Abstract
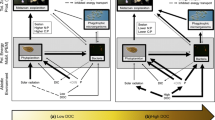
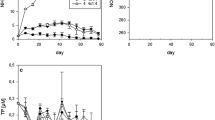
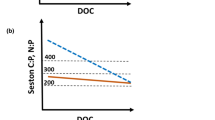
Atmospheric nitrogen deposition predominantly influences ecosystems by shifting their available nutrient budgets towards excess nitrogen conditions. In temperate lakes nitrogen is often naturally in excess and phosphorus is deficient, when compared with the optimal Redfield ratio of 16:1. To investigate effects of future increasing nitrogen conditions on lake plankton communities, we performed mesocosm experiments in three different nitrogen rich lakes, all characterised by high nitrogen to phosphorus ratios. In order to determine functional responses to increased nitrogen loading, we conducted six nitrogen fertilization treatments. Nitrogen fertilization was based upon existing nitrate and ammonium concentrations in natural wet deposition and multiple loadings of these concentrations. Despite the initial conditions of excess nitrogen, removal of additional nitrogen by the plankton community was observed in all of the lakes. In one lake, an increasing phosphorus limitation became visible in seston stoichiometry. Over all of the lakes and within each lake’s experimental nitrogen gradient, we found evidence for decreased mesozooplankton due to nitrogen enrichment. The negative responses of mesozooplankton to N enrichment were mainly restricted to cladocerans and nauplii. The results indicate that nitrogen enrichment within the magnitudes of projected future atmospheric nitrogen depositions may lead to a long-term reduction of mesozooplankton in phosphorus deficient lakes. The transfer of nitrogen enrichment effects on lower food-web dynamics could have consequences for higher trophic levels, such as fish.




Similar content being viewed by others
References
Aber JD, Goodale CL, Ollinger SV, Smith M-L, Magill AH, Martin ME, Hallett RA, Stoddard JL (2003) Is nitrogen deposition altering the nitrogen status of northeastern forests? Bioscience 53:375–389
Andersen T, Hessen DO (1991) Carbon, nitrogen, and phosphorus content of freshwater zooplankton. Limnol Oceanogr 36:807–814
Andersen T, Saloranta TM, Tamminen T (2007) A statistical procedure for unsupervised classification of nutrient limitation bioassay experiments with natural phytoplankton communities. Limnol Oceanogr Methods 5:111–118
Berger SA, Diehl S, Kunz TJ, Albrecht D, Oucible AM, Ritzer S (2006) Light supply, plankton biomass, and seston stoichiometry in a gradient of lake mixing depths. Limnol Oceanogr 51:1898–1905
Bergström A-K, Jansson M (2006) Atmospheric nitrogen deposition has caused nitrogen enrichment and eutrophication of lakes in the northern hemisphere. Glob Change Biol 12:635–643
Bergström A-K, Blomqvist P, Jansson M (2005) Effects of atmospheric nitrogen deposition on nutrient limitation and phytoplankton biomass in unproductive Swedish lakes. Limnol Oceanogr 50:987–994
Beutler M, Wiltshire KH, Meyer B, Moldaenke C, Lüring C, Meyerhöfer M, Hansen U-P, Dau H (2002) A fluorometric method for the differentiation of algal populations in vivo and in situ. Photosynth Res 72:39–53
Bottrell HH, Duncan A, Gliwicz ZM, Grygierek E, Herzig A, Hillbricht-Ilkowska A, Kurasawa H, Larsson P, Weglenska T (1976) A review of some problems in zooplankton production studies. Norw J Zool 24:419–456
