Abstract
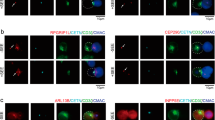
The ZAP70 protein tyrosine kinase (PTK) couples stimulated T cell antigen receptors (TCRs) to their downstream signal transduction pathways and is sine qua non for T cell activation and differentiation. TCR engagement leads to activation-induced post-translational modifications of ZAP70, predominantly by kinases, which modulate its conformation, leading to activation of its catalytic domain. Here, we demonstrate that ZAP70 in TCR/CD3-activated mouse spleen and thymus cells, as well as human Jurkat T cells, is regulated by the peptidyl-prolyl cis–trans isomerase (PPIase), cyclophilin A (CypA) and that this regulation is abrogated by cyclosporin A (CsA), a CypA inhibitor. We found that TCR crosslinking promoted a rapid and transient, Lck-dependent association of CypA with the interdomain B region, at the ZAP70 regulatory domain. CsA inhibited CypA binding to ZAP70 and prevented the colocalization of CypA and ZAP70 at the cell membrane. In addition, imaging analyses of antigen-specific T cells stimulated by MHC-restricted antigen-fed antigen-presenting cells revealed the recruitment of ZAP70-bound CypA to the immunological synapse. Enzymatically active CypA downregulated the catalytic activity of ZAP70 in vitro, an effect that was reversed by CsA in TCR/CD3-activated normal T cells but not in CypA-deficient T cells, and further confirmed in vivo by FRET-based studies. We suggest that CypA plays a role in determining the activity of ZAP70 in TCR-engaged T cells and impact on T cell activation by intervening with the activity of multiple downstream effector molecules.






Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author on reasonable request.
Abbreviations
- C3G:
-
CrkII binding of guanine-nucleotide releasing factor
- c-Cbl:
-
Casitas B-lineage lymphoma
- cfb3:
-
Cytoplasmic fragment of human erythrocyte band 3
- CrkII:
-
CT10 regulator of kinase II
- CypA:
-
Cyclophilin A
- CsA:
-
Cyclosporin A
- FRET:
-
Fluorescence resonance energy transfer
- HRP:
-
Horseradish peroxidase
- IP3 :
-
Inositol trisphosphate
- IS:
-
Immunological synapse
- ITAM:
-
Immunoreceptor tyrosine-based activation motifs
- Itk:
-
Interleukin-2-inducible T cell kinase
- JNK:
-
Jun N-terminal kinase
- LAT:
-
Linker of activated T cells
- Lck:
-
Lymphocyte-specific protein tyrosine kinase
- MAPK:
-
Mitogen-activated protein kinase
- NFAT:
-
Nuclear factor of activated T cells
- Pin1:
-
Peptidyl-prolyl cis–trans isomerase NIMA-interacting 1
- PKCα:
-
Protein kinase C α
- PLC:
-
Phospholipase C
- PPIase:
-
Peptidyl-prolyl cis–trans isomerases
- SCID:
-
Severe combined immunodeficiency
- Sts1:
-
Suppressor of TCR signaling 1
- TCR:
-
T cell antigen receptor
- WCL:
-
Whole-cell lysates
- ZAP70:
-
ζ-Chain-associated protein of 70 kDa
References
Chan AC, Irving BA, Fraser JD, Weiss A (1991) The zeta chain is associated with a tyrosine kinase and upon T-cell antigen receptor stimulation associates with ZAP-70, a 70-kDa tyrosine phosphoprotein. Proc Natl Acad Sci USA 88(20):9166–9170
Arpaia E, Shahar M, Dadi H, Cohen A, Roifman CM (1994) Defective T cell receptor signaling and CD8+ thymic selection in humans lacking zap-70 kinase. Cell 76(5):947–958
Chan AC, Kadlecek TA, Elder ME, Filipovich AH, Kuo WL, Iwashima M et al (1994) ZAP-70 deficiency in an autosomal recessive form of severe combined immunodeficiency. Science 264(5165):1599–1601
Elder ME, Lin D, Clever J, Chan AC, Hope TJ, Weiss A et al (1994) Human severe combined immunodeficiency due to a defect in ZAP-70, a T cell tyrosine kinase. Science 264(5165):1596–1599
Negishi I, Motoyama N, Nakayama K, Nakayama K, Senju S, Hatakeyama S et al (1995) Essential role for ZAP-70 in both positive and negative selection of thymocytes. Nature 376(6539):435–438
Wiest DL, Ashe JM, Howcroft TK, Lee HM, Kemper DM, Negishi I et al (1997) A spontaneously arising mutation in the DLAARN motif of murine ZAP-70 abrogates kinase activity and arrests thymocyte development. Immunity 6(6):663–671
Barber EK, Dasgupta JD, Schlossman SF, Trevillyan JM, Rudd CE (1989) The CD4 and CD8 antigens are coupled to a protein-tyrosine kinase (p56lck) that phosphorylates the CD3 complex. Proc Natl Acad Sci USA 86(9):3277–3281
Fasbender F, Claus M, Wingert S, Sandusky M, Watzl C (2017) Differential requirements for Src-family kinases in SYK or ZAP70-mediated SLP-76 phosphorylation in lymphocytes. Front Immunol 8:789
van Oers NS, Killeen N, Weiss A (1996) Lck regulates the tyrosine phosphorylation of the T cell receptor subunits and ZAP-70 in murine thymocytes. J Exp Med 183(3):1053–1062
