Abstract
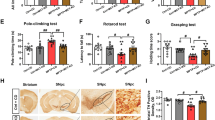
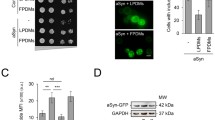
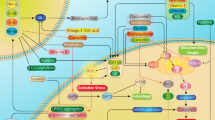
Parkinson’s disease (PD) is a progressive movement disorder characterized by dopaminergic (DA) neuron degeneration and the existence of Lewy bodies formed by misfolded α-synuclein. Emerging evidence supports the benefits of dietary interventions in PD due to their safety and practicality. Previously, dietary intake of α-ketoglutarate (AKG) was proved to extend the lifespan of various species and protect mice from frailty. However, the mechanism of dietary AKG’s effects in PD remains undetermined. In the present study, we report that an AKG-based diet significantly ameliorated α-synuclein pathology, and rescued DA neuron degeneration and impaired DA synapses in adeno-associated virus (AAV)-loaded human α-synuclein mice and transgenic A53T α-synuclein (A53T α-Syn) mice. Moreover, AKG diet increased nigral docosahexaenoic acid (DHA) levels and DHA supplementation reproduced the anti-α-synuclein effects in the PD mouse model. Our study reveals that AKG and DHA induced microglia to phagocytose and degrade α-synuclein via promoting C1q and suppressed pro-inflammatory reactions. Furthermore, results indicate that modulating gut polyunsaturated fatty acid metabolism and microbiota Lachnospiraceae_NK4A136_group in the gut-brain axis may underlie AKG’s benefits in treating α-synucleinopathy in mice. Together, our findings propose that dietary intake of AKG is a feasible and promising therapeutic approach for PD.











Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are present in the paper and/or the supplementary files, and the RNA-seq data can be publicly found at the Gene Expression Omnibus database under accession number GSE214446. Requests for any materials in this study should be directed to Yunlong Zhang and obtained through an MTA.
References
Kalia LV, Lang AE (2015) Parkinson’s disease. Lancet 386(9996):896–912
Samii A, Nutt JG, Ransom BR (2004) Parkinson’s disease. Lancet 363(9423):1783–1793
Armstrong MJ, Okun MS (2020) Diagnosis and treatment of Parkinson disease: a review. JAMA 323(6):548–560
Fox SH, Lang AE (2008) Levodopa-related motor complications–phenomenology. Mov Disord 23(Suppl 3):S509-514
Petzinger GM, Fisher BE, McEwen S, Beeler JA, Walsh JP, Jakowec MW (2013) Exercise-enhanced neuroplasticity targeting motor and cognitive circuitry in Parkinson’s disease. Lancet Neurol 12(7):716–726
Domenighetti C, Sugier PE, Ashok Kumar Sreelatha A, Schulte C, Grover S, Mohamed O et al (2022) Dairy intake and Parkinson’s disease: a Mendelian randomization study. Mov Disord 37(4):857–864
Tan AH, Lim SY, Chong KK, Manap MAAA, Hor JW, Lim JL et al (2021) Probiotics for constipation in Parkinson disease: a randomized placebo-controlled study. Neurology 96(5):e772–e782
Goya ME, Xue F, Sampedro-Torres-Quevedo C, Arnaouteli S, Riquelme-Dominguez L, Romanowski A et al (2020) Probiotic bacillus subtilis protects against alpha-synuclein aggregation in C. elegans. Cell Rep 30(2):367-380 e367
Maraki MI, Yannakoulia M, Stamelou M, Stefanis L, Xiromerisiou G, Kosmidis MH et al (2019) Mediterranean diet adherence is related to reduced probability of prodromal Parkinson’s disease. Mov Disord 34(1):48–57
Phillips MCL, Murtagh DKJ, Gilbertson LJ, Asztely FJS, Lynch CDP (2018) Low-fat versus ketogenic diet in Parkinson’s disease: a pilot randomized controlled trial. Mov Disord 33(8):1306–1314