Brett MT, Müller-Navarra DC, Park S-K (2000) Empirical analysis of the effect of phosphorus limitation on algal food quality for freshwater zooplankton. Limnol Oceanogr 45:1564–1575
Brooks LJ (1968) The effects of prey size selection by lake planktivores. Syst Biol 17:273–291
Camargo JA, Alonso A, Salamanca A (2005) Nitrate toxicity to aquatic animals: a review with new data for freshwater invertebrates. Chemosphere 58:1255–1267
Carrillo P, Villar-Argaiz M, Medina-Sanchez JM (2001) Relationship between N:P ratio and growth rate during the life cycle of calanoid copepods: an in situ measurement. J Plankton Res 23:537–547
Ciais P, Sabine C, Bala G, Bopp L, Brovkin V, Canadell J, Chhabra A, DeFries R, Galloway J, Heimann M, Jones C, Le Quéré C, Myneni RB, Piao S, Thornton P (2013) Carbon and other biogeochemical cycles. In: Stocker TF, Qin D, Plattner G-K, Tignor M, Allen SK, Boschung J, Nauels A, Xia Y, Bex V, Midgley PM (eds) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Collos Y, Harrison PJ (2014) Acclimation and toxicity of high ammonium concentrations to unicellular algae. Marine Poll Bull 80:8–23
Crowley KF, McNeil BE, Lovett GM, Canham D, Driscoll CT, Rustad LE, Denny E, Hallett RA, Arthur MA, Boggs JL, Goodale CL, Kahl JS, Nulty SG, Ollinger SV, Pardo LH, Schaberg PG, Stoddard JL, Weand MP, Weathers KC (2012) Do nutrient limitation patterns shift from nitrogen toward phosphorus with increasing nitrogen deposition across the northeastern United States? Ecosystems 15:940–957
Darchambeau F, Faerovig PJ, Hessen DO (2003) How Daphnia copes with excess carbon in its food. Oecologia 136:336–346
Denman KL, Brasseur G, Chidthaisong A, Ciais P, Cox PM, Dickinson RE, Hauglustaine D, Heinze C, Holland E, Jacob D, Lohmann U, Ramachandran S, da Silva Dias PL, Wofsy SC, Zhang X (2007) Couplings between changes in the climate system and biogeochemistry. In: Solomon S, Qin D, Manning M, Chen Z, Marquis M, Averyt KB, Tignor M, Miller HL (eds) Climate change 2007: the physical science basis. Contribution of working group I to the fourth assessment report of the intergovernmental panel on climate change. Cambridge University Press, Cambridge
Donald D, Bogard M, Finlay K, Bunting L, Leavitt PR (2013) Phytoplankton-specific response to enrichment of phosphorus-rich surface waters with ammonium, nitrate, and urea. PLoS One 8:e53277
Dortch Q (1990) The interaction between ammonium and nitrate uptake in phytoplankton. Mar Ecol Prog Ser 61:183–201
Elser JJ, Marzolf ER, Goldman CR (1990) Phosphorus and nitrogen limitation of phytoplankton growth in the freshwaters of North America: a review and critique of experimental enrichments. Can J Fish Aquat Sci 46:1468–1477
Elser JJ, Fagan WF, Denno RF, Dobberfuhl DR, Folarin A, Huberty A, Interlandi S, Kilham SS, McCauley E, Schulz KL, Siemann EH, Sterner RW (2000) Nutritional constraints in terrestrial and freshwater food webs. Nature 408:578–580
Elser JJ, Hayakawa K, Urabe J (2001) Nutrient limitation reduces food quality for zooplankton: Daphnia response to seston phosphorus enrichment. Ecology 82:898–903
Elser JJ, Bracken MES, Cheland EE, Gruner DS, Harpole WS, Hildebrand H, Ngai JT, Seabloom EW, Shurin JB, Smith JE (2007) Global analysis of nitrogen and phosphorus limitation of primary producers in freshwater, marine and terrestrial ecosystems. Ecol Lett 10:1–8
