Abstract
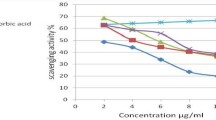
Chalcones featuring an analgesic/anti-inflammatory pharmacophore, i.e., the 2(3H)-benzoxazolone heterocycle, on the one hand, and a radical scavenger moiety, i.e., 2,6-di-t-butylphenol, on the other hand were synthesized by condensation of a ketone 2(3H)-benzoxazolone precursor with 3,5-di-t-butyl-4-hydroxybenzaldehyde. Among the various methods explored (acid homogenous or heterogenous catalysis, base catalysis), heterogenous catalysis conditions using KSF Montmorillonite were found to be the most convenient. The E-geometry of the so-obtained chalcones was ascertained both by 1H and 13C-nuclear magnetic resonance (NMR) spectroscopy as well as B3LYP/6-31G** quantum mechanics calculations. Chalcones 1–8 were pharmacologically evaluated in vitro for their ability to prevent human low-density lipoprotein (LDL) copper-induced oxidation using Cu2+ as oxidizing agent. Compound 4 emerged as the most promising agent as it was able to inhibit copper-mediated human LDL oxidation with an activity ten times greater than that of Probucol, a reference antioxidant drug.



Similar content being viewed by others
References
Aichaoui H, Poupaert JH, Lesieur D, Henichart JP (1992) Aluminium trichloride-DMF reagent in the Friedel-Crafts reaction. Application to 2(3H) benzoxazolones in the Haworth reaction. Bull Soc Chim Belg 101(12):1053–1060
Ballini R, Bosica G, Ricciutelli M, Maggi R, Sartori G, Sartorio R, Righi P (2001) Clay-catalyzed solvent less synthesis of trans-chalcones. Green Chem 3(4):178–180. doi:10.1039/b101355f
Becke AD (1988) Density-functional exchange-energy approximation with correct asymptotic behavior. Phys Rev A At Mol Opt Phys 38(6):3098–3100
Blanc-Delmas E, Lebegue N, Wallez V, Leclerc V, Yous S, Carato P, Farce A, Bennejean C, Renard P, Caignard DH, Audinot-Bouchez V, Chomarat P, Boutin J, Hennuyer N, Louche K, Carmona MC, Staels B, Penicaud L, Casteilla L, Lonchampt M, Dacquet Chavatte Ph, Berthelot P, Lesieur D (2006) Novel 1,3-dicarbonyl compounds having 2(3H)-benzoxazolonic heterocycles as PPARγ agonists. Bioorg Med Chem 14(22):7377–7391. doi:10.1016/j.bmc.2006.07.029
Climent MJ, Corma A, Iborra S, Velty A (2004) Activated hydrotalcites as catalysts for the synthesis of chalcones of pharmaceutical interest. J Catal 221(2):474–482. doi:10.1016/j.jcat.2003.09.012
Depreux P, Aichaoui H, Lesieur I (1993) Synthesis of indanyl analogs of 3-[3-(4-arylpiperazin-1-yl) propyl]-2-oxo-2,3-dihydro [1,3]benzoxazoles. Heterocycl 36(5):1051–1058
Ilieva S, Atanasov Y, Kalcheva V, Galabov B (2003) Computational study of the general base catalyzed aminolysis of 2-benzoxazolinone. J Mol Struct THEOCHEM 633(1):49–55. doi:10.1016/S0166-1280(03)00310-5
Lebeau J, Furman Ch, Bernier JL, Duriez P, Teissier E, Cotelle N (2000) Anti-oxidant properties of di-tert-butylhydroxylated flavonoids. Free Rad Biol Med 29:900–912
Leyrer J, Zaki MI, Knoezinger H (1986) Solid/solid interactions. Monolayer formation in molybdenum trioxide-alumina physical mixtures. J Phys Chem 90(20):4775–4780. doi:10.1021/j100411a013
Lutz RE, Jordan RH (1950) cis-Benzalacetophenone. J Am Chem Soc 72:4090. doi:10.1021/ja01165a069
Orhan H, Dogruer DS, Cakir B, Sahin G, Sahin MF (1999) The in vitro effects of new non-steroidal antiinflammatory compounds on antioxidant system of human erythrocytes. Experimental and toxicologic pathology: official journal of the Ges. Toxicol Pathol 51(4–5):397–402
Pilli G, Erdogan H, Sunal R (1993) Some new benzoxazolinone derivatives with analgesic and anti-inflammatory activities. Arz-For 43:1351–1354
Poupaert JH, Carato P, Colacino E (2005) 2(3H)-Benzoxazolone and bioisosters as “privileged scaffold” in the design of pharmacological probes. Curr Med Chem 12:877–885. doi:10.2174/0929867053507388
Rendy R, Zhang Y, McElrea A, Gomez A, Klumff DA (2004) Superacid-catalyzed reactions of cinnamic acids and the role of superelectrophiles. J Org Chem 69(7):2340–2347. doi:10.1021/jo030327t
Safak C, Erdogan H, Palaska E, Rumeysa S, Suna D (1992) Synthesis of 3-(2-pyridylethyl)benzoxazolinone derivatives : potent analgesic and antiinflammatory compounds inhibiting prostaglandin E2. J Med Chem (35):1296–1299. doi:10.1021/jm00085a018
Sgaragli GP, Valoti M, Gorelli B, Fusi F, Palmi M, Mantovani P (1993) Calcium antagonist and antiperoxidant properties of some hindered phenols. Br J Pharm 110(1):369–377
Stella L, Janousek Z, Merenyi R, Viehe HG (1978) Stabilization of radicals by capto-dative substitution—carbon-carbon addition to radicophilic olefins. Angew Chem 90(9):741–742. doi:10.1002/ange.19780900934
Vosko SH, Wilk L, Nusair M (1980) Accurate spin-dependent electron liquid correlation energies for local spin density calculations: a critical analysis. Can J Phys 58(8):1200–1211
Yous S, Poupaert JH, Chavatte Ph, Bizot Espiard Jean-Guy, Caignard D-H, Lesieur D (2001) Synthesis and pharmacological evaluation of analgesic 6-substituted 2(3H)-benzothiazolones. Drug Des Disc 17(4):331–336
Yous S, Lebegue N, Poupaert JH, Chavatte Ph, Berthelot P (2005) Lipid-lowering properties of 6-benzoyl-2(3H)-benzothiazolone and structurally related compounds. J Enz Inhib Med Chem 20(6):525–532
Acknowledgement
This work was supported by a research grand from the FNRS (FRSM 3.4.625.07 and FRFC 2.4.654.06).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Aichaoui, H., Guenadil, F., Kapanda, C.N. et al. Synthesis and pharmacological evaluation of antioxidant chalcone derivatives of 2(3H)-benzoxazolones. Med Chem Res 18, 467–476 (2009). https://doi.org/10.1007/s00044-008-9143-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-008-9143-y