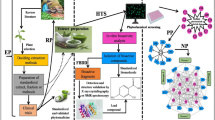
Abstract
Two naturally new cyclic urea and amide derivatives, [1,3]-diazepan-2-one (1) and (S)-1,4-diaza-cyclododecane-2,3-dion (2), were isolated from the organic extract of the sponge Hemimycale aff arabica collected from the Red Sea. This was together with 4-acetamido-2,6-dibromo-4-hydroxy-1,1-dimethoxycyclohexa-2,5-diene (3), 2,4-bis(1-methyl-1-phenylethyl)-phenol (4), β-sitosterol (5), bis-[2-ethyl]-hexyl-phthylester, triglyceride fatty acid ester, and linoleic acid. The chemical structures of compounds 1 and 2 were determined based on 1D and 2D Nuclear Magnetic Resonance (NMR) and HR-ESIMS. Compounds 1 and 2 revealed high potency as antiglycated agents. The molecular docking study of compounds 1 and 2 using α-glucosidase, α-amylase, dipeptidyl peptidase-IV, and glycogen synthase kinase was performed to check their efficiency as hypoglycemic agents. Both the sponge extract and compound 1 showed no cytotoxicity vs. the RPE-1 as a model of normal cell line.





Similar content being viewed by others
References
Alcaraz MJ, Payá M (2006) Marine sponge metabolites for the control of inflammatory diseases. Curr Opin Investig Drugs 7:974–979
Aoki Y, Umezawa N, Asano Y, Hatano K, Yano Y, Kato N, Higuchi T (2007) Bioorg Med Chem 15:7108
Barde SR, Sakhare RS, Kanthale SB, Chandak PG, Jamkhande PG (2015) Marine bioactive agents: a short review on new marine antidiabetic compounds. Asian Pac J Trop Dis 5:S209–S213
Belarbi H, Contreras GA, Chisti Y, García CF, Molina GE (2003) Producing drugs from marine sponges. Biotechnol Adv 21:585–598
Bidon-Chanal A, Fuertes A, Alonso D, Perez DI, Martinez A, Luque FJ, Medina M (2013) Evidence for a new binding mode to GSK-3: allosteric regulation by the marine compound palinurin. Eur J Med Chem 60:479–489
Burkholder PR, Burkholder LM, Almodovar LR (1960) Antibiotic activity of some marine algae of Puerto Rico. Bot Mar 2:149–156
Chapman & Hall Chemical Database (2016) Dictionary of Natural Products on CD-ROM.
Chia-Chen L, Bao-Hong L, Ying-Jang L (2015) Antioxidation and antiglycation of Fagopyrum tataricum ethanol extract. J Food Sci Technol 52:1110–1116
Dávalos A, de la Peña G, Sánchez-Martín CC, Teresa Guerra M, Bartolomé B, Lasunción MA (2009) Effects of red grape juice polyphenols in NADPH oxidase subunit expression in human neutrophils and mononuclear blood cells. Br J Nutr 102:1125–1135
Darko AK, Curran FC, Copin C, McElwee-White L (2011) Carbonylation of functionalized diamine diols to cyclic ureas: application to derivatives of DMP 450 Tetrahedron 67:3976
Dhar A, Desai KM, Wu L (2010) Alagebrium attenuates acute methylglyoxal-induced glucose intolerance in Sprague–Dawley rats. Br J Pharmacol 159:166–175
Dubey S, Ganeshpurkar A, Ganeshpurkar A, Bansal D, Dubey N (2017) Glycolytic enzyme inhibitory and antiglycation potential of rutin. Future J Pharm Sci 3:158–162
Gordaliza M (2010) Cytotoxic terpene quinones from marine sponges. Mar Drugs 8:2849–2870
Haefner B (2003) Drugs from the deep: marine natural products as drug candidates. Drug Discov Today 8:536–544
Hansmannel F, Sillaire A, Kamboh MI, Lendon C, Pasquier F, Hannequin D, Laumet G, Mounier A, Ayral A-M, DeKosky ST, Hauw J-J, Berr C, Mann D, Amouyel P, Campion D, Lambert J-C (2010) Is the urea cycle involved in Alzheimer’s disease? J Alzheimers Dis 21:1013
Higa T, Tanak J, Kitamura A, Koyama T, Takahash M (1994) Bioactive compounds from marine sponges. Pure Appl Chem 66:2227–2230
Hussein SR, Marzouk MM, Soltan MM, Ahmed EK, Said MM, Hamed AR (2017) Phenolic constituents of Pulicaria undulata (L.) C.A. Mey. sub sp. undulata (Asteraceae): antioxidant protective effects and chemosystematic significances. J Food Drug Anal 25:333–339