Elser JJ, Kyle M, Steger L, Nydick KR, Baron JS (2009a) Nutrient availability and phytoplankton nutrient limitation across a gradient of atmospheric nitrogen deposition. Ecology 90:3062–3073
Elser JJ, Andersen T, Baron JS, Bergström AK, Jansson M, Kyle M, Nydick KR, Steger L, Hessen DO (2009b) Shifts in lake N:P stoichiometry and nutrient limitation driven by atmospheric nitrogen deposition. Science 326:835–837
Elser JJ, Peace AL, Kyle M, Wojewodzic M, McCrackin ML, Andersen T, Hessen DO (2010) Atmospheric nitrogen deposition is associated with elevated phosphorus limitation of lake zooplankton. Ecol Lett 13:1256–1261
Felip M, Catalan J (2000) The relationship between phytoplankton biovolume and chlorophyll in a deep oligotrophic lake: decoupling spatial and temporal maxima. J Plankton Res 22:91–105
Galloway JN, Townsend AR, Erisman JW, Bekunda M, Cai Z, Freney JR, Martinelli LA, Seitzinger SP, Sutton MA (2008) Transformation of the nitrogen cycle: recent trends, questions, and potential solutions. Science 320:889–892
Gersich FM, Hopkins DL (1986) Site-specific acute and chronic toxicity of ammonia to Daphnia magna Straus. Environ Toxicol Chem 5:443–447
Glibert PM, Wilkerson FP, Dugdale RC, Raven JA, Dupont CL, Leavitt PR, Parker AE, Burkholder JM, Kana TM (2016) Pluses and minuses of ammonium and nitrate uptake and assimilation by phytoplankton and implications for productivity and community composition, with emphasis on nitrogen-enriched conditions. Limnol Oceanogr 61:165–197
Goll DS, Brovkin V, Parida BR, Reick CH, Kattge J, Reich PB, van Bodegom PM, Niinemets Ü (2012) Nutrient limitation reduces land carbon uptake in simulations with a model of combined carbon, nitrogen and phosphorus cycling. Biogeosciences 9:3547–3569
Gundersen P, Callesen I, De Vries W (1998) Nitrate leaching in forest ecosystems is related to forest floor C/N ratios. Environ Pollut 102:403–407
Hammer O, Harper DAT, Ryan PD (2001) PAST: paleontological Statistics software package for education and data analysis, version 2.17. http://folk.uio.no/ohammer/past/. Accessed 5 Jan 2014
Hansen B, Bjornsen PK, Hansen PJ (1994) The size ratio between planktonic predators and their prey. Limnol Oceanogr 39:395–403
Hessen DO (1992) Nutrient element limitation of zooplankton production. Am Nat 140:799–814
Hessen DO (2013) Inorganic nitrogen deposition and its impacts on N:P-ratios and lake productivity. Water 5:327–341
Hessen DO, Faerovig PJ, Andersen T (2002) Light, nutrients, and P:C ratios in algae: grazer performance related to food quality and quantity. Ecology 83:1886–1898
Hessen DO, Elser JJ, Sterner RW, Urabe J (2013) Ecological stoichiometry: an elementary approach using basic principles. Limnol Oceanogr 58:2219–2236
Holmes RM, Aminot A, Kerouel R, Hooker BA, Peterson BJ (1999) A simple and precise method for measuring ammonium in marine and freshwater ecosystems. Can J Fish Aquat Sci 56:1801–1808
Ilic M (2014) Nutrient limitation under increased phosphorus limitation in two Bavarian lakes. Master Thesis, Ludwig-Maximilians University Munich pp. 61
Kim I-N, Lee K, Gruber N, Karl DM, Bullister JL, Yang S, Kim T-W (2014) Increasing anthropogenic nitrogen in the North Pacific Ocean. Science 346:1102–1106
Klausmeier C, Litchman E, Daufresne T, Levin SA (2004) Optimal nitrogen-to-phosphorus stoichiometry of phytoplankton. Nature 429:171–174
Legendre P, Legendre L (1998) Numerical ecology. 2nd English. Elsevier, Amsterdam
