Abstract
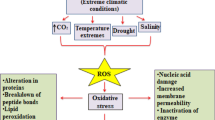
The central Chilean Andes are located in a Mediterranean-type climate zone, characterized by dry summers and high irradiance. This creates a contrasting elevational gradient because higher elevations get more solid precipitation and lower temperatures, resulting in higher soil humidity along the growing season compared with severe drought at lower elevations. Therefore, species with wide elevational distributions, such as Phacelia secunda, must have developed specific adaptations to cope with contrasting severity of drought stress-induced photoinhibition at different elevations. We hypothesize that P. secunda from lower elevation, is more tolerant to drought stress-induced photo-damage than plants from high elevation. This higher tolerance will be associated to a higher diversity of photoprotective strategies in plants that naturally suffers severe drought every growing season. To test this hypothesis, plants from 2700 and 3600 m in the central Chilean Andes were grown under the common garden and then subjected to water restriction. We measured stress indicators, photochemistry of PSII and PSI and estimate alternative electron sinks. Drought affected P. secunda photosynthetic performance differentially depending on the elevation of provenance. Plants from lower elevation exhibited higher drought tolerance than higher elevation ones. This was related to higher levels of heat dissipation and alternative electron sinks exhibited by plants from lower elevation under drought stress. We concluded that plants naturally subjected to recurrent drought are better adapted to respond to drought stress using additional photochemical photoprotective mechanisms and confirm the role of alternative electron sinks ameliorating photodamage.






Similar content being viewed by others
References
Ananyev G, Gates C, Kaplan A, Dismukes GC (2017) Photosystem II-cyclic electron flow powers exceptional photoprotection and record growth in the microalga Chlorella ohadii. Biochim Biophys Acta Bioenerg 1858:873–883
Arroyo MTK, Armesto JJ, Villagrán C (1981) Plant phenological patterns in the high Andean cordillera of central Chile. J Ecol 69:205–222
Asada K (1999) The water–water cycle in chloroplasts: scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Phys 50:601–639
Asada K (2006) Production and Scavenging of Reactive Oxygen Species in Chloroplasts and Their Functions. Plant Physiol 41:391–396
Berninger F, Streb P, Ensminger I (2012) Ecophysiology of photosynthesis in boreal, arctic and alpine ecosystems. Terrestrial photosynthesis in a changing environment—a molecular, physiological and Ecological approach, 1st edn. Cambridge University Press, Cambridge, pp 488–501
Biehler K, Fock H (1996) Evidence for the contribution of the Mehler-peroxidase reaction in dissipating excess electrons in drought-stressed wheat. Plant Physiol 112:265–272
Bravo LA, Saavedra-Mella FA, Vera F, Guerra A, Cavieres LA, Ivanov AG, Huner NPA, Corcuera LJ (2007) Effect of cold acclimation on the photosynthetic performance of two ecotypes of Colobanthus quitensis (Kunth) Bartl. J Exp Bot 58:3581–3590
Brestic M, Cornic G, Fryer MJ, Baker NR (1995) Does photorespiration protect the photosynthetic apparatus in French bean leaves from photoinhibition during drought stress? Planta 196:450–457
Buchner O, Holzinger A, Lütz C (2007) Effects of temperature and light on the formation of chloroplast protrusions in leaf mesophyll cells of high alpine plants. Plant Cell Environ 30:1347–1356
Cavieres LA (2000) Variación morfológica de Phacelia secunda J.F. Gmel. (Hydrophyllaceae) a lo largo de un gradiente altitudinal en Chile central. Gayana Bot 57:89–96
Cavieres LA, Badano E, Sierra-Almeida A, González-Gómez S, Molina-Montenegro M (2006) Positive interactions between alpine plant species and the nurse cushion plant Laretia acaulis do not increase with elevation in the Andes of central Chile. New Phytol 169:59–69
Cavieres LA, Badano EI, Sierra-Almeida A, Molina-Montenegro MA (2007) Microclimatic modifications of cushion plants and their consequences for seedling survival of native and non-native herbaceous species in the high Andes of central Chile. Arct Antarct Alp Res 39:229–236
Chen HX, Gao HY, An SZ, Li WJ (2004) Dissipation of excess energy in Mehler-peroxidase reaction in Rumex leaves during salt shock. Photosynthetica 42:117–122
