Abstract
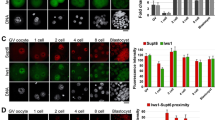
N-acetyltransferase 10 (NAT10)-mediated N4-acetylcytidine (ac4C) modification is crucial for mRNA stability and translation efficiency, yet the underlying function in mammalian preimplantation embryos remains unclear. Here, we characterized the ac4C modification landscape in mouse early embryos and found that the majority of embryos deficient in ac4C writer-NAT10 failed to develop into normal blastocysts. Through single-cell sequencing, RNA-seq, acetylated RNA immunoprecipitation combined with PCR (acRIP-PCR), and embryonic phenotype monitoring, Nop2 was screened as a target gene of Nat10. Mechanistically, Nat10 knockdown decreases the ac4C modification on Nop2 mRNA and reduces RNA and protein abundance by affecting the mRNA stability of Nop2. Then, depletion of NOP2 may inhibit the translation of transcription factor TEAD4, resulting in defective expression of the downstream lineage-specific gene Cdx2, and ultimately preventing blastomeres from undergoing the trophectoderm (TE) fate. However, exogenous Nop2 mRNA partially reverses this abnormal development. In conclusion, our findings demonstrate that defective ac4C modification of Nop2 mRNA hinders the morula-to-blastocyst transition by influencing the first cell fate decision in mice.







Similar content being viewed by others
Data Availability
All data supporting this study are available within the article and the Supplementary Materials. The RNA-seq data of Nat10-depleted morulae in mice have been deposited in the Gene Expression Omnibus database (GEO) under the accession number GSE236943.
References
Watson AJ, Natale DR, Barcroft LC (2004) Molecular regulation of blastocyst formation. Anim Reprod Sci 82–83:583–592
Fleming TP, Pickering SJ (1985) Maturation and polarization of the endocytotic system in outside blastomeres during mouse preimplantation development. J Embryol Exp Morphol 89:175–208
Johnson MH, McConnell JM (2004) Lineage allocation and cell polarity during mouse embryogenesis. Semin Cell Dev Biol 15:583–597
Johnson MH, Ziomek CA (1983) Cell interactions influence the fate of mouse blastomeres undergoing the transition from the 16- to the 32-cell stage. Dev Biol 95:211–218
Dahl JA, Jung I, Aanes H et al (2016) Broad histone h3k4me3 domains in mouse oocytes modulate maternal-to-zygotic transition. Nature 537:548–552
Inoue A, Zhang Y (2011) Replication-dependent loss of 5-hydroxymethylcytosine in mouse preimplantation embryos. Science 334:194
Sui X, Hu Y, Ren C et al (2020) Mettl3-mediated m(6)a is required for murine oocyte maturation and maternal-to-zygotic transition. Cell Cycle 19:391–404
Yang Y, Wang L, Han X et al (2019) Rna 5-methylcytosine facilitates the maternal-to-zygotic transition by preventing maternal mrna decay. Mol Cell 75(1188–1202):e1111
Cohn WE (1960) Pseudouridine, a carbon-carbon linked ribonucleoside in ribonucleic acids: Isolation, structure, and chemical characteristics. J Biol Chem 235:1488–1498
Kumbhar BV, Kamble AD, Sonawane KD (2013) Conformational preferences of modified nucleoside n(4)-acetylcytidine, ac4c occur at “wobble” 34th position in the anticodon loop of trna. Cell Biochem Biophys 66:797–816
Ito S, Horikawa S, Suzuki T et al (2014) Human nat10 is an atp-dependent rna acetyltransferase responsible for n4-acetylcytidine formation in 18 s ribosomal rna (rrna). J Biol Chem 289:35724–35730
Arango D, Sturgill D, Alhusaini N et al (2018) Acetylation of cytidine in mrna promotes translation efficiency. Cell 175(1872–1886):e1824
Tardu M, Jones JD, Kennedy RT et al (2019) Identification and quantification of modified nucleosides in saccharomyces cerevisiae mrnas. ACS Chem Biol 14:1403–1409
Yang W, Li HY, Wu YF et al (2021) Ac4c acetylation of runx2 catalyzed by nat10 spurs osteogenesis of bmscs and prevents ovariectomy-induced bone loss. Mol Ther Nucleic acids 26:135–147
