Abstract
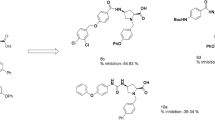
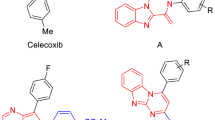
A series of chromone derivatives have been evaluated as potential cyclooxygenase-2 (COX-2) inhibitors. The four most potent compounds, 48, 41, 39, and 35 displayed IC50 values of 3.30, 6.86, 7.36 and 7.46 µM, respectively. Compounds 35 and 38 showed higher selectivity for COX-2 (selectivity index, SI = 7.48 and 5.46, respectively) than celecoxib (SI = 4.17 in the same test) whereas compound 39 showed comparable selectivity (SI = 4.19) to celecoxib. The molecular volumes of compounds 35 (312.84 Å3) and 38 (314.18 Å3) were similar to celecoxib (299.28 Å3) but larger than ibuprofen (211.83 Å3). Docking results were in good agreement with the experimental biological data in terms of evaluation of binding energy and binding mode. Compounds 35, 38, and 39 had higher binding affinity against COX-2 (binding energy between −9.77 and −11.42 kcal/mole) than COX-1 (binding energy between −6.28 and −7.88 kcal/mole). These three chromone compounds also displayed active conformation in the same orientation as that of celecoxib. Thus, compounds in this series has the potential to be a new class of selective COX-2 inhibitor.





Similar content being viewed by others
References
Cheong E, Ivory K, Doleman J, Parker ML, Rhodes M, Johnson IT (2004) Synthetic and naturally occurring COX-2 inhibitors suppress proliferation in a human oesophageal adenocarcinoma cell line (OE33) by inducing apoptosis and cell cycle arrest. Carcinogenesis 25:1945–1952
Crofford L, Wilder R, Ristimaki A, Sano H, Remmers E, Epps H, Hla T (1994) Cyclooxygenase-1 and -2 expression in rheumatoid synovial tissues. Effects of interleukin-1 beta, phorbol ester, and corticosteroids. J Clin Invest 93:1095–1101
DeWitt DL (1999) Cox-2-selective inhibitors: the new super aspirins. Mol Pharm 55:625–631
Flower RJ (2003) The development of COX2 inhibitors. Nat Rev Drug Discov 2:179–191
Fu JY, Masferrer JL, Seibert K, Raz A, Needleman P (1990) The induction and suppression of prostaglandin H2 synthase (cyclooxygenase) in human monocytes. J Biol Chem 265:16737–16740
Gacche RN, Meshram RJ, Shegokar HD, Gond DS, Kamble SS, Dhabadge VN, Utage BG, Patil KK, More RA (2015) Flavonoids as a scaffold for development of novel anti-angiogenic agents: an experimental and computational enquiry. Arch Biochem Biophys 577:35–48
Gierse JK, McDonald JJ, Hauser SD, Rangwala SH, Koboldt CM, Seibert K (1996) A single amino acid difference between cyclooxygenase-1 (COX-1) and -2 (COX-2) reverses the selectivity of COX-2 specific inhibitors. J Biol Chem 271:15810–15814
Guo Q, Wang LH, Ruan KH, Kulmacz RJ (1996) Role of Val509 in time-dependent inhibition of human prostaglandin H synthase-2 cyclooxygenase activity by isoform-selective agents. J Biol Chem 271:19134–19139
Habeeb G, Rao PNP, Knaus EE (2001) Design and synthesis of 4,5-diphenyl-4-isoxazolines: novel inhibitors of cyclooxygenase-2 with analgesic and antiinflammatory activity. J Med Chem 44:2921–2927
Havsteen B (1983) Flavonoids, a class of natural products of high pharmacological potency. Biochem Pharmacol 32:1141–1148
Kurumbail RG, Stevens AM, Gierse JK, McDonald JJ, Stegeman RA, Pak JY, Gildehaus D, Miyashiro JM, Penning TD, Seibert K, Isakson PC, Stallings WC (1996) Structural basis for selective inhibition of cyclooxygenase-2 by anti-inflammatory agents. Nature 384:644–648
Lerdsirisuk P, Maicheen C, Ungwitayatorn J (2014) Antimalarial activity of HIV-1 protease inhibitor in chromone series. Bioorg Chem 57:142–147
Lin S-J, Tsai W-J, Chiou W-F, Yang T-H, Yang L-M (2008) Selective COX-2 inhibitors. Part 2: synthesis and biological evaluation of 4-benzylideneamino- and 4-phenyliminomethyl-benzenesulfonamides. Bioorg Med Chem 16:2697–2706