Chow WS, Hope AB (2004) Electron fluxes through photosystem I in cucumber leaf discs probed by far-red fight. Photosynth Res 81:77–89
Cornic G, Massacci A (1996) Leaf Photosynthesis under drought Stress. In: Baker NR (ed) Photosynthesis and the Environment. Kluwer Academic Publishers, Dordrecht
Demmig-Adams B (1999) Survey of thermal energy dissipation and pigment composition in sun and shade leaves. Plant Cell Physiol 39:474–482
Demmig-Adams B, Adams W (1996) The role of xanthophyll cycle carotenoids in the protection of photosynthesis. Trends Plant Sci 1:21–26
Essemine J, Govindachary S, Ammar S, Bouzid S, Carpentier R (2011) Abolition of photosystem I cyclic electron flow in Arabidopsis thaliana following thermal-stress. Plant Physiol Biochem 49:235–243
Flexas J, Badger M, Chow WS, Medrano H, Osmond CB (1999) Analysis of the relative increase in photosynthetic O2 uptake when photosynthesis in grapevine leaves is inhibited following low night temperatures and/or water stress. Plant Physiol 121:675–684
Flores-Bavestrello A, Król M, Ivanov AG, Hüner NPA, García-Plazaola JI, Corcuera LJ, Bravo LA (2016) Two Hymenophyllaceae species from contrasting natural environments exhibit a homoiochlorophyllous strategy in response to desiccation stress. J Plant Physiol. https://doi.org/10.1016/j.jplph.2015.12.003
Galmés J, Abadía A, Cifré J, Medrano H, Flexas J (2007) Photoprotection processes under water stress and recovery in Mediterranean plants with different growth forms and leaf habits. Physiol Plant 130:495–510
Gao S, Wang G (2012) The enhancement of cyclic electron flow around photosystem I improves the recovery of severely desiccated Porphyra yezoensis (Bangiales, Rhodophyta). J Exp Bot 63:4349–4358
Gao S, Niu J, Chen W, Wang G, Xie X, Pan G, Gu W, Zhu D (2013) The physiological links of the increased photosystem II activity in moderately desiccated Porphyra haitanensis (Bangiales, Rhodophyta) to the cyclic electron flow during desiccation and re-hydration. Photosynth Res 116:45–54
García-Plazaola JI, Rojas R, Christie DA, Coopman RE (2015) Photosynthetic responses of trees in high-elevation forests: comparing evergreen species along an elevation gradient in the Central Andes. AoB Plants 7:plv058. https://doi.org/10.1093/aobpla/plv058
Genty B, Briantais JM, Baker NR (1989) The relationship between the quantum yield of photosynthetic electron transport and quenching of chlorophyll fluorescence. Biochim Biophys Acta 990:87–92
Golding AJ, Johnson JN (2003) Down-regulation of linear and activation of cyclic electron transport during drought. Planta 218:107–114
Golding AJ, Finazzi G, Johnson GN (2004) Reduction of the thylakoid electron transport chain by stromal reductants-evidence for activation of cyclic electron transport upon dark adaptation or under drought. Planta 220:356–363
Guan X, Gu S (2009) Photorespiration and photoprotection of grapevine (Vitis vinifera L. cv. Cabernet Sauvignon) under water stress. Photosynthetica 47:437–444
Guan X, Zhao S, Li D, Shu H (2004) Photoprotective function of photorespiration in several grapevine cultivars under drought stress. Photosynthetica 42:31–36
Habibi G, Ajory N (2015) The effect of drought on photosynthetic plasticity in Marrubium vulgare plants growing at low and high altitudes. J Plant Res. https://doi.org/10.1007/s10265-015-0748-1
Haupt-Herting S, Fock HP (2002) Oxygen exchange in relation to carbon assimilation in water-stressed leaves during photosynthesis. Ann Bot London 89:851–859
Heber U (2002) The Mehler reaction in relation to cyclic electron transport in C3 plants. Photosynth Res 73:223–231
Heber U, Bligny R, Streb P, Douce R (1996) Photorespiration is essential for the protection of the photosynthetic apparatus of C3 plants against photoinactivation under sunlight. Bot Acta 109:307–315
Hernández-Fuentes C, Bravo LA, Cavieres LA (2015) Photosynthetic responses and photoprotection strategies of Phacelia secunda plants exposed to experimental warming at different elevations in the central Chilean Andes. Alp Bot 125:87–99
Huang W, Zhang SB, Cao KF (2010) Stimulation of cyclic electron flow during recovery after chilling-induced photoinhibition of PSII. Plant Cell Physiol 51:1922–1928
Huang W, Zhang SB, Cao KF (2011) Cyclic electron flow plays an important role in photoprotection of tropical trees illuminated at temporal chilling temperature. Plant Cell Physiol 52:297–305