Chin RM, Fu X, Pai MY, Vergnes L, Hwang H, Deng G et al (2014) The metabolite alpha-ketoglutarate extends lifespan by inhibiting ATP synthase and TOR. Nature 510(7505):397–401
Su Y, Wang T, Wu N, Li D, Fan X, Xu Z et al (2019) Alpha-ketoglutarate extends drosophila lifespan by inhibiting mTOR and activating AMPK. Aging (Albany NY) 11(12):4183–4197
Burdyliuk N, Bayliak M (2017) Effects of long-term cultivation on medium with alpha-ketoglutarate supplementation on metabolic processes of Saccharomyces cerevisiae. J Aging Res 2017:8754879
Zhang Z, He C, Gao Y, Zhang L, Song Y, Zhu T et al (2021) Alpha-ketoglutarate delays age-related fertility decline in mammals. Aging Cell 20(2):e13291
Asadi Shahmirzadi A, Edgar D, Liao CY, Hsu YM, Lucanic M, Asadi Shahmirzadi A et al (2020) Alpha-ketoglutarate, an endogenous metabolite, extends lifespan and compresses morbidity in aging mice. Cell Metab 32(3):447-456 e446
An D, Zeng Q, Zhang P, Ma Z, Zhang H, Liu Z et al (2021) Alpha-ketoglutarate ameliorates pressure overload-induced chronic cardiac dysfunction in mice. Redox Biol 46:102088
Wang Y, Deng P, Liu Y, Wu Y, Chen Y, Guo Y et al (2020) Alpha-ketoglutarate ameliorates age-related osteoporosis via regulating histone methylations. Nat Commun 11(1):5596
Salminen A, Kauppinen A, Hiltunen M, Kaarniranta K (2014) Krebs cycle intermediates regulate DNA and histone methylation: epigenetic impact on the aging process. Ageing Res Rev 16:45–65
Volpicelli-Daley LA, Luk KC, Lee VM (2014) Addition of exogenous alpha-synuclein preformed fibrils to primary neuronal cultures to seed recruitment of endogenous alpha-synuclein to lewy body and lewy neurite-like aggregates. Nat Protoc 9(9):2135–2146
Zhang W, Ding L, Chen H, Zhang M, Ma R, Zheng S et al (2023) Cntnap4 partial deficiency exacerbates alpha-synuclein pathology through astrocyte-microglia c3–c3ar pathway. Cell Death Dis 14(4):285
Polinski NK, Volpicelli-Daley LA, Sortwell CE, Luk KC, Cremades N, Gottler LM et al (2018) Best practices for generating and using alpha-synuclein pre-formed fibrils to model Parkinson’s disease in rodents. J Parkinsons Dis 8(2):303–322
Zhou C, Zhong W, Zhou J, Sheng F, Fang Z, Wei Y et al (2012) Monitoring autophagic flux by an improved tandem fluorescent-tagged lc3 (mtagrfp-mwasabi-lc3) reveals that high-dose rapamycin impairs autophagic flux in cancer cells. Autophagy 8(8):1215–1226
Giasson BI, Duda JE, Quinn SM, Zhang B, Trojanowski JQ, Lee VM (2002) Neuronal alpha-synucleinopathy with severe movement disorder in mice expressing a53t human alpha-synuclein. Neuron 34(4):521–533
Liu JH, Wang Q, You QL, Li ZL, Hu NY, Wang Y et al (2020) Acute EPA-induced learning and memory impairment in mice is prevented by DHA. Nat Commun 11(1):5465
Gong J, Zhang W, Ding L, Zhang M, Zheng S, Ma R et al (2021) 4,4’-dimethoxychalcone regulates redox homeostasis by targeting riboflavin metabolism in Parkinson’s disease therapy. Free Radic Biol Med 174:40–56
Faustini G, Longhena F, Varanita T, Bubacco L, Pizzi M, Missale C et al (2018) Synapsin III deficiency hampers alpha-synuclein aggregation, striatal synaptic damage and nigral cell loss in an AAV-based mouse model of Parkinson’s disease. Acta Neuropathol 136(4):621–639
Zhang W, Chen H, Ding L, Gong J, Zhang M, Guo W et al (2021) Trojan horse delivery of 4,4’-dimethoxychalcone for parkinsonian neuroprotection. Adv Sci (Weinh) 8(9):2004555
Zhang W, Zhou M, Lu W, Gong J, Gao F, Li Y et al (2020) Cntnap4 deficiency in dopaminergic neurons initiates parkinsonian phenotypes. Theranostics 10(7):3000–3021
