Abstract
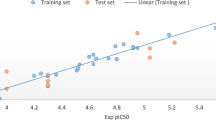
Growth suppression of many non-COX-2 expressing tumor cells can be exhibited by COX-2 inhibitors, where supplementation of cells with exogenous prostaglandins fails to rescue the cells from growth inhibition. It can, therefore, be speculated that anti-cancer properties of some COX-2 inhibitors may be contributed by the COX-2-independent effects also. Some of the derivatives obtained from certain COX-2 inhibitors which show non-COX-2 inhibitory mechanism have revealed some significant anti-cancer activities. From a COX-2 selective inhibitor nimesulide, an analog JCC76 is derived which is a non-COX-2 active compound and shows inhibition of SKBR-3 breast cancer cell growth. Other JCC76 derived inhibitors also played significant role in SKBR-3 cell inhibition. An analog-based study was done using pharmacophore modeling and 3D-QSAR to provide clues for potential lead compound designing. A five point pharmacophore ADHRR was generated using 39 JCC76-derived SKBR-3 inhibitors. The validated pharmacophore alignment was used for further 3D-QSAR analysis, which presented a good R 2 value of 0.562, 0.982, and 0.848 for atom-based QSAR, CoMFA, and CoMSIA model, respectively. All the QSAR models presented good statistical significance and predictivity. The corresponding Q 2 values for each model are 0.513, 0.649, and 0.518, respectively. Both the pharmacophore and CoMSIA results displayed that the H-bond donor and acceptor sites are the key structural feature for JCC76-derived non-COX-2-dependent inhibitors with high activity.






Similar content being viewed by others
References
Almerico AM, Tutone M, Lauria A (2010) 3D-QSAR pharmacophore modeling and in silico screening of newBcl-xl inhibitors. Eur J Med Chem 45:4774–4782
Boelsterli UA, Ho HK, Zhou S, Leow KY (2006) Bioactivation and hepatotoxicity of nitroaromatic drugs. Curr Drug Metab 7:715–727
Boland GP, Butt IS, Prasad R, Knox WF, Bundred NJ (2004) COX-2 expression is associated with an aggressive phenotype in ductal carcinoma in situ. Br J Cancer 90:423–429
Chen B, Su B, Chen S (2009) A COX-2 inhibitor nimesulide analog selectively induces apoptosis in Her2 overexpressing breast cancer cells via cytochrome c dependent mechanisms. Biochem Pharmacol 77:1787–1794
Cramer RD, Patterson DE, Bunce JD (1988) Comparative molecular field analysis (CoMFA). 1. Effect of shape on binding of steroids to carrier proteins. J Am Chem Soc 110:5959–5967
Deasy BM, O’Sullivan-Coyne G, O’Donovan TR, McKenna SL, O’Sullivan GC (2007) Cyclooxygenase-2 inhibitors demonstrate anti-proliferative effects in oesophageal cancer cells by prostaglandin E(2)-independent mechanisms. Cancer Lett 256:246–258
Dixon SL, Smondyrev AM, Rao SN (2006) PHASE: a novel approach to pharmacophore modeling and 3D database searching. Chem Biol Drug Des 67:370–372
Eberhart CE, Coffey RJ, Radhika A, Giardiello FM, Ferrenbach S, DuBois RN (1994) Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 107:1183–1188
Eibl G, Reber HA, Wente MN, Hines OJ (2003) The selective cyclooxygenase-2 inhibitor nimesulide induces apoptosis in pancreatic cancer cells independent of COX-2. Pancreas 26:33–41
Furukawa F, Nishikawa A, Lee IS, Kanki K, Umemura T, Okazaki K, Kawamori T, Wakabayashi K, Hirose M (2003) A cyclooxygenase-2 inhibitor, nimesulide, inhibits postinitiation phase of N-nitrosobis(2-oxopropyl)amineinduced pancreatic carcinogenesis in hamsters. Int J Cancer 104:269–273