Huang W, Yang SJ, Zhang SB (2012) Cyclic electron flow plays an important role in photoprotection for the resurrection plant Paraboea rufescensunder drought stress. Planta 235:819–828
Huang W, Zhang SB, Hong H (2015) Insusceptibility of oxygen-evolving complex to high light in Betula platyphylla. J Plant Res 128:307–315
Huner NPA, Maxwell DP, Gray GR, Savitch LV, Krol M, Ivanov AG, Falk S (1996) Sensing environmental temperature change through imbalances between energy supply and energy consumption: redox state of photosystem II. Physiol Plant 98:358–364
Johnson GN (2011) Physiology of PSI cyclic electron transport in higher plants. Biochim Biophys Acta 1807:384–389
Klughammer C, Schreiber U (2008) Complementary PS II quantum yields calculated from simple fluorescence parameters measured by PAM fluorometry and the saturation pulse method. PAM Appl Not 1:27–35
Kohzuma K, Cruz JA, Akashi K, Hoshiyasu S, Munekage YN, Yokota A, Kramer DM (2009) The long-term responses of the photosynthetic proton circuit to drought. Plant Cell and Environ 32:209–219
Körner C (2003) Alpine plant life, 2da edn. Springer, Berlin, p 344
Kramer DM, Johnson G, Kiirats O, Edwards GE (2004) New fluorescence parameters for the determination of QA redox state and excitation energy fluxes. Photosynth Res 79:209–218
Kuhn M (2012) Rain and snow at high elevations: the interaction of water, energy and trace substances. In: Lütz C (ed) Plant in alpine regions: cell physiology of adaptation and survival strategies. Springer, Wien, pp 1–10
Li XG, Meng QW, Jiang GQ, Zou Q (2003) The susceptibility of cucumber and sweet pepper to chilling under low irradiance is related to energy dissipation and water-water cycle. Photosynthetica 41:259–265
Machler F, Nosberger J (1978) The adaptation to temperature of photorespiration and of the photosynthetic carbon metabolism of altitudinal ecotypes of Trifolium repens L. Oecologia 35:267–276
Machler F, Nosberger J, Erismann KH (1977) Photosynthetic 14CO2 fixation products in altitudinal ecotypes of Trifolium repens L. with different temperature requirements. Oecologia 31:79–84
Makino A, Miyake C, Yokota A (2002) Physiological functions of the water–water cycle (Mehler reaction) and the cyclic electron flow around PSI in rice leaves. Plant Cell Physiol 43:1017–1026
Manuel N, Cornic G, Aubert S, Choler P, Bligny G, Heber U (1999) Protection against Photoinhibition in the Alpine Plant Geum montanum. Oecologia 119:149–158
Maxwell K, Johnson GN (2000) Chlorophyll fluorescence-a practical guide. J Exp Bot 51:659–668
Miyake C (2010) Alternative electron flows (water–water cycle and cyclic electron flow around PSI) in photosynthesis: molecular mechanisms and physiological functions. Plant Cell Physiol 51(12):1951–1963
Miyake C, Horiguchi S, Makino A, Shinzaki Y, Yamamoto H, Tomizawa K (2005) Effects of light intensity on cyclic electron flow around PSI and its relationship to non-photochemical quenching of Chl fluorescence in tobacco leaves. Plant Cell Physiol 46:1819–1830
Moss DA, Bendall DS (1984) Cyclic electron transport in chloroplasts: the Q-cycle and the site of action of antimycin. Biochim Biophys Acta 767:389–395
Müller P, Xiao-Ping L, Niyogi KK (2001) Non-photochemical quenching. A response to excess light energy. Plant Physiol 125:1558–1566
Munekage Y, Hojo M, Meurer J, Endo T, Tasaka M, Shikanai T (2002) PGR5 is involved in cyclic electron flow around photosystem I and is essential for photoprotection in Arabidopsis. Cell 110:361–371
Munekage Y, Hashimoto M, Miyake C (2004) Cyclic electron flow around photosystem I is essential for photosynthesis. Nature 429:579–582
Munekage Y, Genty B, Peltier G (2008) Effect of PGR5 impairment on photosynthesis and growth in Arabidopsis thaliana. Plant Cell Physiol 49:1688–1698
Nogués S, Alegre L (2002) An increase in water deficit has no impact on the photosynthetic capacity of field-grown Mediterranean plants. Funct Plant Biol 29:621–630
Ogren WL (1984) Photorespiration: pathways, regulation, and modification. Ann Rev Plant Physiol 35:415–442
Öquist G, Huner NP (2003) Photosynthesis of overwintering evergreen plants. Annu Rev Plant Biol 54:329–355
Ort D, Baker N (2002) A photoprotective role for O2 as an alternative electron sink in photosynthesis? Curr Opin Plant Biol 3:193–198
Osmond CB, Grace SC (1995) Perspectives on photoinhibition and photorespiration in the field: quintessential inefficiencies of the light and dark reactions of photosynthesis? J Exp Bot 46:1351–1362