Thill PA, Weiss A, Chakraborty AK (2016) Phosphorylation of a tyrosine residue on Zap70 by Lck and its subsequent binding via an SH2 domain may be a key gatekeeper of T cell receptor signaling in vivo. Mol Cell Biol 36(18):2396–2402
Duplay P, Thome M, Herve F, Acuto O (1994) p56lck interacts via its src homology 2 domain with the ZAP-70 kinase. J Exp Med 179(4):1163–1172
Watts JD, Affolter M, Krebs DL, Wange RL, Samelson LE, Aebersold R (1994) Identification by electrospray ionization mass spectrometry of the sites of tyrosine phosphorylation induced in activated Jurkat T cells on the protein tyrosine kinase ZAP-70. J Biol Chem 269(47):29520–29529
Chan AC, Dalton M, Johnson R, Kong GH, Wang T, Thoma R et al (1995) Activation of ZAP-70 kinase activity by phosphorylation of tyrosine 493 is required for lymphocyte antigen receptor function. EMBO J 14(11):2499–2508
Kong G, Dalton M, Bubeck Wardenburg J, Straus D, Kurosaki T, Chan AC (1996) Distinct tyrosine phosphorylation sites in ZAP-70 mediate activation and negative regulation of antigen receptor function. Mol Cell Biol 16(9):5026–5035
Zhang W, Sloan-Lancaster J, Kitchen J, Trible RP, Samelson LE (1998) LAT: the ZAP-70 tyrosine kinase substrate that links T cell receptor to cellular activation. Cell 92(1):83–92
Bubeck Wardenburg J, Fu C, Jackman JK, Flotow H, Wilkinson SE, Williams DH et al (1996) Phosphorylation of SLP-76 by the ZAP-70 protein-tyrosine kinase is required for T-cell receptor function. J Biol Chem 271(33):19641–19644
Salvador JM, Mittelstadt PR, Guszczynski T, Copeland TD, Yamaguchi H, Appella E et al (2005) Alternative p38 activation pathway mediated by T cell receptor-proximal tyrosine kinases. Nat Immunol 6(4):390–395
Au-Yeung BB, Shah NH, Shen L, Weiss A (2018) ZAP-70 in signaling, biology, and disease. Annu Rev Immunol 36:127–156
Wange RL, Guitian R, Isakov N, Watts JD, Aebersold R, Samelson LE (1995) Activating and inhibitory mutations in adjacent tyrosines in the kinase domain of ZAP-70. J Biol Chem 270(32):18730–18733
Rao N, Lupher ML Jr, Ota S, Reedquist KA, Druker BJ, Band H (2000) The linker phosphorylation site Tyr292 mediates the negative regulatory effect of Cbl on ZAP-70 in T cells. J Immunol 164(9):4616–4626
Zhao Q, Weiss A (1996) Enhancement of lymphocyte responsiveness by a gain-of-function mutation of ZAP-70. Mol Cell Biol 16(12):6765–6774
Yan Q, Barros T, Visperas PR, Deindl S, Kadlecek TA, Weiss A et al (2013) Structural basis for activation of ZAP-70 by phosphorylation of the SH2-kinase linker. Mol Cell Biol 33(11):2188–2201
Neumeister EN, Zhu Y, Richard S, Terhorst C, Chan AC, Shaw AS (1995) Binding of ZAP-70 to phosphorylated T-cell receptor zeta and eta enhances its autophosphorylation and generates specific binding sites for SH2 domain-containing proteins. Mol Cell Biol 15(6):3171–3178
Katzav S, Sutherland M, Packham G, Yi T, Weiss A (1994) The protein tyrosine kinase ZAP-70 can associate with the SH2 domain of proto-Vav. J Biol Chem 269(51):32579–32585
Plas DR, Johnson R, Pingel JT, Matthews RJ, Dalton M, Roy G et al (1996) Direct regulation of ZAP-70 by SHP-1 in T cell antigen receptor signaling. Science 272(5265):1173–1176
Pacini S, Ulivieri C, Di Somma MM, Isacchi A, Lanfrancone L, Pelicci PG et al (1998) Tyrosine 474 of ZAP-70 is required for association with the Shc adaptor and for T-cell antigen receptor-dependent gene activation. J Biol Chem 273(32):20487–20493
Gelkop S, Isakov N (1999) T cell activation stimulates the association of enzymatically active tyrosine-phosphorylated ZAP-70 with the Crk adapter proteins. J Biol Chem 274(31):21519–21527
Pelosi M, Di Bartolo V, Mounier V, Mege D, Pascussi JM, Dufour E et al (1999) Tyrosine 319 in the interdomain B of ZAP-70 is a binding site for the Src homology 2 domain of Lck. J Biol Chem 274(20):14229–14237
Wu J, Zhao Q, Kurosaki T, Weiss A (1997) The Vav binding site (Y315) in ZAP-70 is critical for antigen receptor-mediated signal transduction. J Exp Med 185(10):1877–1882
Gelkop S, Gish GD, Babichev Y, Pawson T, Isakov N (2005) T cell activation-induced CrkII binding to the Zap70 protein tyrosine kinase is mediated by Lck-dependent phosphorylation of Zap70 tyrosine 315. J Immunol 175(12):8123–8132
Dong G, Kalifa R, Nath PR, Gelkop S, Isakov N (2017) TCR crosslinking promotes Crk adaptor protein binding to tyrosine-phosphorylated CD3zeta chain. Biochem Biophys Res Commun 488(3):541–546
Xie J, Han X, Zhao C, Canonigo-Balancio AJ, Yates JR, 3rd, Li Y, et al (2019) Phosphotyrosine-dependent interaction between the kinases PKCtheta and Zap70 promotes proximal TCR signaling. Sci Signal 12(577).
