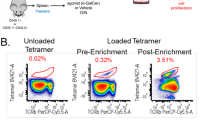
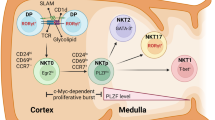
Abstract
Invariant natural killer T (iNKT) cells correspond to a population of thymus-generated T cells with innate-like characteristics and effector functions. Among the various iNKT subsets, NKT17 is the only subset that produces the proinflammatory cytokine IL-17. But, how NKT17 cells acquire this ability and what would selectively trigger their activation remain incompletely understood. Here, we identified the cytokine receptor DR3 being specifically expressed on thymic NKT17 cells and mostly absent on other thymic iNKT subsets. Moreover, DR3 ligation promoted the in vivo activation of thymic NKT17 cells and provided costimulatory effects upon agonistic α-GalCer stimulation. Thus, we identified a specific surface marker for thymic NKT17 cells that triggers their activation and augments their effector functions both in vivo and in vitro. These findings provide new insights for deciphering the role and function of murine NKT17 cells and for understanding the development and activation mechanisms of iNKT cells in general.




Similar content being viewed by others
Data availability
All data generated or analyzed during this study are included in this published article and its supplementary information files.
Code availability
Not applicable.
References
Bendelac A, Savage PB, Teyton L (2007) The biology of NKT cells. Annu Rev Immunol 25:297–336. https://doi.org/10.1146/annurev.immunol.25.022106.141711
Crosby CM, Kronenberg M (2018) Tissue-specific functions of invariant natural killer T cells. Nat Rev Immunol 18:559–574. https://doi.org/10.1038/s41577-018-0034-2
Lee YJ, Holzapfel KL, Zhu J, Jameson SC, Hogquist KA (2013) Steady-state production of IL-4 modulates immunity in mouse strains and is determined by lineage diversity of iNKT cells. Nat Immunol 14:1146–1154. https://doi.org/10.1038/ni.2731
Dai H et al (2015) Syndecan-1 identifies and controls the frequency of IL-17-producing naive natural killer T (NKT17) cells in mice. Eur J Immunol 45:3045–3051. https://doi.org/10.1002/eji.201545532
Luo S, Kwon J, Crossman A, Park PW, Park JH (2021) CD138 expression is a molecular signature but not a developmental requirement for RORgammat+ NKT17 cells. JCI Insight. https://doi.org/10.1172/jci.insight.148038
Tsagaratou A (2019) Unveiling the regulation of NKT17 cell differentiation and function. Mol Immunol 105:55–61. https://doi.org/10.1016/j.molimm.2018.11.013
Lee YJ et al (2015) Tissue-specific distribution of iNKT cells impacts their cytokine response. Immunity 43:566–578. https://doi.org/10.1016/j.immuni.2015.06.025
Wang H, Hogquist KA (2018) CCR7 defines a precursor for murine iNKT cells in thymus and periphery. Elife. https://doi.org/10.7554/eLife.34793
Bittner S, Ehrenschwender M (2017) Multifaceted death receptor 3 signaling-promoting survival and triggering death. FEBS Lett 591:2543–2555. https://doi.org/10.1002/1873-3468.12747
Alexander CM et al (2000) Syndecan-1 is required for Wnt-1-induced mammary tumorigenesis in mice. Nat Genet 25:329–332. https://doi.org/10.1038/77108
Ligons DL et al (2018) RORgammat limits the amount of the cytokine receptor gammac through the prosurvival factor Bcl-xL in developing thymocytes. Sci Signal. https://doi.org/10.1126/scisignal.aam8939
Lahl K et al (2007) Selective depletion of Foxp3+ regulatory T cells induces a scurfy-like disease. J Exp Med 204:57–63. https://doi.org/10.1084/jem.20061852
Moran AE et al (2011) T cell receptor signal strength in Treg and iNKT cell development demonstrated by a novel fluorescent reporter mouse. J Exp Med 208:1279–1289. https://doi.org/10.1084/jem.20110308
Park JY et al (2019) CD24(+) cell depletion permits effective enrichment of thymic iNKT cells while preserving their subset composition. Immune Netw 19:e14. https://doi.org/10.4110/in.2019.19.e14
Park JY, DiPalma DT, Kwon J, Fink J, Park JH (2019) Quantitative difference in PLZF protein expression determines iNKT lineage fate and controls innate CD8 T cell generation. Cell Rep 27:2548-2557 e2544. https://doi.org/10.1016/j.celrep.2019.05.012
Schreiber TH et al (2010) Therapeutic treg expansion in mice by TNFRSF25 prevents allergic lung inflammation. J Clin Invest 120:3629–3640. https://doi.org/10.1172/JCI42933
Park JY et al (2022) In vivo availability of the cytokine IL-7 constrains the survival and homeostasis of peripheral iNKT cells. Cell Rep 38:110219. https://doi.org/10.1016/j.celrep.2021.110219
Meylan F, Richard AC, Siegel RM (2011) TL1A and DR3, a TNF family ligand-receptor pair that promotes lymphocyte costimulation, mucosal hyperplasia, and autoimmune inflammation. Immunol Rev 244:188–196. https://doi.org/10.1111/j.1600-065X.2011.01068.x
