Abstract
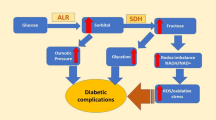
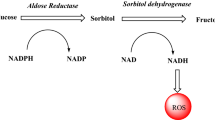
Aldose reductase (AR) is an enzyme that catalyzes the conversion of glucose to sorbitol, which is in turn converted to fructose by sorbitol dehydrogenase. The increased glucose flux through this metabolic pathway has been linked to the development of diabetic complications such as neuropathy, nephropathy, retinopathy, and cataract. Inhibitors of AR thus seem to have the potential to prevent or treat diabetic complications. AR inhibitors belong to different chemical classes, one of which comprises pyridazinone analogues. At present, however, side effects and/or insufficient pharmacokinetic profiles have made most of the drug candidates undesirable. We evaluated a series of 2H-pyridazine-3-one and 6-chloropyridazine analogues via an in vitro spectrophotometric assay for their ability to inhibit rat kidney AR. The study showed that the introduction of a pyrazole ring on pyridazinone led to a marked decrease in AR inhibitory potency. Moreover, introduction of an acetic acid side chain on 2H-pyridazine-3-one and 6-chloropyridazine did not improve the AR inhibitory activity, which was an unexpected result. On the basis of preliminary AR inhibitory screening results on 2H-pyridazine-3-one and 6-chloropyridazine derivatives, we embarked on the synthesis of more derivatives to discover more active molecules.



Similar content being viewed by others
References
Banavara LM (2005) Pyridazinone aldose reductase inhibitors. US Patent 6849629 issued on February 1, 2005
Banoğlu E, Akoğlu C, Ünlü S, Küpeli E, Yeşilada E, Şahin MF (2004) Amide derivatives of [6-(5-methyl-3-phenylpyrazol-1-yl)-3(2H)-pyrazinone-2-yl]acetic acids as potential analgesic and anti-inflammatory compounds. Arch Pharm Pharm Med Chem 337:7–14
Banoğlu E, Akoğlu C, Ünlü S, Çalışkan-Ergun B, Küpeli E, Yeşilada E, Şahin MF (2005) Synthesis of amide derivatives of [6-(3,5-dimethylpyrazol-1-yl)-3(2H)-pyrazinone-2-yl]acetic acid and their analgesic and anti-inflammatory properties. Arzheim Forsch Drug Res 55:520–527
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Buyukbingol E, Suzen S, Klopman G (1994) Studies on the synthesis and structure-activity relationships of 5-(3′-indolyl)-2-thiohydantoin derivatives as aldose reductase enzyme ınhibitors. Il Farmaco 49:443–447
Cerelli KJ, Curtis DL, Dunn PH, Nelson PH, Peak TM, Waterbury LD (1986) Antiinflammatory and aldose reductase inhibitory activity of some tricyclic arylacetic acids. J Med Chem 29:2347–2351
Constantino L, Rastelli G, Vescovivni K, Cignarella G, Vianello P, Del Corso A, Cappiello M, Mura U, Barlocco D (1996) Synthesis, activity and molecular modeling of a new series of tricyclic pyridazinones as selective aldose reductase inhibitors. J Med Chem 39:4396–4405
Constantino L, Rastelli G, Gamberini MC, Giovannoni MP, Dal Piaz V, Vianello P, Barlocco D (1999) Isoxazolo-[3,4-d]-pyridazin-7-(6H)-one as a potential substrate for new aldose reductase inhibitors. Med Res Rev 19:3–23
Courdet P, Doroux E, Bastide P, Couquelet JD, Tronche P (1991) Synthesis and evaluation of the aldose reductase inhibitory activity of new diaryl pyridazine-3-ones. J Pharm Belg 46:375–380
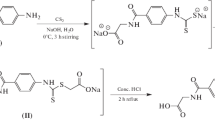
Courdet P, Rubat C, Doroux E, Bastide P, Couquelet JD, Tronche P, Albuisson E (1992) Synthesis and aldose reductase inhibitory activity of pyridazine derivatives possessing acetic acid group. Il Farmaco 47:37–46
Guo Z, Su W, Allen S, Pang H, Daugherty A, Smart E, Gong MC (2005) COX-2 Up-regulation and vascular smooth muscle contractile hyperreactivity in spontaneous diabetic db/db mice. Cadiovasc Res 67:723–735
Klebe G, Kramer O, Sotriffer C (2004) Strategies for the design of inhibitors of aldose reductase, an enzyme showing pronounced induced-fit adaptations. Cell Mol Life Sci 61:783–793
Lee SH, Woo HG, Baik EJ, Moon CH (2000) High glucose enhances IL-1beta-induced cyclooxygenase-2 expression in rat vascular smooth muscle cells. Life Sci 68:57–67
Maccari R, Ottana R, Curing C, Vigorita MG, Rakowitz D, Steindl T, Langer T (2005) Structure-activity relationships and molecular modelling of 5-arylidene-2,4-thiazolidinediones active as aldose reductase inhibitors. Bioorg Med Chem 13:2809–2823
Mylari B, Armento SJ, Beebe DA, Conn EL, Coutcher JB, Dina BS (2003) A highly selective, non-hydantoin, non carboxylic acid inhibitor of aldose reductase with potent oral activity in diabetic rat models. J Med Chem 46:2283–2286
Pau A, Asproni B, Boatto G, Grella GE, Caprariis P De, Costantino L, Pinna GA (2004) Synthesis and aldose reductase inhibitory activities of novel thienocinnolinone derivatives. Pharm Sci 21:545–552
Sarges R, Oates PJ (1993) Aldose reductase inhibitors: Recent developments. Prog Drug Res 40:99–156
Steuber H, Zentgraf M, Podjarny A, Heine A, Klebe G (2006) High resolution crystal structure of aldose reductase complexed with the novel sulfonyl pyridazinone inhibitor exhibiting an alternative active site anchoring group. J Mol Biol 356:45–561
Şüküroğlu M, Çalışkan-Ergun B, Ünlü S, Şahin MF, Küpeli E, Yeşilada E, Banoğlu E, Akoğlu C (2005) Synthesis, analgesic and anti-inflammatory activities of [6-(3,5-dimethyl-4-pyrazol-1-yl)-3(2H)-pyrazinone-2-yl]acetamides. Arch Pharm Res 28:509–517
Suzen S, Buyukbingol E (2003) Recent studies of aldose reductase enzyme inhibition for diabetic complications. Curr Med Chem 10:1329–1352
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Şüküroğlu, M., Çalişkan-Ergün, B., Das-Evcimen, N. et al. Screening and evaluation of rat kidney aldose reductase inhibitory activity of some pyridazine derivatives. Med Chem Res 15, 443–451 (2007). https://doi.org/10.1007/s00044-006-0021-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00044-006-0021-1