Abstract
Objectives
This study assesses the risk of exposure to hazardous chemical residues in pork meat, liver, and kidney collected at wet markets in Nghe An and Hung Yen provinces and discusses health impact implication.
Methods
514 pig feed, kidney, liver, and pork samples were pooled and qualitatively and quantitatively analyzed for tetracyclines, fluoroquinolones, sulphonamide, chloramphenicol, β-agonists, and heavy metals. We compare the results with current regulations on chemical residues and discuss health implications.
Results
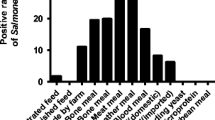
Legal antibiotics were found in feed. Tetracycline and fluoroquinolones were not present in pork, but 11% samples were positive with sulfamethazine above maximum residue limits (MRL); 11% of packaged feed and 4% of pork pooled samples were positive for chloramphenicol, a banned substance; two feed, two liver, and one pork samples were positive for β-agonists but did not exceed current MRL; 28% of pooled samples had lead, but all were below MRL; and all samples were negative for cadmium and arsenic. Thus, the health risks due to chemical hazards in pork in Hung Yen and Nghe An seemed not as serious as what were recently communicated to the public on the mass media.
Conclusions
There is potential exposure to sulphonamide, chloramphenicol, and β-agonists from pork. Risk communication needs to focus on banned chemicals, while informing the public about the minimal risks associated with heavy metals.


Similar content being viewed by others
References
Andrée S, Jira W, Schwind KH, Wagner H, Schwägele F (2010) Chemical safety of meat and meat products. Meat Sci 86(1):38–48. doi:10.1016/j.meatsci.2010.04.020
Baynes RE, Dedonder K, Kissell L, Mzyk D, Marmulak T, Smith G, Riviere JE (2016) Health concerns and management of select veterinary drug residues. Food Chem Toxicol 88:112–122. doi:10.1016/j.fct.2015.12.020
Beyene T (2016) Veterinary drug residues in food-animal products: its risk factors and potential effects on public health. J Veterinar Sci Technol 7(1):285. doi:10.4172/2157-7579.1000285
Circular to Promulgate the List of Drugs, Chemicals, Antibiotics Banned for Uses, Limited to Use (2009)
Circular to revise and add some articles on the Circular Number 57/2012/TT-BNNPTNT dated 07/11/2012 on the monitoring, inspection and apply fines for banned veterinary drugs of the Beta-agonist group in feed (2016)
EMC (2015) Salbutamol Syrup 2 mg/5 ml. Retrieved 30 April, 2016, from http://www.medicines.org.uk/emc/medicine/24470
FAO and WHO (2002) Joint FAO/WHO expert committee on food additives, Limit test for heavy metals in food additive specifications, 59th meeting. Geneva: FAO and WHO
Heffernan A, Aylward L, Toms L-M, Sly P, MacLeod M, Mueller JF (2014) Pooled biological specimens for human biomonitoring of environmental chemicals: opportunities and limitations. J Eposure Sci Environ Epidemiol 24:225–232
Kim DP, Degand G, Douny C, Pierret G, Philippe Delahaut V, Ton D, Scippo M-L (2013) Preliminary evaluation of antimicrobial residue levels in marketed pork and chicken meat in the Red River Delta Region of Vietnam. Food Public Health 3(6):267–276. doi:10.5923/j.fph.20130306.02
National technical regulation Animal feeding stuffs—maximum level of antibiotics, drugs, microorganism and heavy metals in completed feeds for pigs (2009)
Nguyen A (2015) 80% of suspected feed companies used prohibited veterinary drugs, Dan Tri. Retrieved from http://dantri.com.vn/kinh-doanh/80-doanh-nghiep-thuc-an-chan-nuoi-su-dung-chat-cam-20151204105046746.htm
Nguyen Van Viet (2015) Report on preventing and applying fines on the violations of using banned veteranary drugs in feeds in provinces in the South. Hanoi
Nhiem DV, Peter P, Witaya S, Frans JMS, Moses NK, Maximilian POB, Ngan PH (2006) Preliminary analysis of tetracycline residues in marketed pork in Hanoi, Vietnam. Ann N Y Acad Sci 1081:534–542
