joshi SD (2017) Tetrazolylmethyl quinolines: Design, docking studies, synthesis, anticancer and antifungal analyses. Eur J Med Chem 128:258–273
Suh D, Chaires JB (1995) Criteria for the mode of binding of DNA binding agents. Bioorg Med Cehm 3:723–728
Tomczyk MD, Walczak KZ (2018) l,8-Naphthalimide based DNA intercalators and anticancer agents. A systematic review from 2007 to 2017. Eur J Med Chem 159:393–422
Tumiatti V, Milelli A, Minarini A, Micco M, Gasperi Campani A, Roncuzzi L, Baiocchi D, Marinello J, Capranico G, Zini M, Stefanelli C (2009) Design, synthesis, and biological evaluation of substituted naphthalene imides and diimides as anticancer agent. J Med Chem 52:7873–7877
Wang LJ, Geng CA, MA YB, Luo J, Huang XY, Chen H, Zhou NJ, Zhang XM, Chen JJ (2012) Design, synthesis, and molecular hybrids of caudatin and cinnamic acids as novel anti-hepatitis B virus agents. Eur J Med Chem 54:352–365
Westrip SP (2010) publCIF: Software for editing, validating and formatting crystallographic information files. J Appl Crys 43:920–925
Xiao H, Chen M, Shi G, Wang L, Yin H, Mei C (2010) A novel fluorescent molecule based on 1, 8-naphthalimide: synthesis, spectral properties, and application in cell imaging. Res Chem Intermed 36:1021–1026
Yazdani M, Edraki N, Badri R, Khoshneviszadeh M, Iraji A, Firuzi O (2019) Multi-target inhibitors against Alzheimer disease derived from 3-hydrazinyl 1, 2, 4-triazine scaffold containing pendant phenoxy methyl-1, 2, 3-triazole: Design, synthesis and biological evaluation. Bioorg Chem 84:363–371
Zanoza SO, Klimenko KO, Maltzev GV, Bykova TI, Levandovskiy IA, Lyakhov SA (2019) Aminoalkoxyfluorenones and aminoalkoxybiphenyls: DNA binding modes. Bioorg Chem 86:52–60
Zhang G, Shen J, Cheng H, Zhu L, Fang L, Luo S, Muller MT, Lee GE, Wei L, Du Y, Sun D (2005) Syntheses and biological activities of rebeccamycin analogues with uncommon sugars. J Med Chem 48:2600–2611
Zsila F, Bikádi Z, Simonyi M (2004) Circular dichroism spectroscopic studies reveal pH dependent binding of curcumin in the minor groove of natural and synthetic nucleic acids. Org Biomol Chem 2:2902–2910
Acknowledgements
The authors would like to thank the Shiraz University Research Council for financial support (Grant no. 97-GR-SC-23) and Dr. Attila Benyei for collecting X-ray data.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
About this article
Cite this article
Borazjani, N., Behzadi, M., Dadkhah Aseman, M. et al. Cytotoxicity, anticancer, and antioxidant properties of mono and bis-naphthalimido β-lactam conjugates. Med Chem Res 29, 1355–1375 (2020). https://doi.org/10.1007/s00044-020-02552-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-020-02552-1