Guasch L, Sala E, Valls C, Mulero M, Pujadas G, Garcia-Vallve S (2012) Development of docking-based 3D-QSAR models for PPARgamma full agonists. J Mol Graph Model 36:1–9
Han S, Roman J (2005) COX-2 inhibitors suppress integrin alpha5 expression in human lung carcinoma cells through activation of Erk: involvement of Sp1 and AP-1 sites. Int J Cancer 116:536–546
Han S, Roman J (2006) COX-2 inhibitors suppress lung cancer cell growth by inducing p21 via COX-2 independent signals. Lung Cancer 51:283–296
Hanif R, Pittas A, Feng Y, Koutsos MI, Qiao L, Staiano-Coico L, Shiff SI, Rigas B (1996) Effects of nonsteroidal anti-inflammatory drugs on proliferation and on induction of apoptosis in colon cancer cells by a prostaglandinindependent pathway. Biochem Pharmacol 52:237–245
Hida T, Kozaki K, Muramatsu H, Masuda A, Shimizu S, Mitsudomi T, Sugiura T, Ogawa M, Takahashi T (2000) Cyclooxygenase-2 inhibitor induces apoptosis and enhances cytotoxicity of various anticancer agents in non-small cell lung cancer cell lines. Clin Cancer Res 6:2006–2011
Johnson AJ, song X, Hsu A, Chen C (2001) Apoptosis signaling pathways mediated by cyclooxygenase-2 inhibitors in prostate cancer cells. Adv Enzym Regul 41:221–235
Julemont F, de Leval X, Michaux C, Damas J, Charlier C, Durant F, Pirotte B, Dogne JM (2002) Spectral and crystallographic study of pyridinic analogues of nimesulide: determination of the active form of methanesulfonamides as COX-2 selective inhibitors. J Med Chem 45:5182–5185
Kawamori T, Nakatsugi S, Ohta T, Sugimura T, Wakabayashi K (2002) Chemopreventive effects of nimesulide, a selective cyclooxygenase-2 inhibitor, against PhIP-induced mammary carcinogenesis. Adv Exp Med Biol 507:371–376
Keller JJ, Giardiello FM (2003) Chemoprevention strategies using NSAIDs and COX-2 inhibitors. Cancer Biol Ther 2:140–149
Klebe G, Abraham U, Mietzner T (1994) Molecular similarity indices in a comparative molecular field analysis (CoMFA) of drug molecules to correlate and predict their biological activity. J Med Chem 37:4130–4146
Li JY, Wang XZ, Chen FL, Yu JP, Luo HS (2003) Nimesulide inhibits proliferation via induction of apoptosis and cell cycle arrest in human gastric adenocarcinoma cell line. World J Gastroenterol 9:915–920
Liang M, Yang H, Fu J (2009) Nimesulide inhibits IFN-gamma-induced programmed death-1-ligand 1 surface expression in breast cancer cells by COX-2 and PGE2 independent mechanisms. Cancer Lett 276:47–52
Liu CH, Chang SH, Narko K, Trifan OC, Wu MT, Smith E, Haudenschild C, Lane TF, Hla T (2001) Overexpression of cyclooxygenase-2 is sufficient to induce tumorigenesis in transgenic mice. J Biol Chem 276:18563–18569
Mazhar D, Gillmore R, Waxman J (2005) COX and cancer. QJM 98:711–718
Mehta N, Chand S, Bahia MS, Silakari O (2012) Elaboration of new anti-inflammatory agents using pharmacophore based 3D QSAR of 4, 5-diaryl imidazoline as P2X(7) receptor antagonists. Lett Drug Des Discov 9:185–198
Nagamani S, Muthusamy K, Kirubakaran P, Singh KD, Krishnasamy G (2012) Theoretical studies on benzimidazole derivatives as E. coli biotin carboxylase inhibitors. Med Chem Res 21:2169–2180