Zhang W, Huang J, Gao F, You Q, Ding L, Gong J et al (2022) Lactobacillus reuteri normalizes altered fear memory in male Cntnap4 knockout mice. EBioMedicine 86:104323
Zhang M, Chen H, Zhang W, Liu Y, Ding L, Gong J et al (2023) Biomimetic remodeling of microglial riboflavin metabolism ameliorates cognitive impairment by modulating neuroinflammation. Adv Sci (Weinh). https://doi.org/10.1002/advs.202300180
Chang PK, Khatchadourian A, McKinney RA, Maysinger D (2015) Docosahexaenoic acid (DHA): a modulator of microglia activity and dendritic spine morphology. J Neuroinflammation 12:34
Chitre NM, Wood BJ, Ray A, Moniri NH, Murnane KS (2020) Docosahexaenoic acid protects motor function and increases dopamine synthesis in a rat model of Parkinson’s disease via mechanisms associated with increased protein kinase activity in the striatum. Neuropharmacology 167:107976
Serrano-Garcia N, Fernandez-Valverde F, Luis-Garcia ER, Granados-Rojas L, Juarez-Zepeda TE, Orozco-Suarez SA et al (2018) Docosahexaenoic acid protection in a rotenone induced Parkinson’s model: prevention of tubulin and synaptophysin loss, but no association with mitochondrial function. Neurochem Int 121:26–37
Holden SS, Grandi FC, Aboubakr O, Higashikubo B, Cho FS, Chang AH et al (2021) Complement factor c1q mediates sleep spindle loss and epileptic spikes after mild brain injury. Science 373(6560):eabj2685
Webster SD, Galvan MD, Ferran E, Garzon-Rodriguez W, Glabe CG, Tenner AJ (2001) Antibody-mediated phagocytosis of the amyloid beta-peptide in microglia is differentially modulated by c1q. J Immunol 166(12):7496–7503
Webster SD, Park M, Fonseca MI, Tenner AJ (2000) Structural and functional evidence for microglial expression of C1qr(p), the c1q receptor that enhances phagocytosis. J Leukoc Biol 67(1):109–116
Webster SD, Yang AJ, Margol L, Garzon-Rodriguez W, Glabe CG, Tenner AJ (2000) Complement component C1q modulates the phagocytosis of abeta by microglia. Exp Neurol 161(1):127–138
Choi I, Zhang Y, Seegobin SP, Pruvost M, Wang Q, Purtell K et al (2020) Microglia clear neuron-released alpha-synuclein via selective autophagy and prevent neurodegeneration. Nat Commun 11(1):1386
Cao S, Theodore S, Standaert DG (2010) Fcgamma receptors are required for nf-kappab signaling, microglial activation and dopaminergic neurodegeneration in an AAV-synuclein mouse model of Parkinson’s disease. Mol Neurodegener 5:42
Sampson TR, Debelius JW, Thron T, Janssen S, Shastri GG, Ilhan ZE et al (2016) Gut microbiota regulate motor deficits and neuroinflammation in a model of Parkinson’s disease. Cell 167(6):1469-1480 e1412
Sampson TR, Challis C, Jain N, Moiseyenko A, Ladinsky MS, Shastri GG et al (2020) A gut bacterial amyloid promotes alpha-synuclein aggregation and motor impairment in mice. Elife. https://doi.org/10.7554/eLife.53111
He L, Xu Z, Yao K, Wu G, Yin Y, Nyachoti CM et al (2015) The physiological basis and nutritional function of alpha-ketoglutarate. Curr Protein Pept Sci 16(7):576–581
Hong S, Beja-Glasser VF, Nfonoyim BM, Frouin A, Li S, Ramakrishnan S et al (2016) Complement and microglia mediate early synapse loss in Alzheimer mouse models. Science 352(6286):712–716
Absinta M, Maric D, Gharagozloo M, Garton T, Smith MD, Jin J et al (2021) A lymphocyte-microglia-astrocyte axis in chronic active multiple sclerosis. Nature 597(7878):709–714
