Abstract
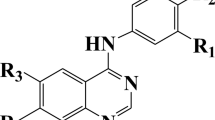
Two quantitative structure–activity relationship (QSAR) models, multiple linear regression (MLR) and radial basis function neural network (RBFNN), have been developed for predicting agonist activity of a series of potent and selective bombesin receptor subtype-3 (BRS-3) containing a biarylethylimidazole pharmacophore by performing density functional theory calculations at the B3LYP/6-311G(d,p) level. The investigated results have demonstrated that the excitation activity of investigated compounds can be reflected by quantum descriptors such as hardness, the total dipole moment, electrophilicity, the highest occupied molecular orbital energy (E HOMO), and the lowest unoccupied molecular orbital energy (E LUMO). The results showed that the pEC50 values calculated by RBFNN model are in good agreement with the experimental data, and the performance of the RBFNN regression model is superior to the MLR-based model. The developed RBFNN model was applied for the prediction of the biological activities of biarylethylimidazole derivatives, which were not in the modeling procedure. The resulted model showed high prediction ability with root mean square error of prediction of 0.224 for RBFNN. Therefore, the QSAR models based on quantum descriptors are reliable in predicting BRS-3 agonist activity for unknown biarylethylimidazole derivatives.





Similar content being viewed by others
References
Atkinson AC (1985a) Plots, transformations and regression. Clarendon Press, Oxford
Atkinson AC (1985b) Plots, transformations, and regression: an introduction to graphical methods of diagnostic regression analysis. Clarendon Press, Oxford
Becke AD (1993) Density-functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648
Chattaraj PK, Sarkar U, Roy DR (2006) Electrophilicity index. Chem Rev 106(6):2065–2091
El Ashry ESH, El Nemr A, Esawy SA, Ragab S (2006) Corrosion inhibitors: Part II: Quantum chemical studies on the corrosion inhibitions of steel in acidic medium by some triazole, oxadiazole and thiadiazole derivatives. Electrochim Acta 51(19):3957–3968
El-Deredy W, Ashmore SM, Branston NM, Darling JL, Williams SR, Thomas DGT (1997) Pretreatment prediction of the chemotherapeutic response of human glioma cell cultures using nuclear magnetic resonance spectroscopy and artificial neural networks. Cancer Res 57(19):4196
Fassihi A, Shahlaei M, Moeinifard B, Sabet R (2012) QSAR study of anthranilic acid sulfonamides as methionine aminopeptidase-2 inhibitors. Chem Mon 143(2):189–198
Frisch M, Trucks G, Schlegel H, Scuseria G, Robb M, Cheeseman J, Montgomery J, Vreven T, Kudin K, Burant J (2008) Gaussian 03, revision C.02. Gaussian Inc., Pittsburgh
Golbraikh A, Tropsha A (2002) Beware of q2! J Mol Graph Model 20(4):269
Gramatica P, Pilutti P, Papa E (2004) Validated QSAR prediction of OH tropospheric degradation of VOCs: splitting into training-test sets and consensus modeling. J Chem Inf Comput Sci 44(5):1794–1802
Guan XM, Chen H, Dobbelaar PH, Dong Y, Fong TM, Gagen K, Gorski J, He S, Howard AD, Jian T (2010) Regulation of energy homeostasis by bombesin receptor subtype-3: selective receptor agonists for the treatment of obesity. Cell Metab 11(2):101–112
Hadden M, Goodman A, Guo C, Guzzo PR, Henderson AJ, Pattamana K, Ruenz M, Sargent BJ, Swenson B, Yet L (2010) Synthesis and SAR of heterocyclic carboxylic acid isosteres based on 2-biarylethylimidazole as bombesin receptor subtype-3 (BRS-3) agonists for the treatment of obesity. Bioorg Med Chem Lett 20(9):2912–2915
Haykin SP (1999) Neural networks: a comprehensive foundation, 2nd edn. Hall PTR, Upper Saddle River
Hohenberg P, Kohn W (1964) Inhomogeneous electron gas. Phys Rev 136(3B):B864
Howlett RJ, Jain LC (2001) Radial basis function networks 1: recent developments in theory and applications, vol 1. Physica-Verlag HD, Heidelberg
Jennings C, Harrison D, Maycox P, Crook B, Smart D, Hervieu G (2003) The distribution of the orphan bombesin receptor subtype-3 in the rat CNS. Neuroscience 120(2):309–324
Karelson M, Lobanov VS, Katritzky AR (1996) Quantum-chemical descriptors in QSAR/QSPR studies. Chem Rev 96(3):1027–1044
Katritzky AR, Dobchev DA, Fara DC, Karelson M (2005) QSAR studies on 1-phenylbenzimidazoles as inhibitors of the platelet-derived growth factor. Bioorg Med Chem 13(24):6598–6608
Kennard R, Stone L (1969) Computer aided design of experiments. Technometrics 11(1):137–148
