Abstract
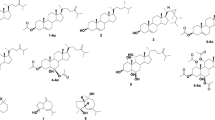
From diethyl ether fraction of the Red Sea soft coral, Litophyton arboreum, two new terpenoidal derivatives, himachalene-type sesquiterpene (3α,6α-epidioxyhimachal-1-ene, 1) and secosteroidal derivative (22-norerogostane derivative, 13,14-seco-22-norergosta-4,24(28)-dien-19-hydroperoxide-3-one, 2), were identified. In addition to di-acetyl cembranoid derivative, (3) was isolated for the first time from the nature. Along with three known metabolites, sarcophytol M (4), 7β-acetoxy-24-methylcholesta-5-24(28)-diene-3,19-diol (5) and erythro-N-dodecanoyl-docosasphinga-(4E,8E)-dienine (6) were obtained. Spectroscopic measurements including 1D and 2D NMR were used for identification of chemical structures. All compounds were evaluated to assess their antiproliferative effect toward three different cancer cell lines MCF-7 (human breast carcinoma), HCT116 (human colon cancer) and HepG-2 (human hepatic carcinoma) in comparison with standard anticancer drug (Doxorubicin) by employing SRD assay. Compound 3 showed significant antiproliferative activities to MCF-7 (IC50 19.1 ± 0.032 µM), to HepG2 (IC50 24.0 ± 0.032 µM) and to HCT116 (IC50 22.0 ± 0.092 µM). The antiproliferative activities of 3 can be attributed, at least partly, to their ability to do cell cycle arrest, particularly, in G0/G1 and S phases.



Similar content being viewed by others
References
Al-Abdin OZ, Rabah DM, Badr G, Kotb A, Aprikian A (2013) Differences in prostate cancer detection between Canadian and Saudi populations. Braz J Med Biol Res 46:539–545
Boyle P, Severi G, Giles GG (2003) The epidemiology of prostate cancer. Urol Clin North Am 30:209–217
Ellithey MS, Lall N, Hussein AA, Meyer D (2013) Cytotoxic, cytostatic and HIV-1 PR inhibitory activities of the soft coral Litophyton arboretum. Mar Drugs 11:4917–4936
Ginsburg OM (2013) Breast and cervical cancer control in low and middle-income countries: human rights meet sound health policy. J Cancer Policy 1:e35–e41
Goad LG (1978) The sterols of marine inverterbrates: composition, biosynthesis, and metabolites. In: Scheuer PJ (ed) Marine natural products, chemical and biological perspectives, vol 2. Academic Press, New York, p 76
Grote D, Dahse H-M, Seifert K (2008) Furanocembranoids from the soft corals Sinularia asterolobata and Litophyton arboretum. Chem Biodivers 5:2449–2456
Li R, Wang S, Tan G, Long K (1994) Two polyhydroxylated steroids from the Chinese soft coral Litophyton arboretum. Steroids 59:503–505
Narula APS, Sukh DEV (1977) Studies in sesquiterpenes-LI: β-Himachalene epoxide-stereochemistry and solvolysis. Tetrahedron 33:813–816
Rezanka TV, Dembitsky VM (2001) γ-Lactones from the soft corals Sarcophyton trocheliophorum and Lithophyton arboreum. Tetrahedron 57:8743–8749
Schmitz FJ (1978) Uncommon marine steroids. In: Scheuer PJ (ed) Marine natural products, chemical and biological perspectives, vol 1. Academic Press, New York, p 241
Shaker KH, Mueller M, Ghani MA, Dahse H-M, Seifert K (2010) Terpenes from the Soft Corals Litophyton arboreum and Sarcophyton ehrenbergi. Chem Biodivers 7:2007–2015
Siegel R, DeSantis C, Virgo Stein K, Mariotto A, Smith T, Dr Cooper, Gansler T, Lerro C, Fedewa S, Lin Ch, Leach C, CannadyRl S, Cho H, Scoppa S, Hachey M, Kirch R, Jemal A, Ward E (2012) Cancer treatment and survivorship statistics. Cancer J Clin 62:220–241
Skehan P, Storeng R, Scudiero D, Monks A, McMahon J, Vistica D, Warren JT, Bokesch H, Kenney S, Boyd MR (1990) New colorimetric cytotoxicity assay for anticancer-drug screening. J Natl Cancer Inst 82:1107–1112
Soliman FM, Fathy MM, Salama MM, Al-Abd AM, Saber FR, El-Halawany AM (2014) Cytotoxic activity of acyl phloroglucinols isolated from the leaves of Eucalyptus cinerea F. Muell. exBenth. cultivated in Egypt. Sci Rep 4:5410–5415
Su B-N, Takaishi Y (1999) Morinins H-K, four novel phenylpropanol ester lipid metabolites from morinachonesis. J Nat Prod 62:1325–1327
Wei X, Rodríguez AD, Baran P, Raptis RG, Sánchez JA, Ortega-Barria E, González J (2004) Antiplasmodial cembradiene diterpenoids from a Southwestern Caribbean gorgonian octocoral of the genus Eunicea. Tetrahedron 60:11813–11819
Acknowledgments
This project was funded by the Deanship of Scientific Research (DSR), King Abdulaziz University, Jeddah, under Grant No. (279/150/1434). The authors, therefore, acknowledge with thanks DSR for technical and financial support and Mr. Kamal Aldahody, Faculty of maritime studies, King Abdulaziz University, for collection of the black coral sample.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ghandourah, M.A., Alarif, W.M., Abdel-Lateff, A. et al. Two new terpenoidal derivatives: a himachalene-type sesquiterpene and 13,14-secosteroid from the soft coral Litophyton arboreum . Med Chem Res 24, 4070–4077 (2015). https://doi.org/10.1007/s00044-015-1456-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-015-1456-z