Lu X, Zhang H, Li X, Chen G, Li Q-S, Luo Y, Ruan B-F, Chen X-W, Zhu H-L (2011) Design, synthesis and biological evaluation of pyridine acyl sulfonamide derivatives as novel COX-2 inhibitors. Bioorg Med Chem 19:6827–6832
Luong C, Miler A, Barnett J, Chow J, Ramesha C, Browner MF (1996) Flexibility of the NSAID binding site in the structure of human cyclooxygenase-2. Nat Struct Bio 3:927–933
O’Leary KA, de Pascual-Teresa S, Needs PW, Bao YP, O’Brien NM, Williamson G (2004) Effect of flavonoids and vitamin E on cyclooxygenase-2 (COX-2) transcription. Mutat Res 551:245–254
Phosrithong N, Samee W, Nunthanavanit P, Ungwitayatorn J (2012) In Vitro antioxidant activity study of novel chromone derivatives. Chem Biol Drug Des 79:981–989
Simmons DL, Botting RM, Hla T (2004) Cyclooxygenase Isozymes: the biology of prostaglandin synthesis and inhibition. Pharmacol Rev 56:387–437
Suh Y, Afaq F, Johnson JJ, Mukhtar H (2009) A plant flavonoid fisetin induces apoptosis in colon cancer cells by inhibition of COX2 and Wnt/EGFR/NF-kB-signaling pathways. Carcinogenesis 30:300–307
Uddin MJ, Rao PNP, Rahim MA, McDonald R, Knaus EE (2004) A new class of acyclic 2-alkyl-1,2-diaryl (E)-olefins as selective cyclooxygenase-2 (COX-2) inhibitors. Bioorg Med Chem Lett 14:4911–4914
Uddin MJ, Rao PNP, McDonald R, Knaus EE (2005) Design and synthesis of (E)-1,1,2-triarylethenes: novel inhibitors of the cyclooxygenase- 2 (COX-2) isozyme. Bioorg Med Chem Lett 15:439–442
Ungwitayatorn J, Wiwat C, Samee W, Nunthanavanit P, Phosrithong N (2011) Synthesis, in vitro evaluation, and docking studies of novel chromone derivatives as HIV-1 protease inhibitor. J Mol Struct 1001:152–161
Vane JR, Bakhle YS, Botting RM (1998) Cyclooxygenase 1 and 2. Annu Rev Pharmacol Toxicol 38:97–120
Wang JL, Carter J, Kiefer JR, Kurumbail RG, Pawlitz JL, Brown D, Hartmann SJ, Graneto MJ, Seibert K, Talley JJ (2010) The novel benzopyran class of selective cyclooxygenase-2 inhibitors-part I: the first clinical candidate. Bioorg Med Chem Lett 20:7155–7158
Xie WL, Chipman JG, Robertson DL, Erikson RL, Simmons DL (1991) Expression of a mitogen-responsive gene encoding prostaglandin synthase is regulated by mRNA splicing. Proc Natl Acad Sci USA 88:2692–2696
Yucel T, Ahola H, Carlsted J, Modeer T (1999) Involvement of tyrosine kinases on cyclooxygenase expression and prostaglandin E2 production in human gingival fibroblasts stimulated with interleukin-1 beta and epidermal growth factor. Biochem Biophys Res Commun 257:528–532
Zarghi A, Arfaee S, Rao PNP, Knaus EE (2006) Design, synthesis and biological evaluation of 1,3-diarylprop-2-en-1-ones: a novel class of cyclooxygenase-2 inhibitors. Bioorg Med Chem 14:2600–2605
Zarghi A, Rao PNP, Knaus EE (2007) Design and synthesis of new rofecoxib analogs as selective cyclooxygenase-2 (COX-2) inhibitors: replacement of the methanesulfonyl pharmacophore by an N-acetylsulfonamido bioisostere. J Pharm Pharmaceut Sci 10:159–167
Zarghi A, Kakhgi S, Hadipoor A, Daraee B, Dadrass OG, Hedayati M (2008) Design and synthesis of 1,3-diarylurea derivatives as selective cyclooxygenase (COX-2) inhibitors. Bioorg Med Chem Lett 18:1336–1339
Acknowledgements
The authors thank the High Performance Computer Center (HPCC), National Electronic and Computer Technology Center (NECTEC) of Thailand for providing SYBYL facilities.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Rights and permissions
About this article
Cite this article
Maicheen, C., Phosrithong, N. & Ungwitayatorn, J. Biological activity evaluation and molecular docking study of chromone derivatives as cyclooxygenase-2 inhibitors. Med Chem Res 26, 662–671 (2017). https://doi.org/10.1007/s00044-017-1786-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-017-1786-0