Pisalyaput K, Tenner AJ (2008) Complement component C1q inhibits beta-amyloid- and serum amyloid p-induced neurotoxicity via caspase- and calpain-independent mechanisms. J Neurochem 104(3):696–707
Fonseca MI, Chu SH, Hernandez MX, Fang MJ, Modarresi L, Selvan P et al (2017) Cell-specific deletion of C1qa identifies microglia as the dominant source of C1q in mouse brain. J Neuroinflammation 14(1):48
Nemani VM, Lu W, Berge V, Nakamura K, Onoa B, Lee MK et al (2010) Increased expression of alpha-synuclein reduces neurotransmitter release by inhibiting synaptic vesicle reclustering after endocytosis. Neuron 65(1):66–79
Plotegher N, Berti G, Ferrari E, Tessari I, Zanetti M, Lunelli L et al (2017) Dopal derived alpha-synuclein oligomers impair synaptic vesicles physiological function. Sci Rep 7:40699
Chen MK, Kuwabara H, Zhou Y, Adams RJ, Brasic JR, McGlothan JL et al (2008) VMAT2 and dopamine neuron loss in a primate model of Parkinson’s disease. J Neurochem 105(1):78–90
Makrides M, Gibson RA, McPhee AJ, Yelland L, Quinlivan J, Ryan P et al (2010) Effect of DHA supplementation during pregnancy on maternal depression and neurodevelopment of young children: a randomized controlled trial. JAMA 304(15):1675–1683
Borsini A, Nicolaou A, Camacho-Munoz D, Kendall AC, Di Benedetto MG, Giacobbe J et al (2021) Omega-3 polyunsaturated fatty acids protect against inflammation through production of LOX and CYP450 lipid mediators: relevance for major depression and for human hippocampal neurogenesis. Mol Psychiatry 26(11):6773–6788
Patrick RP (2019) Role of phosphatidylcholine-DHA in preventing APOE4-associated Alzheimer’s disease. FASEB J 33(2):1554–1564
Qiao Y, Mei Y, Han H, Liu F, Yang XM, Shao Y et al (2018) Effects of omega-3 in the treatment of violent schizophrenia patients. Schizophr Res 195:283–285
Coulombe K, Kerdiles O, Tremblay C, Emond V, Lebel M, Boulianne AS et al (2018) Impact of DHA intake in a mouse model of synucleinopathy. Exp Neurol 301(Pt A):39–49
Lamontagne-Proulx J, Coulombe K, Dahhani F, Cote M, Guyaz C, Tremblay C et al (2021) Effect of docosahexaenoic acid (DHA) at the enteric level in a synucleinopathy mouse model. Nutrients. https://doi.org/10.3390/nu13124218
Coulombe K, Saint-Pierre M, Cisbani G, St-Amour I, Gibrat C, Giguere-Rancourt A et al (2016) Partial neurorescue effects of dha following a 6-ohda lesion of the mouse dopaminergic system. J Nutr Biochem 30:133–142
Hernando S, Requejo C, Herran E, Ruiz-Ortega JA, Morera-Herreras T, Lafuente JV et al (2019) Beneficial effects of n-3 polyunsaturated fatty acids administration in a partial lesion model of Parkinson’s disease: the role of glia and NRf2 regulation. Neurobiol Dis 121:252–262
Bradbury J (2011) Docosahexaenoic acid (DHA): an ancient nutrient for the modern human brain. Nutrients 3(5):529–554
Liu R, Chen L, Wang Z, Zheng X, Hou Z, Zhao D et al (2021) Omega-3 polyunsaturated fatty acids prevent obesity by improving tricarboxylic acid cycle homeostasis. J Nutr Biochem 88:108503
Bahety P, Tan YM, Hong Y, Zhang L, Chan EC, Ee PL (2014) Metabotyping of docosahexaenoic acid—treated Alzheimer’s disease cell model. PLoS ONE 9(2):e90123
Machiels K, Joossens M, Sabino J, De Preter V, Arijs I, Eeckhaut V et al (2014) A decrease of the butyrate-producing species Roseburia hominis and Faecalibacterium prausnitzii defines dysbiosis in patients with ulcerative colitis. Gut 63(8):1275–1283
Kasahara K, Krautkramer KA, Org E, Romano KA, Kerby RL, Vivas EI et al (2018) Interactions between Roseburia intestinalis and diet modulate atherogenesis in a murine model. Nat Microbiol 3(12):1461–1471
