Abstract
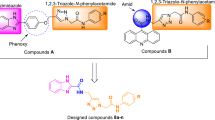
A novel series of benzoxazolyl linked benzylidene based rhodanine and their cyclic analogs were synthesized, characterized and evaluated for their α-amyloglucosidase inhibitory activity. Out of eight target compounds, two compounds (4b and 5b) displayed potent inhibitory activity against α-amyloglucosidase with IC50 values in the range of 0.24 ± 0.01–0.94 ± 0.01 µM as compared to standard drug acarbose. Among all the tested compounds, compound 5b containing rhodanine at 3-position of phenyl was found to be the most active inhibitor of α-amyloglucosidase. Docking studies showed the existence of potential H-bonding interactions between synthesized compounds and α-glucosidase which might be responsible for good biological activity.



Similar content being viewed by others
References
Ertan-Bolelli T, Yildiz I, Ozgen-Ozgacar S (2016) Synthesis, molecular docking and antimicrobial evaluation of novel benzoxazole derivatives. Med Chem Res 25:553–567
Ghani U (2015) Re-exploring promising alpha-glucosidase inhibitors for potential development into oral anti-diabetic drugs: Finding needle in haystack. Eur J Med Chem 103:133–162
Gray GM (1975) Carbohydrates digestion and absorption-role of small intestine. N Eng J Med 292:1225–1230
Kasabri V, Afifi FU, Hamdan I (2011) In vitro and in vivo acute antihypergycemic effects of five selected indigenous plants from Jordan used in tradiational medicine. J Ethpharm 133:888–896
Lazer ES, Miao CK, Wong HC, Sorcek R, Spero DM, Gilman A, Pal K, Behnke M, Graham AG, Watrous JM, Homon CA, Nagel J, Shah A, Guindon Y, Farina PR, Adams J (1994) Benzoxazolamines and benzthiazolamines: potent, enantioselective inhibitors of leukotriene biosynthesis with a novel mechanism of action. J Med Chem 37:913–923
Notkins AL (2002) Immunologic and genetic factors in type 1 diabetes. J Biol Chem 277:43545–43548
Rida SM, Ashour FA, El-Hawash SAM, ElSemary MM, Badr MH, Shalaby MA (2005) Synthesis of some novel benzoxazole derivatives as anticancer anti-HIV-1 and antimicrobial agents. Eur J Med Chem 40:949–959
Seth K, Garg SK, Kumar R, Purohit P, Meena VS, Goyal R, Banergee UC, Chakraborti AK (2014) 2-(2-Arylphenyl)benzoxazole as a novel anti-inflammatory scaffold: synthesis and biological evaluation. Med Chem Lett 5:512–516
Shaw JE, Sicree RA, Zimmet PZ (2010) Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 87:4–14
Singh A, Verma RK, Kuhad A, Mall R (2017) Novel indol-2-carboxalic acid linked 3-phenyl-2-alkoxy propanoic acids: Synthesis, molecular docking and in vivo antidiabetic studies. Med Chem Res 26:745–759
Sybyl 7. 3 (2006) Tripose Inc. 1699 Hanley Road, St. Louis63144, USA
Tipparaju SK, Joyasawal S, Pieroni M, Kaiser M, Brun R, Kozikowski AP (2008) In Prursuit of Natural Product Leads: Synthesis and Biological Evaluation of 2-[3-hydroxy-2-[(3-hydroxy pyridine-2-carbonyl)amino]phenyl]benzoxazole-4-carboxylic acid (A-33853) and Its Analogues: Discovery of N-(2-Benzoxazole-2-ylphenyl)benzamides as Novel Antileishmanial Chemotypes. J Med Chem 51:7344–7347
Verma RK, Ghosh P, Kumar V, Wadhwa LK (2012a) The application of comparative molecular field analysis for the design of α–anilino substituted-3-phenyl propanoic acids as novel PPARα/γ dual ligands. Med Chem Res 21:2873–2884
Verma RK, Ghosh P, Kumar V, Wadhwa LK (2013a) Design, synthesis and computational validation of novel benzimidazole/indole-based PPARα and PPARγ partial agonists. J Chem Sci 125:1555–1571
Verma RK, Kumar V, Ghosh P, Wadhwa LK (2012b) Heterocyclyl linked anilines and benzaldehydes as precursors for biologically significant new chemical entities. J Chem Sci 124:1063–1069
Verma RK, Kumar V, Ghosh P, Wadhwa LK (2013b) 3D-QSAR study of tyrosine and propanoic acid derivatives as PPARα/γ dual agonists using CoMSIA. Med Chem Res 22:287–302
Verma RK, Mall R, Ghosh P, Kumar V (2013c) Design and synthesis of benzimidazole-linked meta-substituted benzylidenes/benzyls as biologically significant new chemical entities. Synth Commun 43:1882–1895
Verma RK, Mall R, Singh A (2015) Indole Linked meta-subsituted benzylidene-based novel PPAR ligands: synthetic and docking studies. Med Chem Res 24:1396–1407
Acknowledgements
Authors are thankful to the Punjabi University, Patiala authorities for providing the necessary research facilities. We are also grateful to Director and Mr. Avtar Singh of Sophisticated Analytical Instrumentation Facility (SAIF), Panjab University, Chandigarh, respectively, for extending the facilities for spectral analysis of the compounds reported in this paper. One of the authors, Mr. Varinder Singh, is thankful to UGC for providing Maulana Azad National fellowship.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Electronic supplementary material
Rights and permissions
About this article
Cite this article
Singh, V., Singh, A., Singh, G. et al. Novel benzoxazole derivatives featuring rhodanine and analogs as antihypergycemic agents: synthesis, molecular docking, and biological studies. Med Chem Res 27, 735–743 (2018). https://doi.org/10.1007/s00044-017-2097-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-2097-1