Abstract
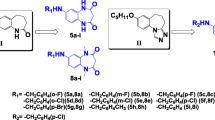
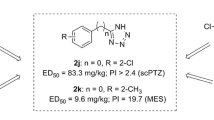
In the present study on the development of new anticonvulsants, twelve new aryl sulfonyl semicarbazide derivatives were synthesized and tested for anticonvulsant activity using maximal electroshock (MES), subcutaneous pentylenetetrazole screens. Their neurotoxicity was determined by the rotorod test. The most active compound 5i showed the MES-induced seizures with ED50 value of 7.3 mg/kg and TD50 value of 402.3 mg/kg after intraperitoneally injection to mice, which provided compound 5i with a protective index (TD50/ED50) of 55.1 in the MES test.




Similar content being viewed by others
References
Audrieth LF, Brhuchitsch MV (1956) Hydrazides of sulfuric acid and their derivatives. II. The sulfamyl hydrazides. J Org Chem 21:426–428
Bialer M (2006) New antiepileptic drugs that are second generation to existing antiepileptic drugs. Expert Opin Investig Drugs 15:637–647
Bojic U, Elmazar MMA, Hauck RS, Nau H (1996) Further branching of valproate-related carboxylic acids reduces the teratogenic activity, but not the anticonvulsant effect. Chem Res Toxicol 9:866–870
Brown TR, Holmes GI (2001) Primary care: epilepsy. N Engl J Med 344:1145–1151
Dimmock JR, Pandey SN, Quail JW, Pugazhenthi U, Allen TM, Kao GY, Balzarini J, DeClercq E (1995) Evaluation of the semicarbazones, thiosemicarbazones and bis-carbohydrazones of some aryl alicyclic ketones for anticonvulsant and other biological propertie. Eur J Med Chem 30:303–314
Dunham NW, Miya TA, Edwards LD (1957) The pharmacological activity of a series of basic esters of mono- and dialkylmalonic acids. J Am Pharm Assoc 46:64–66
Emilio P (1999) The clinical pharmacokinetics of the new antiepileptic drugs. Epilepsia 40(Suppl 9):7–13
Finney DJ (1971) Statisical logic in the monitoring of reactions to therapeutic drugs. Methods Inf Med 10:237–245
He X, Zhong M, Zhang T, Wu W, Wu Z, Yang J, Xiao Y, Pan Y, Qiu G, Hu X (2010) Design, synthesis, and anticonvulsant activity of N-3-arylamide substituted 5,5-cyclopropanespirohydantoin derivatives. Eur J Med Chem 45:5870–5877
He X, Zhong M, Zhang T, Yang J, Wu Z, Xiao Y, Guo H, Qiu G, Hu X (2012a) Synthesis and anticonvulsant activity of 1-(8-(benzyloxy)quinolin-2-yl)-6-substituted-4,6-diazaspiro[2,4]heptane-5,7-diones. Eur J Med Chem 48:338–346
He X, Zhong M, Yang J, Wu Z, Xiao Y, Guo H, Hu X (2012b) Synthesis and anticonvulsant activity of 1-(2-(8-(benzyloxy)quinolin-2-yl)-1-butyrycyclopropyl)3-substituted urea derivatives. Chem Biol Drug Des 79:771–779
James ES, Lesley J (2005) Zonisamide. CNS Drugs 19:347–367
Kaminski K, Obniska J (2008) Design, synthesis, and anticonvulsant activity of N-phenylamino derivatives of 3,3-dialkyl-pyrrolidine-2,5-diones and hexahydro-isoindole-1,3-diones. Bioorg Med Chem 16:4921–4931
Krall RL, Penry JK, White BG, Kupferberg HJ, Swinyard EA (1978) Antiepileptic drug development: II. Anticonvulsant drug screening. Epilepsia 19:409–428
Kwan P, Sander JW (2004) The natural history of epilepsy: an epidemiological view. J Neurol Neurosurg Psychiatry 75:1376–1381
Malawska B (2005) New anticonvulsant agents. Curr Top Med Chem 5:69–85