Guo G, Shi X, Wang H et al (2020) Epitranscriptomic n4-acetylcytidine profiling in cd4(+) t cells of systemic lupus erythematosus. Front Cell Dev Biol 8:842
Zhang Y, Jing Y, Wang Y et al (2021) Nat10 promotes gastric cancer metastasis via n4-acetylated col5a1. Signal Transduct Target Ther 6:173
Wang G, Zhang M, Zhang Y et al (2022) Nat10-mediated mrna n4-acetylcytidine modification promotes bladder cancer progression. Clin Transl Med 12:e738
Wang K, Zhou LY, Liu F et al (2022) Piwi-interacting rna haapir regulates cardiomyocyte death after myocardial infarction by promoting nat10-mediated ac(4) c acetylation of tfec mrna. Adv Sci 9:e2106058
Xiang Y, Zhou C, Zeng Y et al (2021) Nat10-mediated n4-acetylcytidine of rna contributes to post-transcriptional regulation of mouse oocyte maturation in vitro. Front Cell Dev Biol 9:704341
Lin J, Xiang Y, Huang J et al (2022) Nat10 maintains oga mrna stability through ac4c modification in regulating oocyte maturation. Front Endocrinol 13:907286
Chen L, Wang WJ, Liu Q et al (2022) Nat10-mediated n4-acetylcytidine modification is required for meiosis entry and progression in male germ cells. Nucleic acids Res 50:10896–10913
Cui W, Dai X, Marcho C et al (2016) Towards functional annotation of the preimplantation transcriptome: an rnai screen in mammalian embryos. Sci Rep 6:37396
Wang Y, Yuan P, Yan Z et al (2021) Single-cell multiomics sequencing reveals the functional regulatory landscape of early embryos. Nat Commun 12:1247
Hao Y, Hao S, Andersen-Nissen E et al (2021) Integrated analysis of multimodal single-cell data. Cell 184(3573–3587):e3529
Zhou Y, Zhou B, Pache L et al (2019) Metascape provides a biologist-oriented resource for the analysis of systems-level datasets. Nat Commun 10:1523
Arango D, Sturgill D, Oberdoerffer S (2019) Immunoprecipitation and sequencing of acetylated rna. Bio-Protoc 9:e3278
Chen CY, Ezzeddine N, Shyu AB (2008) Messenger rna half-life measurements in mammalian cells. Methods Enzymol 448:335–357
Wang X, Lu Z, Gomez A et al (2014) N6-methyladenosine-dependent regulation of messenger rna stability. Nature 505:117–120
Lasman L, Krupalnik V, Viukov S et al (2020) Context-dependent functional compensation between ythdf m(6)a reader proteins. Genes Dev 34:1373–1391
Strumpf D, Mao CA, Yamanaka Y et al (2005) Cdx2 is required for correct cell fate specification and differentiation of trophectoderm in the mouse blastocyst. Development 132:2093–2102
Jedrusik A, Bruce AW, Tan MH et al (2010) Maternally and zygotically provided cdx2 have novel and critical roles for early development of the mouse embryo. Dev Biol 344:66–78
Nishioka N, Yamamoto S, Kiyonari H et al (2008) Tead4 is required for specification of trophectoderm in pre-implantation mouse embryos. Mech Dev 125:270–283
Nishioka N, Inoue K, Adachi K et al (2009) The hippo signaling pathway components lats and yap pattern tead4 activity to distinguish mouse trophectoderm from inner cell mass. Dev Cell 16:398–410
Sas-Chen A, Thomas JM, Matzov D et al (2020) Dynamic rna acetylation revealed by quantitative cross-evolutionary mapping. Nature 583:638–643
Thalalla Gamage S, Sas-Chen A, Schwartz S et al (2021) Quantitative nucleotide resolution profiling of rna cytidine acetylation by ac4c-seq. Nat Protoc 16:2286–2307
Arango D, Sturgill D, Yang R et al (2022) Direct epitranscriptomic regulation of mammalian translation initiation through n4-acetylcytidine. Mol Cell 82(2797–2814):e2711
Jiang X, Cheng Y, Zhu Y et al (2023) Maternal nat10 orchestrates oocyte meiotic cell-cycle progression and maturation in mice. Nat Commun 14:3729
Wu R, Li A, Sun B et al (2019) A novel m(6)a reader prrc2a controls oligodendroglial specification and myelination. Cell Res 29:23–41
Lan T, Li H, Zhang D et al (2019) Kiaa1429 contributes to liver cancer progression through n6-methyladenosine-dependent post-transcriptional modification of gata3. Mol Cancer 18:186
