Abstract
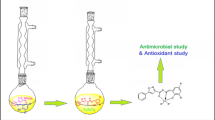
An efficient one pot chemoselective N-benzylation of 2-thiohydantoins and 2-thiobarbituric acids catalyzed by PEG-stabilized Ni nanoparticles has been reported, wherein the NH group of thiohydantoins and thiobarbituric acids is selectively benzylated and no S-benzylation is observed. Mildness and environment friendly approach of the protocol gives it an edge over other conventional methods. These compounds were tested in vitro for their antibacterial activity against Gram-positive bacteria namely, Staphylococcus aureus and Bacillus subtilis and their inhibitory action against two strains of fungus.





Similar content being viewed by others
References
Agasimundin YS, Mumper MW, Hasmane RS (1998) Inhibitors of glycogen phosphorylase B: synthesis, biochemical screening, and molecular modeling studies of novel analogues of hydantocidin. Bioorg Med Chem 6:911–923
Al- Burtamani SKS, Fatope MO, Marwah RG, Onifade AK, Al-Saidi SH (2005) Chemical composition, antibacterial and antifungal activities of the essential oil of Haplophyllum tuberculatum. J Ethnopharmocol 96:107–112
Aly YL, El-Barbary AA, El-Shehawya AA (2004) Alkylation of thiohydantoins including synthesis, conformational and configurational studies of some acetylated S-pyranosides. Phosphorus Sulfur Silicon Relat Elem 179:185–202
Aneja KR, Sharma C, Joshi R (2011) In vitro efficacy of amaltas (Cassia fistula L.) against the pathogens causing otitis externa. J Microbiol 4:175–183
Bojarski JT, Mokros JL, Barton HJ, Paluchowska MH (1985) Recent progress in barbituric acid chemistry. Adv Heterocycl Chem 38:229–297
Chui W-K, Wong T-H, Thenomozhiyal JC (2004) Anticonvulsant activity of phenylmethylenehydantoins: a structure-activity relation study. J Med Chem 47:1527–1535
Figueroa-Villar JD, Rangel CE, Dos Santos LN (1992) Synthesis of oxadeazaflavines from barbituric acid and aromatic aldehydes. Synth Commun 22:1159–1164
Frangin Y, Guimbal C, Wissocq F, Zamarlik H (1986) Synthesis of substituted barbituric acids via organozinc reagents. Synthesis 1986:1046–1050
Gadwood RC, Kamdar BV, Dubray LAC, Wolfe ML, Smith MP, Watt W, Mizsak SA, Groppi VE (1993) Synthesis and biological activity of spirocyclic benzopyran imidazolone potassium channel openers. J Med Chem 36:1480–1487
Gulati D, Chauhan PMS, Pratap R, Bhakuni DS (1994) A new synthesis of 5-bromoaplysinopsin, 6-bromoaplysinopsin and 3’-demethylaplysinopsin and their biological activities. Ind J Chem 33B:10–16
Handzlik J, Spengler G, Mastek B, Dela A, Molnar J, Amaral L, Kieć-Kononowicz K (2012) 5-Arylidene(thio)hydantoin derivatives as modulators of cancer efflux pump. Acta Poloniae Pharmaceutica Drug Research 69:149–153
He S, Kuang R, Venkataraman R, Tu J, Truong TM, Chan HT, Groutas WC (2000) Potent inhibition of serine proteases by heterocyclic sulfide derivatives of 1,2,5-thiadiazolidin-3-one-1,1-dioxide. Bioorg Med Chem 8:1713–1717
Kandra L, Remenyik J, Batta G, Somsák L, Gyémánt G, Park KH (2005) Enzymatic synthesis of a new inhibitor of α-amylases: acarviosinyl-isomaltosyl-spiro-thiohydantoin. Carbohydr Res 340:1311–1317
Khurana JM, Sneha, Vij K (2012) Ni nanoparticles: mild and efficient catalyst for the chemoselective synthesis of 2-arylbenzimidazoles, 2-arylbenzothiazoles and azomethines. Synth Commun 42:2606–2616
Khurana JM, Vij K (2010) Nickel nanoparticles catalyzed knoevenagel condensation of aromatic aldehydes with barbituric acids and 2-thiobarbituric acids. Catal Lett 138:104–110
Khurana JM, Vij K (2011) Nickel nanoparticles catalyzed chemoselective knoevenagel condensation of meldrum’s acid and tandem enol lactonizations via cascade cyclization sequence. Tetrahedron Lett 52:3666–3669
Kieć-Kononowicz K, Szymańska E (2002) Antimycobacterial activity of 5-arylidene derivatives of hydantoin. Farmaco 57:909–916
Mizuno T, Kino T, Ito T, Miyata T (2000) Synthesis of aromatic urea herbicides by the selenium-assisted carbonylation using carbon monoxide with sulfur. Synth Commun 30:1675–1688
Muccioli GG, Fazio N, Scriba GKE, Poppitz W, Cannata F, Poupaert JH, Wouters J, Lambert DM (2006) Substituted 2-thioxoimidazolidin-4-ones and imidazolidine-2,4-diones as fatty acid amide hydrolase inhibitors templates. J Med Chem 49:417–425
Ono M, Hayashi S, Matsumura K, Kimura H, Okamoto Y, Ihara M, Takahashi R, Mori H, Saji H (2011) Rhodanine and thiohydantoin derivatives for detecting tau pathology in alzheimer’s brains. ACS Chem Neurosci 2:269–275
Shih RU, Wu J, Liu Y, Liang YC, Lin SY, Sheu MT, Lee WS (2004) Anti-proliferation effect of 5,5-diphenyl-2-thiohydantoin (DPTH) in human vascular endothelial cells. Biochem Pharmacol 67:67–75
Spengler G, Evaristo M, Handzlik J, Serly J, Moln·r J, Viveiros M, KieÊ-Kononowicz K, Amaral L (2010) Biological activity of hydantoin derivatives on P-glycoprotein (ABCB1) of mouse lymphoma cells. Anticancer Res 30:4867–4871
Szymańska E, Kieć-Kononowicz K, Białecka A, Kasprowicz A (2002) Antimicrobial activity of 5- arylidene aromatic derivatives of hydantoin. Farmaco 57:39–44
Takahashi A, Matsuoka H, Yamada K, Uda Y (2005) Characterization of antimutagenic mechanism of 3-allyl-5-substituted 2-thiohydantoins against 2-amino-3-methylimidazo[4,5-f]quinoline. Food Chem Toxicol 43:521–528
Tanaka K, Cheng X, Yoneda F (1988) Oxidation of thiol with 5arylidene-1,3-dimethylbarbituric acid: application to synthesis of unsymmetrical disulfide. Tetrahedron 44:3241–3249
Tompkin JE (1986) 5,5-Diaryl-2-thiohydantoins and 5,5-diaryl-N3-substituted-2-thiohydantoins as potential hypolipidemic agents. J Med Chem 29:855–859
Yan Q, Cao R, Yi W, Chen Z, Wen H, Ma L, Song H (2009) Inhibitory effects of 5-benzylidene barbiturate derivatives on mushroom tyrosinase and their antibacterial activities. Eur J Med Chem 44:4235–4243
Acknowledgments
Sneha is thankful to CSIR, New Delhi, India for the award of Junior and Senior Research Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sneha, Khurana, J.M., Sharma, C. et al. Chemoselective N-benzylation of 2-thiohydantoins and 2-thiobarbituric acids catalyzed by PEG-stabilized Ni nanoparticles and their anti-microbial activities. Med Chem Res 23, 4595–4606 (2014). https://doi.org/10.1007/s00044-014-1008-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-1008-y