Pfündel E, Klughammer C, Schreiber U (2008) Monitoring the effects of reduced PS II antenna size on quantum yields of photosystems I and II using the Dual-PAM-100 measuring system. PAM Appl Not 1:21–24
Rumeau D, Peltier G, Cournac L (2007) Chlororespiration and cyclic electron flow around PSI during photosynthesis and plant stress response. Plant Cell Environ 30:1041–1051
Ruuska SA, Badger MR, Andrews TJ, von Caemmerer S (2000) Photosynthetic electron sinks in transgenic tobacco with reduced amounts of Rubisco: little evidence for significant Mehler reaction. J Exp Bot 51:357–368
Sanfuentes C, Sierra-Almeida A, Cavieres LA (2012) Efecto del aumento de la temperatura en el desempeño fotosintético de una especie alto-andina en dos altitudes contrastantes. Gayan Bot 69:38–46
Santibáñez F, Uribe JM (1990) Atlas agroclimático de Chile. Regiones VIII y IX. Ediciones Universidad de Chile, Santiago
Shikanai T (2014) Central role of cyclic electron transport around photosystem I in the regulation of photosynthesis. Curr Opin Biotechnol 26:25–30
Sierra-Almeida A, Cavieres LA (2010) Summer freezing resistance decreased in high-elevation plants exposed to experimental warming in the central Chilean Andes. Oecologia 163:267–276
Sierra-Almeida A, Reyes-Bahamonde C, Cavieres LA (2016) Drought increases the freezing resistance of high-elevation plants of the Central Chilean Andes. Oecologia 181:1011–1023
Strand DD, Livingston AK, Satoh-Cruza M, Froehlich JE, Maurino VG, Kramer DM (2015) Activation of cyclic electron flow by hydrogen peroxide in vivo. Proc Natl Acad Sci 12:5539–5544
Streb P, Cornic G (2012) Photosynthesis and antioxidative protection in alpine herbs. In: Lütz C (ed) Plant in Alpine regions: cell physiology of adaptation and survival strategies. Springer, Wien, pp 75–97
Streb P, Feierabend J, Bligny R (1997) Resistance to photoinhibition of photosystem II and catalase and antioxidative protection in high mountain plants. Plant Cell Environ 20:1030–1040
Streb P, Feierabend Y, Bligny R (1998) Divergent strategies of photoprotection in high mountain plants. Planta 207:313–324
Streb P, Josse EM, Gallouët E, Baptist F, Kuntz M, Cornic G (2005) Evidence for alternative electron sinks to photosynthetic carbon assimilation in the high mountain plant species Ranunculus glacialis. Plant Cell Environ 28:1123–1135
Takahashi S, Milward SE, Fan DY, Chow WS, Badger M (2009) How does cyclic electron flow alleviate photoinhibition in arabidopsis? Plant Physiol 149:1560–1567
Walker BJ, Strand DD, Kramer DM, Cousins AB (2014) The response of cyclic electron flow around photosystem i to changes in photorespiration and nitrate assimilation. Plant Physiol 165:453–462
Wiese C, Shi LB, Heber U (1998) Oxygen reduction in the Mehler reaction is insufficient to protect photosystems I and II of leaves against photoinhibition. Physiol Plant 102:437–446
Wilhelm C, Selmar D (2011) Energy dissipation is an essential mechanism to sustain the viability of plants: the physiological limits of improved photosynthesis. J Plant Physiol 168:79–87
Williams EL, Hovenden MJ, Close DC (2003) Strategies of light energy utilization, dissipation and attenuation in six co-occurring alpine heath species in Tasmania. Funct Plant Biol 30:1205–1218
Wingler A, Lea PJ, Quick WP, Leegood RC (2000) Photorespiration: metabolic pathways and their role in stress protection. Philos Trans R Soc B 355:1517–1529
Acknowledgements
The research was supported by Fondecyt 1060910, CONICYT AFB170008 and NEXER-UFRO (NXR17-0002). Authors thank Dan Harris-Pascal for English corrections in the manuscript.
Author information
Authors and Affiliations
Contributions
C.H.F and L.A.B conceived the idea, and all authors made substantial contributions to the study design. All authors contributed to acquisition of data and C.H.F and R.E.C led analyses. C.H.F led manuscript preparation with substantial critical and editorial input from all authors.
Corresponding author
Ethics declarations
Conflict of interest
This study is in compliance with ethical standards and the authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Hernández-Fuentes, C., Coopman, R.E., Cavieres, L.A. et al. Photoprotective strategies against drought are depending on the elevation provenance in Phacelia secunda. Alp Botany 129, 123–135 (2019). https://doi.org/10.1007/s00035-019-00221-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00035-019-00221-7