Levasseur M, Thompson PA, Harrison PJ (1993) Physiological acclimation of marine phytoplankton to different nitrogen sources. J Phycol 29:587–595
Lovett GM, Arthur MA, Weathers KC, Fitzhugh RD, Templer PH (2013) Nitrogen addition increases carbon storage in soils, but not in trees, in an Eastern U.S. deciduous forest. Ecosystems 16:980–1001
McCarthy SDS, Rafferty SP, Frost PC (2010) Responses of alkaline phosphatase activity to phosphorus stress in Daphnia magna. J Exp Biol 213:256–261
Meunier CL, Boersma M, Wiltshire KH, Malzahn AM (2015) Zooplankton eat what they need: copepod selective feeding and potential consequences for marine systems. Oikos 0:001–009
Mulder K, Bowden WB (2007) Organismal stoichiometry and the adaptive advantage of variable nutrient use and production efficiency in Daphnia. Ecol Modell 202:427–440
Müller-Navarra DC (1995) Biochemical versus mineral limitation in Daphnia. Limnol Oceanogr 40:1209–1214
Müller-Navarra DC, Brett MT, Liston AM, Goldman CR (2000) A highly unsaturated fatty acid predicts carbon transfer between primary producers and consumers. Nature 403:64–77
Paasche E (1971) Effect of ammonia and nitrate on growth, photosynthesis, and Ribulose diphosphate carboxylase content of Dunaliella tertiolecta. Physiol Plant 25:294–299
Paerl H (2009) Controlling eutrophication along the freshwater-marine continuum: dual nutrient (N and P) reductions are essential. Estuaries Coasts 32:593–601
Pauli H-R (1989) A new method to estimate individual dry weights of rotifer. Hydrobiologia 186/187:355–361
Plath K, Boersma M (2001) Mineral limitation of zooplankton: stoichiometric constraints and optimal foraging. Ecology 82:1260–1269
Poxleitner M, Trommer G, Lorenz P, Stibor H (2016) The effect of increased nitrogen load on phytoplankton in a phosphorus-limited lake. Freshw Biol 61:1966–1980
Rabalais NN, Turner RE, Diaz RJ, Justic D (2009) Global change and eutrophication of coastal waters. ICES J Mar Sci 66:1528–1537
Ragueneau O, Tréguer P (1994) Determination of biogenic silica in coastal waters: applicability and limits of the alkaline digestion method. Mar Chem 45:43–51
Reay DS, Dentener F, Smith P, Grace J, Feely RA (2008) Global nitrogen deposition and carbon sinks. Nat Geosci 1:430–437
Reich PB, Knops J, Tilman D, Craine J, Ellsworth D, Tjoelker M, Lee T, Wedin D, Naeem S, Bahauddin D, Hendrey G, Jose S, Wrage K, Goth J, Bengston W (2001) Plant diversity enhances ecosystem responses to elevated CO2 and nitrogen deposition. Nature 410:809–810
Rockström J, Steffen W, Noone K, Persson A, Chapin FS, Lambin EF, Lenton TM, Scheffer M, Folke C, Schellnhuber HJ, Nykvist B, de Wit CA, Hughes T, van der Leeuw S, Rodhe H, Sörlin S, Snyder PK, Costanza R, Svedin U, Falkenmark M, Karlberg L, Corell RW, Fabry VJ, Hansen J, Walker B, Liverman D, Richardson K, Crutzen P, Foley JA (2009) A safe operating space for humanity. Nature 461:472–475
Schindler DW (1977) Evolution of phosphorus limitation in lakes. Science 195:260–262
Schindler DW, Hecky RE, Findlay DL, Stainton MP, Parker BR, Paterson MJ, Beaty KG, Lyng M, Kasian SEM (2008) Eutrophication of lakes cannot be controlled by reducing nitrogen input: Results of a 37-year whole-ecosystem experiment. PNAS 105:11254–11258
Sommer U (1992) Phosphorus-limited Daphnia: Intraspecific facilitation instead of competition. Limnol Oceanogr 37:966–973