Nair SB, Teli MK, Pradeep H, Rajanikant G (2012) Computational identification of novel histone deacetylase inhibitors by docking based QSAR. Comput Biol Med 42:697–705
Nakatsugi S, Ohta T, Kawamori T, Mutoh M, Tanigawa T, Watanabe K, Sugie S, Sugimura T, Wakabayashi K (2000) Chemoprevention by nimesulide, a selective cyclooxygenase-2 inhibitor, of 2-amino-1-methyl-6- phenylimidazo[4,5-b]pyridine (PhIP)-induced mammary gland carcinogenesis in rats. Jpn J Cancer Res 91:886–892
Nam KT, Hahm KB, Oh SY, Yeo M, Han SU, Ahn B, Kim YB, Kang JS, Jang DD, Yang KH, Kim DY (2004) The selective cyclooxygenase-2 inhibitor nimesulide prevents Helicobacter pylori-associated gastric cancer development in a mouse model. Clin Cancer Res 10:8105–8113
Olsen JH, Friis S, Poulsen AH, Fryzek J, Harving H, Tjonneland A, Sorensen HT, Blot W (2008) Use of NSAIDs, smoking and lung cancer risk. Br J Cancer 98:232–237
Pan Y, Zhang JS, Gazi MH, Young CY (2003) The cyclooxygenase 2-specific nonsteroidal anti-inflammatory drugs celecoxib and nimesulide inhibit androgen receptor activity via induction of c-Jun in prostate cancer cells. Cancer Epidemiol Biomarkers Prev 12:769–774
Peter B, Bernard L (2008) World Cancer Report, WHO
PHASE 3.4 User Manual (2012) Schrödinger. http://www.schrodinger.com/
Rao CV, Reddy BS (2004) NSAIDs and chemoprevention. Curr Cancer Drug Targets 4:29–42
Reddy KK, Singh SK, Dessalew N, Tripathi SK, Selvaraj C (2012) Pharmacophore modelling and atom-based 3D-QSAR studies on N-methyl pyrimidones as HIV-1 integrase inhibitors. J Enzyme Inhib Med Chem 27:339–347
Renard JF, Julemont F, de Leval X, Pirotte B (2006) The use of nimesulide and its analogues in cancer chemoprevention. Anticancer Agents Med Chem 6:233–237
Rigas B, Kashfi K (2005) Cancer prevention: a new era beyond cyclooxygenase-2. J Pharmacol Exp Ther 314:1–8
Sano H, Kawahito Y, Wilder RL, Hashiramoto A, Mukai S, Asai K, Kimura S, Kato H, Kondo M, Hla T (1995) Expression of cyclooxygenase-1 and -2 in human colorectal cancer. Cancer Res 55:3785–3789
Sarkar FH, Adsule S, Li Y, Padhye S (2007) Back to the future: COX-2 inhibitors for chemoprevention and cancer therapy. Mini Rev Med Chem 7:599–608
Shattuck-Brandt RL, Varilek GW, Radhika A, Yang F, Washington MK, DuBois RN (2000) Cyclooxygenase 2 expression is increased in the stroma of colon carcinomas from IL-10(-/-) mice. Gastroenterology 118:337–345
Song X, Lin HP, Johnson AJ, Tseng PH, Yang YT, Kulp SK, Chen CS (2002) Cyclooxygenase-2, player or spectator in cyclooxygenase-2 inhibitor-induced apoptosis in prostate cancer cells. J Natl Cancer Inst 94:585–591
Srinath P, Rao PN, Knaus EE, Suresh MR (2003) Effect of cyclooxygenase-2 (COX- 2) inhibitors on prostate cancer cell proliferation. Anticancer Res 23:3923–3928
Su B, Chen S (2009) Lead optimization of COX-2 inhibitor nimesulide analogs to overcome aromatase inhibitor resistance in breast cancer cells. Bioorg Med Chem Lett 19:6733–6735
Su B, Diaz-Cruz ES, Landini S, Brueggemeier RW (2006) Novel sulfonanilide analogues suppress aromatase expression and activity in breast cancer cells independent of COX-2 inhibition. J Med Chem 49:1413–1419
Su B, Darby MV, Brueggemeier RW (2008) Synthesis and biological evaluation of novel sulfonanilide compounds as antiproliferative agents for breast cancer. J Comb Chem 10:475–483
Sun X, Li Y, Li W, Xu Z, Tang Y (2011) Computational investigation of interactions between human H2 receptor and its agonists. J Mol Graph Model 29:693–701
