Abstract
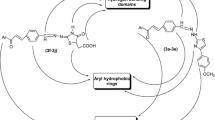
A series of aryl semicarbazones containing a methylene bridge in their skeleton were synthesized as anticonvulsant agents. The strategy of introducing a methylene bridge was to increase the flexibility of the structures because of sp3 hybridization. Pharmacological evaluations of the compounds were performed by determination of their effects on pentylenetetrazole-induced seizure parameters and neurotoxicity in mice. The statistical analysis indicated that most of the synthesized compounds showed significant anticonvulsant activity in comparison with the control group. No remarkable neurotoxicity was observed in rotorod test. A QSAR study was performed using multiple linear regressions. The results of the QSAR study confirmed that the compounds with Br at para position of these new derivatives are more potent than para-ethoxy series.





Similar content being viewed by others
References
Abdel-Aziz M, Abuo-Rahma A, Gamal El-Din A, Hassan AA (2009) Synthesis of novel pyrazole derivatives and evaluation of their antidepressant and anticonvulsant activities. Eur J Med Chem 44:3480–3487
Afiuni-Zadeh S, Azimi G (2010) A QSAR study for modeling of 8-azaadenine analogues proposed as A1 adenosine receptor antagonists using genetic algorithm coupling adaptive Neuro-Fuzzy inference system (ANFIS). Anal Sci 26:897–902
Amin KM, Rahman DEA, Al-Eryani YA (2008) Synthesis and preliminary evaluation of some substituted coumarins as anticonvulsant agents. Bioorg Med Chem 16:5377–5388
Azam F, Alkskas IA, Khokra SL, Prakash O (2009) Synthesis of some novel N4-(naphtha[1,2-d]thiazol-2-yl)semicarbazides as potential anticonvulsants. Eur J Med Chem 44:203–211
Azam F, El-gnidi BA, Alkskas IA (2010) Combating oxidative stress in epilepsy: design, synthesis, quantum chemical studies and anticonvulsant evaluation of 1-(substituted benzylidene/ethylidene)-4-(naphthalen-1-yl)semicarbazides. Eur J Med Chem 45:2817–2826
Bölcskei H, Tarnawa I, Kocsis P (2008) Voltage-gated sodium channel blockers, 2001–2006: an overview. Med Chem Res 17:356–368
Campagna F, Carotti A, Casini G, Palluotto F, Genchi G, De Sarro GB (1993) 2-Aryl-2,5-dihydropyridazino[4,3-b]lindol-3(3H)-ones: novel rigid planar benzodiazepine receptor ligands. Bioorg Med Chem 1:437–446
Dimmock JR, Vashishtha SC, Stables JP (2000) Anticonvulsant properties of various acetylhydrazones, oxamoylhydrazones and semicarbazones derived from aromatic and unsaturated carbonyl compounds. Eur J Med Chem 35:241–248
Dumciute J, Martynaitis V, Holzer W, Mangelinckx S, Kimpe ND, Sackus A (2006) Synthesis and ring transformations of 1-amino-1,2,3,9a-tetrahydroimidazo[1,2-a]indol-2(9H)-ones. Tetrahedron 62:3309–3319
Gholizadeh S, Shafaroodi H, Ghasemi M, Bahremand A, Sharifzadeh M, Dehpour AR (2007) Ultra-low dose cannabinoid antagonist AM251 enhances cannabinoid anticonvulsant effects in the pentylenetetrazole-induced seizure in mice. Neuropharmacology 53:763–770
Hosseini-Zare MS, Salehi F, Seyedi SY, Azami K, Ghadiri T, Mobasseri M, Gholizadeh S, Beyer C, Sharifzadeh M (2011) Effects of pentoxifylline and H-89 on epileptogenic activity of bucladesine in pentylenetetrazol-treated mice. Eur J Pharmacol 670:464–670
Jalali-Heravi M, Asadollahi-Baboli M (2009) Quantitative structure-activity relationship study of serotonin (5-HT7) receptor inhibitors using modified ant colony algorithm and adaptive neuro-fuzzy interference system (ANFIS). Eur J Med Chem 44:1463–1470
Kaushik D, Khan SA, Chawla G, Kumar S (2010) N′-[(5-chloro-3-methyl-1-phenyl-1H-pyrazol-4-yl)methylene] 2/4-substituted hydrazides: synthesis and anticonvulsant activity. Eur J Med Chem 45:3943–3949
Kennedy GM, Lhatoo SD (2008) CNS adverse events associated with antiepileptic drugs. CNS Drugs 22:739–760