Sommer U, Gliwicz ZM, Lampert W, Duncan A (1986) The PEG-model of seasonal succession of planktonic events in fresh waters. Arch Hydrobiol 4:433–471
Sommer U, Adrian A, De Senerpont Domis L, Elser JJ, Gaedke U, Ibelings B, Jeppesen E, Lürling M, Molinero JC, Mooij WM, van Donk E, Winder M (2012) Beyond the plankton ecology group (PEG) model: mechanisms driving plankton succession. Annu Rev Ecol Evol Syst 43:429–448
Steffen W, Richardson K, Rockström J, Cornell SE, Fetzer I, Bennett EM, Biggs R, Carpenter SR, de Vries W, de Wit CA, Folke C, Gerten D, Heinke J, Mace GM, Persson LM, Ramanathan V, Reyers B, Sorlin S (2015) Planetary boundaries: guiding human development on a changing planet. Science. doi:10.1126/science.1259855
Sterner RW, Elser JJ (2002) Ecological stoichiometry: the biology of elements from molecules to the biosphere. Princeton University Press, Princeton, NJ
Sterner RW, Hessen DO (1994) Algal nutrient limitation and the nutrition of aquatic herbivores. Annu Rev Ecol Syst 25:1–29
Sterner RW, Elser JJ, Fee EJ, Guildford SJ, Chrzanowski TH (1997) The light:nutrient ratio in lakes: the balance of energy and materials affects ecosystem structure and process. Am Nat 150:663–684
Trommer G, Pondaven P, Siccha M, Stibor H (2012) Zooplankton-mediated nutrient limitation patterns in marine phytoplankton: an experimental approach with natural communities. Mar Ecol Prog Ser 449:83–94
Tyrrell T (1999) The relative influences of nitrogen and phosphorus on oceanic primary production. Nature 400:525–531
Urabe J, Clasen J, Sterner RW (1997) Phosphorus limitation of Daphnia growth: Is it real? Limnol Oceanogr 42:1436–1443
Vanni MJ (1987) Effects of food availability and fish predation on a zooplankton community. Ecol Monogr 57:61–87
Vitousek PM, Aber JD, Howarth RW, Likens GE, Pamela A, Schindler DW, Schlesinger WH, Tilman DG (1997) Human alteration of the global nitrogen cycle: sources and consequences. Ecol Appl 7:737–750
Vitousek PM, Porder S, Houlton BZ, Chadwick OA (2010) Terrestrial phosphorus limitation: mechanisms, implications, and nitrogen–phosphorus interactions. Ecol Appl 20:5–15
Wacker A, von Elert E (2001) Polyunsaturated fatty acids: evidence for non-substitutable biochemical resources in Daphnia galeata. Ecology 82:2507–2520
Watkins J, Rudstam L, Kristen H (2011) Length-weight regression for zooplankton biomass calculations—a review and a suggestion for standard equations. eCommons Cornell. http://hdl.handle.net/1813/24566. Accessed 1 July 2013
Wetzel RG, Likens GE (1991) Limnological analyses. 2nd edn. Springer, New York
Acknowledgements
We wish to thank A. Wild and A. Weigert for technical support and laboratory analyses. We are grateful for the help of colleagues, students and the commune of Seeon for setting up the experiments. We thank the editors U. Sommer and S. Findlay, and two anonymous reviewers for their helpful remarks to improve this manuscript. This study was funded by the German Research Foundation to G.T. (DFG Tr 1126/1–1).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Trommer, G., Poxleitner, M., Lorenz, P. et al. Altered food-web dynamics under increased nitrogen load in phosphorus deficient lakes. Aquat Sci 79, 1009–1021 (2017). https://doi.org/10.1007/s00027-017-0551-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00027-017-0551-2