Khanfar MA, Asal BA, Mudit M, Kaddoumi A, El Sayed KA (2009) The marine natural-derived inhibitors of glycogen synthase kinase-3β phenylmethylene hydantoins: in vitro and in vivo activities and pharmacophore modelling. Bioorg Med Chem 17:6032–6039
Laatsch H (2016) AntiBase. A Data Base for Rapid Structural Determination of Microbial Natural Products, and annual updates. Wiley-VCH, Weinheim, Germany,
Lauritano C, Ianora A (2016) Marine organisms with anti-diabetes properties. Mar Drugs 14:220–233
Li GQ, Tao Z-F, Tong YS, Przytulinska MK, Kovar P, Merta P, Chen ZH, Zhang HY, Sowin T, Rosenberg SH, Lin N-H (2007) Synthesis and in-vitro biological activity of macrocyclic urea Chk1 inhibitors Bioorg Med Chem Lett 17:6499
Malve H (2016) Exploring the ocean for new developments: marine pharmacology. J Pharm Bioallied Sci 8:83–91
Matsuura N, Aradate T, Sasaki C, Kojima H, Ohara M, Hasegawa J, Ubukata M (2002) Screening system for the Maillard reaction inhibitor from natural product extracts. J Health Sci 48:520–526
Maurus R, Begum A, Williams LK, Fredriksen JR, Zhang R, Withers SG, Brayer GD (2008) Alternative catalytic anions differentially modulate human alpha-amylase activity and specificity. Biochemistry 47:3332–3344
Molinski TF, Dalisay DS, Lievens SL, Saludes JP (2009) Drug development from marine natural products. Nat Rev Drug Discov 8:69–85
Naim MJ, Alam MJ, Nawaz F, Naidu VGM, Aaghaz S, Sahu M, Siddiqui N, Alam O (2017) Synthesis, molecular docking and anti-diabetic evaluation of 2,4-thiazolidinedione based amide derivatives. Bioorg Chem 73:24–36
Nakao Y, Uehara T, Matunaga S, Fusetani N, van Soest RWM (2002) Callyspongynic acid, a polyacetylenic acid which inhibits α-glucosidase, from the marine sponge Callyspongia truncata. J Nat Prod 65:922–924
Nakatani H, Leibbrandt NB, Edgecombe M, Miyasaki JM, Paulo-SP, S (2010) U.S. Patent 0 311 589
Pashikanti S, de Alba DR, Boissonneault GA, Cervantes-Laurean D (2010) Rutin metabolites: novel inhibitors of nonoxidative advanced glycation end products. Free Radic Biol Med 48:656–663
Patel M, Rodgers JD, McHugh RJ,Jr Johnson BL, Cordova BC, Klabe RM, Bacheler LT, Erickson-Viitanen S, Ko SS (1999) Unsymmetrical cyclic ureas as HIV-1 protease inhibitors: Novel biaryl indazoles as P2/P2′ substituents Bioorg Med Chem Lett 9:3217
Pedretti A, Villa L, Vistoli G (2004) VEGA—an open platform to develop chemo-bio-informatics applications, using plug-in architecture and script programming. J Comput Aided Mol Des 8:167–173
Perdicaris S, Vlachogianni T, Valavanidis A (2013) Bioactive natural substances from marine sponges: new developments and prospects for future pharmaceuticals. Nat Prod Chem Res 1:3
Perera PRD, Ekanayake S, Ranaweera KKDS (2013) In vitro study on antiglycation activity, antioxidant activity and phenolic content of Osbeckia octandra L. leaf decoction. J Pharmacogn Phytochem 2:198–201
Phadungkit M, Rattarom R, Rattana S (2010) Phytochemical screening, antioxidant, antibacterial and cytotoxic activities of Knema angustifolia extracts. J Med Plants Res 4:1269–1272
Popovic-Djordjevic JB, Jevtic II, Grozdanic ND, Šegan SB, Zlatovic MV, Ivanovic MD, Stanojkovic TP (2017) α-Glucosidase inhibitory activity and cytotoxic effects of some cyclic urea and carbamate derivatives. J Enzyme Inhib Med Chem 32:298–303
Roig-Zamboni V, Cobucci-Ponzano B, Iacono R, Ferrara MC, Germany S, Bourne Y, Parenti G, Moracci M, Sulzenbacher G (2017) Structure of human lysosomal acid α-glucosidase-a guide for the treatment of Pompe disease. Nat Commun 8:1111
Sajid I, Fondja YCB, Shaaban KA, Hasnain S, Laatsch H (2009) Antifungal and antibacterial activities of indigenous Streptomyces isolates from salinefarmlands: prescreening, ribotyping and metabolic diversity W J Microbiol Biotechnol 25:601–610
Schrodinger (2015) The PyMOL Molecular Graphics System, Version 1.8.
Scifinder (2018) Chemical Abstracts Service: Germany. https://scifinder.cas.org/scifinder.