Kohn W, Sham LJ (1965) Self-consistent equations including exchange and correlation effects. Phys Rev 140:A1133
Liu H, Papa E, Gramatica P (2008) Evaluation and QSAR modeling on multiple endpoints of estrogen activity based on different bioassays. Chemosphere 70(10):1889–1897
Liu J, He S, Jian T, Dobbelaar PH, Sebhat IK, Lin LS, Goodman A, Guo C, Guzzo PR, Hadden M (2010) Synthesis and SAR of derivatives based on 2-biarylethylimidazole as bombesin receptor subtype-3 (BRS-3) agonists for the treatment of obesity. Bioorg Med Chem Lett 20(7):2074–2077
Matsumoto K, Yamada K, Wada E, Hasegawa T, Usui Y, Wada K (2003) Bombesin receptor subtype-3 modulates plasma insulin concentration. Peptides 24(1):83–90
Moody TW, Sancho V, di Florio A, Nuche-Berenguer B, Mantey S, Jensen RT (2011) Bombesin receptor subtype-3 agonists stimulate the growth of lung cancer cells and increase EGF receptor tyrosine phosphorylation. Peptides 32:1677–1684
Ohki-Hamazaki H, Watase K, Yamamoto K, Ogura H, Yamano M, Yamada K, Maeno H, Imaki J, Kikuyama S, Wada E (1997) Mice lacking bombesin receptor subtype-3 develop metabolic defects and obesity. Nature 390(6656):165–169
Qin Y, Deng H, Yan H, Zhong R (2011) An accurate nonlinear QSAR model for the antitumor activities of chloroethylnitrosoureas using neural networks. J Mol Graph Model 29:826–833
Shahlaei M (2013) Descriptor selection methods in quantitative structure–activity relationship studies: a review study. Chem Rev 113:8093–8103
Shahlaei M, Fassihi A (2013) QSAR analysis of some 1-(3,3-diphenylpropyl)-piperidinyl amides and ureas as CCR5 inhibitors using genetic algorithm-least square support vector machine. Med Chem Res 22:4384–4400
Shahlaei M, Pourhossein A (2013) Modeling of CCR5 antagonists as anti HIV agents using combined genetic algorithm and adaptive neuro-fuzzy inference system (GA–ANFIS). Med Chem Res 22:4423–4436
Shahlaei M, Madadkar-Sobhani A, Fassihi A, Saghaie L, Arkan E (2012a) QSAR study of some CCR5 antagonists as anti-HIV agents using radial basis function neural network and general regression neural network on the basis of principal components. Med Chem Res 21(10):3246–3262
Shahlaei M, Madadkar-Sobhani A, Fassihi A, Saghaie L, Shamshirian D, Sakhi H (2012b) Comparative quantitative structure–activity relationship study of some 1-aminocyclopentyl-3-carboxyamides as CCR2 inhibitors using stepwise MLR, FA-MLR, and GA-PLS. Med Chem Res 21(1):100–115
Shahlaei M, Fassihi A, Saghaie L, Arkan E, Madadkar-Sobhani A, Pourhossein A (2013a) Computational evaluation of some indenopyrazole derivatives as anticancer compounds; application of QSAR and docking methodologies. J Enzyme Inhib Med Chem 28(1):16–32
Shahlaei M, Fassihi A, Pourhossein A, Arkan E (2013b) Statistically validated QSAR study of some antagonists of the human CCR5 receptor using least square support vector machine based on the genetic algorithm and factor analysis. Med Chem Res 22(3):1399–1414
Tan N, Rao H, Li Z, Li X (2009) Prediction of chemical carcinogenicity by machine learning approaches. SAR QSAR Environ Res 20(1–2):27–75
Tropsha A, Gramatica P, Gombar V (2003) The importance of being earnest: validation is the absolute essential for successful application and interpretation of QSPR models. QSAR Comb Sci 22(1):69–77
Xiang Y, Liu M, Zhang X, Zhang R, Hu Z, Fan B, Doucet J, Panaye A (2002) Quantitative prediction of liquid chromatography retention of N-benzylideneanilines based on quantum chemical parameters and radial basis function neural network. J Chem Inf Comput Sci 42(3):592–597
Zheng S, Li Z, Wang H (2011) A genetic fuzzy radial basis function neural network for structural health monitoring of composite laminated beams. Expert Syst Appl 38(9):11837–11842
Zupan J, Gasteiger J (1993) Neural networks for chemists. VCH, Weinheim
Acknowledgments
We gratefully acknowledge Vice Chancellor for Research and Technology, Kermanshah University of Medical Sciences for financial support. This article resulted from the Pharm.D thesis of Amin Nowroozi, major of Pharmacy, Kermanshah University of Medical Sciences, Kermanshah, Iran.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shahlaei, M., Nowroozi, A. & Khodarahmi, R. Application of radial basis function neural network and DFT quantum mechanical calculations for the prediction of the activity of 2-biarylethylimidazole derivatives as bombesin receptor subtype-3 (BRS-3) agonists. Med Chem Res 23, 3681–3693 (2014). https://doi.org/10.1007/s00044-014-0948-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-014-0948-6