Sybyl-X 2.0. http://www.tripos.com. St. Louis, MO, USA
Telvekar VN, Chaudhari HK (2012) 3D-QSAR and docking-based combined in silico study on C-5 methyl substituted 4-arylthio and 4-aryloxy-3-iodopyridin-2-(1H)-one as HIV-1 RT inhibitors. Med Chem Res 21:2032–2043
Tian G, Yu JP, Luo HS, Yu BP, Yue H, Li JY, Mei Q (2002) Effect of nimesulide on proliferation and apoptosis of human hepatoma SMMC-7721 cells. World J Gastroenterol 8:483–487
Tong BJ, Tan J, Tajeda L, Das SK, Chapman JA, DuBois RN, Dey SK (2000) Heightened expression of cyclooxygenase-2 and peroxisome proliferatoractivated receptor-delta in human endometrial adenocarcinoma. Neoplasia 2:483–490
Tong Z, Wu X, Chen CS, Kehrer JP (2006) Cytotoxicity of a non-cyclooxygenase-2 inhibitory derivative of celecoxib in non-small-cell lung cancer A549 cells. Lung Cancer 52:117–124
Totzke G, Schulze-Osthoff K, Janicke RU (2003) Cyclooxygenase-2 (COX-2) inhibitors sensitize tumor cells specifically to death receptor-induced apoptosis independently of COX-2 inhibition. Oncogene 22:8021–8030
Tsujii M, Kawano S, Tsuji S, Sawaoka H, Hori M, DuBois RN (1998) Cyclooxygenase regulates angiogenesis induced by colon cancer cells. Cell 93:705–716
Williams CS, Tsujii M, Reese J, Dey SK, DuBois RN (2000) Host cyclooxygenase-2 modulates carcinoma growth. J Clin Invest 105:1589–1594
Yamamoto K, Kitayama W, Denda A, Morisaki A, Kuniyasu H, Kirita T (2003) Inhibitory effects of selective cyclooxygenase-2 inhibitors, nimesulide and etodolac, on the development of squamous cell dysplasias and carcinomas of the tongue in rats initiated with 4-nitroquinoline 1-oxide. Cancer Lett 199:121–129
Yamazaki R, Kusunoki N, Matsuzaki T, Hashimoto S, Kawai S (2002) Selective cyclooxygenase-2 inhibitors show a differential ability to inhibit proliferation and induce apoptosis of colon adenocarcinoma cells. FEBS Lett 531:278–284
Zhong Bo, Cai X, Yi X, Liu L, Pink JJ, Xu Y, Su B (2012) From COX-2 inhibitor nimesulide to potent anti-cancer agent: Syntesis, in vitro, in vivo and pharmacokinetic evaluation. Eur J Med Chem 47:432–444
Zhong B, Cai X, Yi X, Zhou A, Chen S, Su B (2011) In vitro and in vivo effects of a cyclooxygenase-2 inhibitor nimesulide analog JCC76 in aromatase inhibitors-insensitive breast cancer cells. J Steroid Biochem Mol Biol 126:10–18
Zhu J, Song X, Lin HP, Young DC, Yan S, Marquez VE, Chen CS (2002) Using cyclooxygenase-2 inhibitors as molecular platforms to develop a new class of apoptosis-inducing agents. J Natl Cancer Inst 94:1745–1757
Zhu J, Huang JW, Tseng PH, Yang YT, Fowble J, Shiau CW, Shaw YJ, Kulp SK, Chen CS (2004) From the cyclooxygenase-2 inhibitor celecoxib to a novel class of 3-phosphoinositide-dependent protein kinase-1 inhibitors. Cancer Res 64:4309–4318
Acknowledgments
ADC is thankful to Centre for Advanced Scientific Education and Research for usage of all computing facilities and Bioinformatics Centre, Assam University for other related resources.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Das Choudhury, A. Atom-based QSAR and 3D QSAR using pharmacophore based alignment for discovery of nimesulide-derived SKBR-3 cell line inhibitors. Med Chem Res 24, 999–1012 (2015). https://doi.org/10.1007/s00044-014-1187-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1187-6