Abstract.
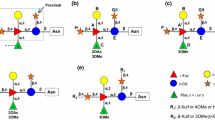
Cyanovirin-N (CV-N), an 11-kDa protein from the cyanobacterium Nostoc ellipsosporum, is a highly potent virucidal agent that has generated interest as a lead natural product for the prevention and chemotherapy of human immunodeficiency virus infection. The antiviral activity of CV-N is mediated through specific, high-affinity interactions with the viral surface envelope glycoproteins. A number of structures of wild-type, mutant and sequence-shuffled CV-N have been solved by nuclear magnetic resonance and crystallography, showing that the protein exists as either a quasi-symmetric two-domain monomer or a domain-swapped dimer. Structures of several complexes of CV-N with oligosaccharides help in explaining the unique mode of high-affinity binding of these molecules to both forms of CV-N.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
RID="*"
ID="*"Corresponding author.
Rights and permissions
About this article
Cite this article
Botos, I., Wlodawer, A. Cyanovirin-N: a sugar-binding antiviral protein with a new twist. CMLS, Cell. Mol. Life Sci. 60, 277–287 (2003). https://doi.org/10.1007/s000180300023
Issue Date:
DOI: https://doi.org/10.1007/s000180300023