Nie Y, Zhong M, Gao Y, Ding F, Li H, Zhang Y, He X (2015) Synthesis and potential anticonvulsant activity of new 5,5-cyclopropanespirohydantoin derivatives. Med Chem Res 24:4207–4216
Perucca E, French J, Bialer M (2007) Development of new antiepileptic drugs: challenges, incentives, and recent advances. Lancet Neurol 6:793–804
Rajak H, Deshmukh R, Aggarwal N, Kashaw S, Kharya MD, Mishra P (2009) Synthesis of novel 2,5-disubstituted 1,3,4-thiadiazoles for their potential anticonvulsant activity: pharmacophoric model studies. Arch Pharm 342:453–461
Salgado A, Huybrechts T, Eeckhaut A, Van der Eycken J, Szakonyi Z, Fülöp Tkachev A, De Kimpe N (2001) Synthesis of (1S)-1-amino-2,2-dimethylcyclopropane-1-carboxylic acid via PLE mediated hydrolysis of bis(2,2,2-trifluoroethyl) 2,2-dimethylcyclopropane-1,1-dicarboxylate. Tetrahedron 57:2781–2786
Smith M, Wilcox KS, White HS (2007) Discovery of antiepileptic drugs. Neurotherapeutics 4:12–17
Swinyard EA (1969) Laboratory evaluation of antiepileptic drugs. Epilepsia 10:107–119
Swinyard EA, Brown WC, Goodman LS (1952) Comparative assays of antiepileptic drugs in mice and rats. J Pharmacol Exp Ther 106:319–330
Thirumurugan R, Sriram D, Saxena A, Stables J, Yogeeswari P (2006) 2,4-Dimethoxyphenylsemicarbazones with anticonvulsant activity against three animal models of seizures: synthesis and pharmacological evaluation. Bioorg Med Chem 14:3106–3112
Ucar H, Kim VD, Cacciaguerra S, Spampinato S, Stables JP, Depovere P, Isa M, Masereel B, Delarge J, Poupaert JH (1998) Synthesis and anticonvulsant activity of 2 (3H)-benzoxazolone and 2 (3H)-benzothiazolone derivatives. J Med Chem 41:1138–1145
Vamecq J, Lambert D, Poupaert JH, Masereel B, Stables JP (1998) Anticonvulsant activity and interactions with neuronal voltage-dependent sodium channel of analogs of ameltolide. J Med Chem 41:3307–3313
Verhe R, De Kimpe N, De Buyck L, Courtheyn D, Schamp N (1977) Nucleophilic substitution reactions of brominated dimethyl alkylidene malonates. Bull Soc Chim Belg 86:55–63
White HS, Woodhead JH, Wilcox KS, Stables JP, Kupferberg HJ, Wolf HH, Levy RH, Mattson RH, Meldrum BS, Perucca E (2002) Antiepileptic drugs. Lippincott Williams & Wilkins Publishers, New York, pp 36–48
Yogeeswari P, Ragavendran JV, Sriram D (2007) Neuropathic pain: strategies in drug discovery and treatment. Expert Opin Drug Discov 2:169–184
Zaccara G, Franciotta D, Perucca E (2007) Idiosyncratic adverse reactions to antiepileptic drugs. Epilepsia 48:1223–1244
Zhong M, Zhang Y, He X (2014) Synthesis and anticonvulsant activity of ethyl 2,2-dimethyl-1-(2-substitutedhydrazinecarboxamido)cyclopropanecarboxylate derivatives. Chem Biol Drug Des 84:234–241
Acknowledgments
This work is supported by National Science Foundation of China (NSFC) (Grant No: 21302065), Scientific Research Foundation for the Returned Overseas Chinese Scholars, State Education Ministry of China and Technology Foundation for Selected Overseas Chinese Scholar, Ministry of Personnel of China.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nie, Y., Zhong, M., Jiang, Z. et al. Synthesis and potential anticonvulsant activity of new aryl sulfonyl semicarbazide derivatives. Med Chem Res 25, 1425–1432 (2016). https://doi.org/10.1007/s00044-016-1582-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-016-1582-2