Wu Y, Xu X, Qi M et al (2022) N(6)-methyladenosine regulates maternal rna maintenance in oocytes and timely rna decay during mouse maternal-to-zygotic transition. Nat Cell Biol 24:917–927
Zeng Y, Wang S, Gao S et al (2018) Refined rip-seq protocol for epitranscriptome analysis with low input materials. PLoS Biol 16:e2006092
Dominissini D, Moshitch-Moshkovitz S, Salmon-Divon M et al (2013) Transcriptome-wide mapping of n(6)-methyladenosine by m(6)a-seq based on immunocapturing and massively parallel sequencing. Nat Protoc 8:176–189
Zhu Z, Xing X, Huang S et al (2021) Nat10 promotes osteogenic differentiation of mesenchymal stem cells by mediating n4-acetylcytidine modification of gremlin 1. Stem Cells Int 2021:8833527
Zhao W, Zhou Y, Cui Q et al (2019) Paces: Prediction of n4-acetylcytidine (ac4c) modification sites in mrna. Sci Rep 9:11112
Alam W, Tayara H, Chong KT (2020) Xg-ac4c: Identification of n4-acetylcytidine (ac4c) in mrna using extreme gradient boosting with electron-ion interaction pseudopotentials. Sci Rep 10:20942
Cui W, Pizzollo J, Han Z et al (2016) Nop2 is required for mammalian preimplantation development. Mol Reprod Dev 83:124–131
Wang H, Wang L, Wang Z et al (2020) The nucleolar protein nop2 is required for nucleolar maturation and ribosome biogenesis during preimplantation development in mammals. FASEB J 34:2715–2729
Ralston A, Cox BJ, Nishioka N et al (2010) Gata3 regulates trophoblast development downstream of tead4 and in parallel to cdx2. Development 137:395–403
Hansen CG, Moroishi T, Guan KL (2015) Yap and taz: a nexus for hippo signaling and beyond. Trends Cell Biol 25:499–513
Yagi R, Kohn MJ, Karavanova I et al (2007) Transcription factor tead4 specifies the trophectoderm lineage at the beginning of mammalian development. Development 134:3827–3836
Hong B, Brockenbrough JS, Wu P et al (1997) Nop2p is required for pre-rrna processing and 60s ribosome subunit synthesis in yeast. Mol Cell Biol 17:378–388
Klinge S, Woolford JL Jr (2019) Ribosome assembly coming into focus. Nat Rev Mol Cell Biol 20:116–131
Ralston A, Rossant J (2008) Cdx2 acts downstream of cell polarization to cell-autonomously promote trophectoderm fate in the early mouse embryo. Dev Biol 313:614–629
Niwa H, Toyooka Y, Shimosato D et al (2005) Interaction between oct3/4 and cdx2 determines trophectoderm differentiation. Cell 123:917–929
Jedrusik A, Parfitt DE, Guo G et al (2008) Role of cdx2 and cell polarity in cell allocation and specification of trophectoderm and inner cell mass in the mouse embryo. Genes Dev 22:2692–2706
Funding
This work was supported by grants from the National Natural Science Foundation of China (31771601, U20A20376, 61972116) and the Applied Technology Research and Development Project of Heilongjiang (GA20C018).
Author information
Authors and Affiliations
Contributions
Conceptualization, MYW and QW; Methodology, HJH and ZBH; Validation, ZBH; Formal Analysis, RC and YZ; Investigation, MYW, HJH and QW; Writing—Original Draft, MYW, RC and HJH; Writing—Review & Editing, MYW, HJH, YZ and QW; Visualization, MYW and RC; Funding Acquisition, YZ and QW. All authors discussed the results and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of Interest
The authors declare that they have no competing interests.
Ethical approval
All operations on experimental animals were carried out in accordance with the Guide for the Care and Use of Laboratory Animals from the Harbin Institute of Technology (HIT) and approved by the Institutional Animal Care and Use Committee or Animal Experimental Ethics Committee of HIT (IACUC-2023001).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, M., Cheng, R., He, H. et al. N4-acetylcytidine of Nop2 mRNA is required for the transition of morula-to-blastocyst. Cell. Mol. Life Sci. 80, 307 (2023). https://doi.org/10.1007/s00018-023-04955-w
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04955-w