Tylichova Z, Slavik J, Ciganek M, Ovesna P, Krcmar P, Strakova N et al (2018) Butyrate and docosahexaenoic acid interact in alterations of specific lipid classes in differentiating colon cancer cells. J Cell Biochem 119(6):4664–4679
Kolar S, Barhoumi R, Jones CK, Wesley J, Lupton JR, Fan YY et al (2011) Interactive effects of fatty acid and butyrate-induced mitochondrial ca(2)(+) loading and apoptosis in colonocytes. Cancer 117(23):5294–5303
Qiao CM, Sun MF, Jia XB, Li Y, Zhang BP, Zhao LP et al (2020) Sodium butyrate exacerbates Parkinson’s disease by aggravating neuroinflammation and colonic inflammation in MPTP-induced mice model. Neurochem Res 45(9):2128–2142
Erny D, Hrabe de Angelis AL, Jaitin D, Wieghofer P, Staszewski O, David E et al (2015) Host microbiota constantly control maturation and function of microglia in the CNS. Nat Neurosci 18(7):965–977
Keshavarzian A, Green SJ, Engen PA, Voigt RM, Naqib A, Forsyth CB et al (2015) Colonic bacterial composition in Parkinson’s disease. Mov Disord 30(10):1351–1360
Segain JP, Raingeard de la Bletiere D, Bourreille A, Leray V, Gervois N, Rosales C et al (2000) Butyrate inhibits inflammatory responses through nfkappab inhibition: implications for Crohn’s disease. Gut 47(3):397–403
Guo TT, Zhang Z, Sun Y, Zhu RY, Wang FX, Ma LJ et al (2023) Neuroprotective effects of sodium butyrate by restoring gut microbiota and inhibiting TLR4 signaling in mice with MPTP-induced Parkinson’s disease. Nutrients. https://doi.org/10.3390/nu15040930
O’Donovan SM, Crowley EK, Brown JR, O’Sullivan O, O’Leary OF, Timmons S et al (2020) Nigral overexpression of alpha-synuclein in a rat Parkinson’s disease model indicates alterations in the enteric nervous system and the gut microbiome. Neurogastroenterol Motil 32(1):e13726
Lawson JA, Patrono C, Ciabattoni G, Fitzgerald GA (1986) Long-lived enzymatic metabolites of thromboxane b2 in the human circulation. Anal Biochem 155(1):198–205
Kiefer JR, Pawlitz JL, Moreland KT, Stegeman RA, Hood WF, Gierse JK et al (2000) Structural insights into the stereochemistry of the cyclooxygenase reaction. Nature 405(6782):97–101
Hammarstrom S (1983) Leukotrienes. Annu Rev Biochem 52:355–377
Ohmura T, Tian Y, Sarich N, Ke Y, Meliton A, Shah AS et al (2017) Regulation of lung endothelial permeability and inflammatory responses by prostaglandin a2: role of ep4 receptor. Mol Biol Cell 28(12):1622–1635
Zhou W, Zhang J, Goleniewska K, Dulek DE, Toki S, Newcomb DC et al (2016) Prostaglandin i2 suppresses proinflammatory chemokine expression, cd4 t cell activation, and stat6-independent allergic lung inflammation. J Immunol 197(5):1577–1586
Ma L, Ni Y, Wang Z, Tu W, Ni L, Zhuge F et al (2020) Spermidine improves gut barrier integrity and gut microbiota function in diet-induced obese mice. Gut Microbes 12(1):1–19
Xia T, Duan W, Zhang Z, Li S, Zhao Y, Geng B et al (2021) Polyphenol-rich vinegar extract regulates intestinal microbiota and immunity and prevents alcohol-induced inflammation in mice. Food Res Int 140:110064
Sheng K, Yang J, Xu Y, Kong X, Wang J, Wang Y (2022) Alleviation effects of grape seed proanthocyanidin extract on inflammation and oxidative stress in ad-galactose-induced aging mouse model by modulating the gut microbiota. Food Funct 13(3):1348–1359
Donati L, Ziegler F, Pongelli G, Signorini MS (1999) Nutritional and clinical efficacy of ornithine alpha-ketoglutarate in severe burn patients. Clin Nutr 18(5):307–311