Kumar M, Thurow K, Stoll N, Stoll R (2007) Robust fuzzy mappings for QSAR studies. Eur J Med Chem 42:675–685
Narayana B, Vijaya Raj KK, Ashalatha BV, Kumari NS (2006) Synthesis of some new substituted triazolo [4,3-a][1,4] benzodiazepine derivatives as potent anticonvulsants. Eur J Med Chem 41:417–422
Pandeya SN, Aggarwal N, Jain JS (1999) Evaluation of semicarbazones for anticonvulsant and sedative hypnotic properties. Pharmazie 54:300–302
Pandeya SN, Yogeeswari P, Stables JP (2000) Synthesis and anticonvulsant activity of 4-bromophenyl substituted aryl semicarbazones. Eur J Med Chem 35:879–886
Perucca E (2005) An introduction to antiepileptic drugs. Epilepsia 46(4):31–37
Rajak H, Deshmukh R, Veerasamy R, Sharma AK, Mishra P, Kharya MD (2010) Novel semicarbazones based 2,5-disubstituted-1,3,4-oxadiazoles: one more step towards establishing four binding site pharmacophoric model hypothesis for anticonvulsant activity. Bioorg Med Chem 20:4168–4172
Sander JW (2003) The epidemiology of epilepsy revisited. Curr Opin Neurol 16:165–170
Shafaroodi H, Samini M, Moezi L, Homayoun H, Sadeghipour H, Tavakoli S, Hajrasouliha AR, Dehpour AR (2004) The interaction of cannabinoids and opioids on pentylenetetrazole-induced seizure threshold in mice. Neuropharmacology 47:390–400
Shafiee A, Rineh A, Kebriaeezadeh A, Foroumadi A, Sheibani V, Afarinesh MR (2009) Synthesis and anticonvulsant activity of 4-(2-phenoxyphenyl)semicarbazones. Med Chem Res 18:758–769
Singh HP, Pandeya SN, Chauhan CS, Sharma CS (2011) Synthesis and pharmacological screening of some novel chalconyl derivatives of substituted phenyl semicarbazide. Med Chem Res 20:74–80
Thirumurugan R, Sriram D, Saxena A, Stables J, Yogeeswari P (2006) 2,4-Dimethoxyphenylsemicarbazones with anticonvulsant activity against three animal models of seizures: synthesis and pharmacological evaluation. Bioorg Med Chem 14:3106–3112
Treiman DM (2001) GABAergic mechanisms in epilepsy. Epilepsia 42(3):8–12
Wagle S, Adhikari AV, Kumari NS (2009) Synthesis of some new 4-styryltetrazolo[1,5-a]quinoxaline and 1-substituted-4-styryl[1,2,4]triazolo[4,3-a]quinoxaline derivatives as potent anticonvulsants. Eur J Med Chem 44:1135–1143
Yogeeswari P, Sriram D, Brahmandam A, Sridharan I, Thirumurugan R, Stables JP (2003) Synthesis of novel aryl semicarbazones as anticonvulsants with GABA-mediated mechanism. Med Chem Res 12:57–68
Yogeeswari P, Thirumurugan R, Kavya R, Samuel JS, Stables J, Sriram D (2004) 3-Chloro-2-methylphenyl-substituted semicarbazones: synthesis and anticonvulsant activity. Eur J Med Chem 39:729–734
Yogeeswari P, Sriram D, Thirumurugan R, Raghavendran JV, Sudhan K, Pavana RK, Stables J (2005a) Discovery of N-(2,6-dimethylphenyl)-substituted semicarbazones as anticonvulsants: hybrid pharmacophore-based design. J Med Chem 48:6202–6211
Yogeeswari P, Sriram D, Veena V, Kavya R, Rakhra K, Ragavendran JV, Mehta S, Thirumurugan R, Stables JP (2005b) Synthesis of aryl semicarbazones as potential anticonvulsant agents. Biomed Pharmacother 59:51–55
Yogeeswari P, Sriram D, Thirumurugan R, Jit LRJS, Ragavendran JV, Kavya R, Rakhra K, Saraswat V (2006) Synthesis of N4-(2,4-dimethylphenyl) semicarbazones as 4-aminobutyrate aminotransferase inhibitors. Acta Pharm 56:259–572
Acknowledgments
This study was supported by grants from Pharmaceutical Sciences Research Center, Tehran University of Medical Sciences and Kerman Neuroscience Research Center, Kerman University of Medical Sciences. We thank Ms. Marjan Jalili Baleh and Ali Kazemi for their technical assistance.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mozaffari, S., Ghasemi, S., Baher, H. et al. Synthesis and evaluation of some novel methylene-bridged aryl semicarbazones as potential anticonvulsant agents. Med Chem Res 21, 3797–3808 (2012). https://doi.org/10.1007/s00044-011-9924-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-011-9924-6