Nishikii H et al (2016) DR3 signaling modulates the function of Foxp3+ regulatory T cells and the severity of acute graft-versus-host disease. Blood 128:2846–2858. https://doi.org/10.1182/blood-2016-06-723783
Valatas V, Kolios G, Bamias G (2019) TL1A (TNFSF15) and DR3 (TNFRSF25): a co-stimulatory system of cytokines with diverse functions in gut mucosal immunity. Front Immunol 10:583. https://doi.org/10.3389/fimmu.2019.00583
Kim JM, Rasmussen JP, Rudensky AY (2007) Regulatory T cells prevent catastrophic autoimmunity throughout the lifespan of mice. Nat Immunol 8:191–197. https://doi.org/10.1038/ni1428
Rangarajan S et al (2020) Heparanase-enhanced shedding of syndecan-1 and its role in driving disease pathogenesis and progression. J Histochem Cytochem 68:823–840. https://doi.org/10.1369/0022155420937087
Georgiev H, Ravens I, Benarafa C, Forster R, Bernhardt G (2016) Distinct gene expression patterns correlate with developmental and functional traits of iNKT subsets. Nat Commun 7:13116. https://doi.org/10.1038/ncomms13116
Won HY et al (2021) The timing and abundance of IL-2Rbeta (CD122) expression control thymic iNKT cell generation and NKT1 subset differentiation. Front Immunol 12:642856. https://doi.org/10.3389/fimmu.2021.642856
Ziegler SF, Ramsdell F, Alderson MR (1994) The activation antigen CD69. Stem Cells 12:456–465. https://doi.org/10.1002/stem.5530120502
Madireddi S et al (2017) Regulatory T cell-mediated suppression of inflammation induced by DR3 signaling is dependent on galectin-9. J Immunol 199:2721–2728. https://doi.org/10.4049/jimmunol.1700575
Uldrich AP et al (2005) NKT cell stimulation with glycolipid antigen in vivo: costimulation-dependent expansion, Bim-dependent contraction, and hyporesponsiveness to further antigenic challenge. J Immunol 175:3092–3101. https://doi.org/10.4049/jimmunol.175.5.3092
Wang J et al (2009) Cutting edge: CD28 engagement releases antigen-activated invariant NKT cells from the inhibitory effects of PD-1. J Immunol 182:6644–6647. https://doi.org/10.4049/jimmunol.0804050
Kim DH et al (2008) 4–1BB engagement costimulates NKT cell activation and exacerbates NKT cell ligand-induced airway hyperresponsiveness and inflammation. J Immunol 180:2062–2068. https://doi.org/10.4049/jimmunol.180.4.2062
Pellicci DG et al (2003) Intrathymic NKT cell development is blocked by the presence of alpha-galactosylceramide. Eur J Immunol 33:1816–1823. https://doi.org/10.1002/eji.200323894
Rossjohn J, Pellicci DG, Patel O, Gapin L, Godfrey DI (2012) Recognition of CD1d-restricted antigens by natural killer T cells. Nat Rev Immunol 12:845–857. https://doi.org/10.1038/nri3328
Cameron G et al (2015) Antigen specificity of type I NKT cells is governed by TCR beta-chain diversity. J Immunol 195:4604–4614. https://doi.org/10.4049/jimmunol.1501222
Dickgreber N et al (2012) Immature murine NKT cells pass through a stage of developmentally programmed innate IL-4 secretion. J Leukoc Biol 92:999–1009. https://doi.org/10.1189/jlb.0512242
Watarai H et al (2012) Development and function of invariant natural killer T cells producing T(h)2- and T(h)17-cytokines. PLoS Biol 10:e1001255. https://doi.org/10.1371/journal.pbio.1001255
Acknowledgements
We thank Joo-Young Park (Seoul National University) for the critical review of the manuscript. We are also grateful for members of the EIB FACS core facility for their help and expertise in flow cytometry data acquisition and analyses.
Funding
This study has been supported by the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research.
Author information
Authors and Affiliations
Contributions
SL and NL: designed and performed the experiments, analyzed the data, and contributed to the writing of the manuscript. CL and AC: performed experiments, analyzed the data, and commented on the manuscript. EW and FM: provided reagents, experimental expertise, and edited the manuscript. JP: conceived the project, analyzed the data, and wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All animal procedures reported in this study that were performed by NCI-CCR affiliated staff were approved by the NCI Animal Care and Use Committee (ACUC) and in accordance with federal regulatory requirements and standards. All components of the intramural NIH ACU program are accredited by AAALAC International. All mice were cared for in accordance with the Public Health Service policy on human care and use of laboratory animals and NIH guidelines.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Luo, S., Liman, N., Li, C. et al. The cytokine receptor DR3 identifies and promotes the activation of thymic NKT17 cells. Cell. Mol. Life Sci. 80, 76 (2023). https://doi.org/10.1007/s00018-023-04726-7
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-023-04726-7