OECD (2016) Meat consumption (indicator). Retrieved 15 May, 2016
Salpeter SR, Ormiston TM, Salpeter EE (2004) Cardiovascular effects of beta-agonists in patients with asthma and COPD: a meta-analysis. Chest 125(6):2309–2321
Strydom PE, Frylinck L, Montgomery JL, Smith MF (2008) The comparison of three beta-agonists for growth performance, carcass characteristics and meat quality of feedlot cattle. Meat Sci 81:557–564
Sundlo SF (2014) Veterinary drugs—general. Encycl Food Safety 3:35–38. doi:10.1016/B978-0-12-378612-8.00248-
The Minister of Health promulgates a Circular on the promulgation of the Regulation on Maximum limits on residues of veterinary medicines in food (2013)
Toản LQ, Hùng NV, Hương BM (2013) Đánh giá nguy cơ thịt lợn nhiễm salmonella ở Hà Nội Tạp chí Y học dự phòng, XXIII (3–140), 10–18
Tuyet Hanh TT, Duc NTM, Phuc PD, Tuat CV, Viet-Hung N (2015) Chemical hazards in pork and health risk: a review. Vietnam J Public Health 35:7–16
USAID (2015) Report National Opinion Survey. USAID, Hanoi, p 9
Van Duan, Huong, Nguyen (2016) Combating the use of prohibited veterinary drugs in pig raising in Vietnam. Retrieved 3 March, 2016, from http://nld.com.vn/kinh-te/tuyen-chien-voi-chat-cam-trong-chan-nuoi-20160303222342423.htm
Vietnam Food Administration (2016) Food poisoning statistic. Retrieved 10th Sept, 2016, from http://vesinhantoanthucpham.com.vn/thong-ke-ngo-doc-thuc-pham-tai-viet-nam
Vietnam National Institute of Nutrition, and Unicef (2010) Vietnam National Nutrition Survey 2009-2010 Hanoi
World Bank (2016) Food safety risk management in Vietnam: Challenges and opportunities. Technical working paper. Hanoi, Vietnam: World Bank
Woodward K (2013) Chapter 8 Antimicrobial Drugs. In: Woodward K (ed) Toxicological Effects of Veterinary Medicinal Products in Humans, pp 305
Acknowledgements
This study was a component of an ongoing PigRISK project (Reducing disease risks and improving food safety in smallholder pig value chains in Vietnam) (2012–2017), which assessed the health risks of chemical hazards in pork and developed incentive-based innovations to improve management of human and animal health risks in smallholder pig value chains in Vietnam. The study was funded by the Australian Centre for International Agricultural Research (ACIAR) and the CGIAR Research Program Agriculture for Nutrition and Health (A4NH). We would like to express our sincerely thanks to Associate Prof. Hoang Van Minh at Hanoi University of Public Health for his support in submitting this manuscript to the special supplement “Health and social determinants of health in Vietnam: local evidence and international implications”. Special thanks also go to Editors and reviewers for valuable comments on the draft manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The study was funded by the Australian Centre for International Agricultural Research (ACIAR) and the CGIAR Research Program Agriculture for Nutrition and Health (A4NH) led by IFPRI.
Ethical approval
All applicable international, national, and/or institutional guidelines for the care and use of animals were followed. All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. Ethical clearance and approval was obtained from the Hanoi University of Public Health’s Institutional Review Board (Number 148/2012/YTCC-HD3).
Conflict of interest
The authors declare no conflict of interest.
Additional information
This article is part of the supplement “Health and social determinants of health in Vietnam: local evidence and international implications.”
Rights and permissions
About this article
Cite this article
Tuyet-Hanh, T.T., Sinh, D.X., Phuc, P.D. et al. Exposure assessment of chemical hazards in pork meat, liver, and kidney, and health impact implication in Hung Yen and Nghe An provinces, Vietnam. Int J Public Health 62 (Suppl 1), 75–82 (2017). https://doi.org/10.1007/s00038-016-0912-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00038-016-0912-y