Coudray-Lucas C, Le Bever H, Cynober L, De Bandt JP, Carsin H (2000) Ornithine alpha-ketoglutarate improves wound healing in severe burn patients: a prospective randomized double-blind trial versus isonitrogenous controls. Crit Care Med 28(6):1772–1776
Demidenko O, Barardo D, Budovskii V, Finnemore R, Palmer FR, Kennedy BK et al (2021) Rejuvant(r), a potential life-extending compound formulation with alpha-ketoglutarate and vitamins, conferred an average 8 year reduction in biological aging, after an average of 7 months of use, in the truage DNA methylation test. Aging (Albany NY) 13(22):24485–24499
Gyanwali B, Lim ZX, Soh J, Lim C, Guan SP, Goh J et al (2022) Alpha-ketoglutarate dietary supplementation to improve health in humans. Trends Endocrinol Metab 33(2):136–146
Cynober L, Coudray-Lucas C, de Bandt JP, Guechot J, Aussel C, Salvucci M et al (1990) Action of ornithine alpha-ketoglutarate, ornithine hydrochloride, and calcium alpha-ketoglutarate on plasma amino acid and hormonal patterns in healthy subjects. J Am Coll Nutr 9(1):2–12
Filip RS, Pierzynowski SG, Lindegard B, Wernerman J, Haratym-Maj A, Podgurniak M (2007) Alpha-ketoglutarate decreases serum levels of c-terminal cross-linking telopeptide of type I collagen (CTX) in postmenopausal women with osteopenia: six-month study. Int J Vitam Nutr Res 77(2):89–97
De Bandt JP, Coudray-Lucas C, Lioret N, Lim SK, Saizy R, Giboudeau J et al (1998) A randomized controlled trial of the influence of the mode of enteral ornithine alpha-ketoglutarate administration in burn patients. J Nutr 128(3):563–569
Funding
This work was supported by the National Natural Science Foundation of China (No. 82174468 to Y.L.Z., No. 81870856, 81870992, 82071416 to P.Y.X., No. 82101325 to W.L.Z.), the Science and Technology Planning Project of Guangzhou (No. 201904010238 to Y.L.Z.), Guangzhou Medical University Discipline Construction Funds (Basic Medicine, No. JCXKJS2022A09 to Y.L.Z.), Central government guiding local science and technology development projects (No. ZYYD2022C17 to P.Y.X.), Key Research and Development Program of Guangzhou (No. 2023B03J0631 to P.Y.X.), Municipal University (Faculty) joint funding project (No. 202102010010 to P.Y.X.), Guangdong Basic and Applied Basic Research Foundation (No. 2022B1515230004 to P.Y.X.), and the China Postdoctoral Science Foundation (No. 2021M700951 to W.L.Z.).
Author information
Authors and Affiliations
Contributions
YLZ designed the research. WLZ and MRZ performed the Western blotting. LYD and RFM carried out the immunostaining assays. JWG and SHZ injected the AAVs and performed the behavioral tests. HXM prepared and provided the human PFF. YLZ, WLZ, LYD and MRZ analyzed the data. YLZ wrote the manuscript. PYX and HXX discussed the manuscript. All authors read and commented on it.
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
The animal study was approved by the guidelines of the Institutional Animal Care and Use Committee of the Guangzhou Medical University and in line with the National Institutes of Health guidelines on the care and use of animals (NIH Publications No. 8023, revised 1978).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, W., Ding, L., Zhang, M. et al. Dietary intake of α-ketoglutarate ameliorates α-synuclein pathology in mouse models of Parkinson’s disease. Cell. Mol. Life Sci. 80, 155 (2023). https://doi.org/10.1007/s00018-023-04807-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04807-7