Shaaban KA, Ghani MA, Shaaban M (2015) New cembranoid diterpenes from sarcophyton trocheliophorum. Brit J Pharm Res 5:192–201
Shaaban M, Abd-Alla HI, Hassan AZ, Aly HF, Ghani MA (2012) Chemical characterization, antioxidant and inhibitory effects of some marine sponges against carbohydrate metabolizing enzymes. Org Med Chem Lett 2(30):1–12
Shaaban M, El-Hagrassi AM, Abdelghani MA, Osman AF (2017b) Diverse bioactive compounds from sarcophtyton glaucom: structure elucidation and cytotoxic activity studies. Z Naturforsch C. https://doi.org/10.1515/znc-2017-0106
Shaaban M, El-Metwally MM, Abdel-Razek AA, Laatsch H (2017a) Terretonin M: a new meroterpenoid from the thermophilic Aspergillus terreus TM8 and revision of the absolute configuration of penisimplicins. Nat Prod Res. https://doi.org/10.1080/14786419.2017.1419230
Shaaban M, Ghani MA, Shaaban KA (2013a) Zahramycins A-B, two new steroids from the coral Sarcophyton trocheliophorum. Z Naturforsch 68b:939–945
Shaaban M, Ghani MA, Shaaban KA (2016) Unusual pyranosyl Cembranoid diterpene from Sarcophyton trocheliophorum. Z Naturforsch B 71:1211–1217
Shaaban M, Issa MY, Ghani MA, Hamed A, Abdelwahab AB (2018) New pyranosyl cembranoid diterpenes from Sarcophyton trocheliophorum. Nat Prod Res. https://doi.org/10.1080/14786419.2018.1431631
Shaaban M, Shaaban KA, Abd-Alla HI, Hanna AG, Laatsch H (2011) Dendrophen, a novel glycyrrhetyl amino acid from Dendronephthya hemprichi. Z Naturforsch 66b:425–432
Shaaban M, Shaaban KA, Ghani MA (2013b) Hurgadacin: a new steroid from Sinularia polydactyla. Steroids 78:866–873
Shue H-J, Chen X, Shih N-Y, Blythin DJ, Paliwal S, Lin L, Gu DL, Schwerdt JH, Shah S, Reichard GA, Piwinski JJ, Duffy RA, Lachowicz JE, Coffin VL, Liu F, Nomeir AA, Morgan CA, Varty GB (2005) Cyclic urea derivatives as potent NK1 selective antagonists Bioorg Med Chem Lett 15:3896
Simmons TL, Andrianasolo E, McPhail K, Flatt P, Gerwick WH (2005) Marine natural products as anticancer drugs. Mol Cancer Ther 4:333–342
Sipkema D, Franssen MC, Osinga R, Tramper J, Wijffels RH (2005) Marine sponges as pharmacy. Mar Biotechnol (NY) 7:142–162
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Bokesch H, Kenney S, Boyd MR (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82:1107–1112
Trott O, Olson AJ (2010) AutoDock Vina: improving the speed and accuracy of docking with a new scoring function, efficient optimization, and multithreading. J Comput Chem 31:455–461
VanKaer L (2001) Drugs from the sea: a marine sponge-derived compound prevents Type1 diabetes. Sci World J 1:630–632
Vichai V, Kirtikara K (2006) Sulforhodamine B colorimetric assay for cytotoxicity screening. Nat Protoc 1:1112–1116
Vogl O, Knight AC (1968) Polyoxamides. I. Preparation and characterization of cyclic oxamides. Macromolecules 1:311–315
Wu C, Huang S, Yen GC (2011) Silymarin: a novel antioxidant with antiglycation and antiinflammatory properties in vitro and in vivo. Antioxid Redox Signal 14:353–366
Wu CH, Yen GC (2005) Inhibitory effect of naturally occurring flavonoids on the formation of advanced glycation endproducts. J Agric Food Chem 53:3167–3173
Zhao C, Sham HL, Sun MH, Stoll VS, Stewart KD, Lin SQ, Mo HM, Vasavanonda S, Saldivar A, Park C, McDonald EJ, Marsh KC, Klein LL, Kempf DJ, Norbeck DW (2005) Inhibitory kinetics of novel 2,3-dihydro-1H-inden-1-one chalcone-like derivatives on mushroom tyrosinase Bioorg Med Chem Lett 15:5499
Acknowledgements
The authors are deeply thankful to Prof. H. Laatsch, Institute of Organic and Biomolecular Chemistry, Göttingen, for his support and lab facilities. We thank Dr. H. Frauendorf and Dr. M. John for the mass and NMR spectra. The authors would like to acknowledge Ms. F. Lissy for antimicrobial activity tests. We are thankful to Dr. Mohamed Abd-Elghany (Hurghada, Red Sea, Egypt) for the collection and morphological taxonomy of the studied marine sponge. M. Shaaban thanks the German Academic Exchange Service (DAAD) for a short-term grant.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
No potential conflict of interest was reported by the authors.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Shaaban, M., Hassan, A.Z., Soltan, M.M. et al. Naturally bioactive compounds from Hemimycale aff arabica: antimicrobial, antiglycation, cytotoxicity, and molecular docking studies. Med Chem Res 27, 2079–2088 (2018). https://doi.org/10.1007/s00044-018-2217-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-018-2217-6