Lupher ML Jr, Songyang Z, Shoelson SE, Cantley LC, Band H (1997) The Cbl phosphotyrosine-binding domain selects a D(N/D)XpY motif and binds to the Tyr292 negative regulatory phosphorylation site of ZAP-70. J Biol Chem 272(52):33140–33144
Meng W, Sawasdikosol S, Burakoff SJ, Eck MJ (1999) Structure of the amino-terminal domain of Cbl complexed to its binding site on ZAP-70 kinase. Nature 398(6722):84–90
Yang M, Chen T, Li X, Yu Z, Tang S, Wang C et al (2015) K33-linked polyubiquitination of Zap70 by Nrdp1 controls CD8(+) T cell activation. Nat Immunol 16(12):1253–1262
Ivanova E, Carpino N (2016) Negative regulation of TCR signaling by ubiquitination of Zap-70 Lys-217. Mol Immunol 73:19–28
Naik E, Dixit VM (2016) Usp9X is required for lymphocyte activation and homeostasis through its control of ZAP70 Ubiquitination and PKCbeta kinase activity. J Immunol 196(8):3438–3451
Hu H, Wang H, Xiao Y, Jin J, Chang JH, Zou Q et al (2016) Otud7b facilitates T cell activation and inflammatory responses by regulating Zap70 ubiquitination. J Exp Med 213(3):399–414
Thurm C, Poltorak MP, Reimer E, Brinkmann MM, Leichert L, Schraven B et al (2017) A highly conserved redox-active Mx(2)CWx(6)R motif regulates Zap70 stability and activity. Oncotarget 8(19):30805–30816
Isakov N, Biesinger B (2000) Lck protein tyrosine kinase is a key regulator of T-cell activation and a target for signal intervention by Herpesvirus saimiri and other viral gene products. Eur J Biochem 267(12):3413–3421
Brazin KN, Mallis RJ, Fulton DB, Andreotti AH (2002) Regulation of the tyrosine kinase Itk by the peptidyl-prolyl isomerase cyclophilin A. Proc Natl Acad Sci USA 99(4):1899–1904
Li L, Guo X, Shi X, Li C, Wu W, Yan C et al (2017) Ionic CD3-Lck interaction regulates the initiation of T-cell receptor signaling. Proc Natl Acad Sci USA 114(29):E5891–E5899
Isakov N, Wange RL, Burgess WH, Watts JD, Aebersold R, Samelson LE (1995) ZAP-70 binding specificity to T cell receptor tyrosine-based activation motifs: the tandem SH2 domains of ZAP-70 bind distinct tyrosine-based activation motifs with varying affinity. J Exp Med 181(1):375–380
Abrahamsen H, O’Neill AK, Kannan N, Kruse N, Taylor SS, Jennings PA et al (2012) Peptidyl-prolyl isomerase Pin1 controls down-regulation of conventional protein kinase C isozymes. J Biol Chem 287(16):13262–13278
Isakov N, Wange RL, Watts JD, Aebersold R, Samelson LE (1996) Purification and characterization of human ZAP-70 protein-tyrosine kinase from a baculovirus expression system. J Biol Chem 271(26):15753–15761
Walsh CT, Zydowsky LD, McKeon FD (1992) Cyclosporin A, the cyclophilin class of peptidylprolyl isomerases, and blockade of T cell signal transduction. J Biol Chem 267(19):13115–13118
Steinmann B, Bruckner P, Superti-Furga A (1991) Cyclosporin A slows collagen triple-helix formation in vivo: indirect evidence for a physiologic role of peptidyl-prolyl cis-trans-isomerase. J Biol Chem 266(2):1299–1303
Cadra S, Gucciardi A, Valignat MP, Theodoly O, Vacaflores A, Houtman JC et al (2015) ROZA-XL, an improved FRET based biosensor with an increased dynamic range for visualizing zeta associated protein 70 kD (ZAP-70) tyrosine kinase activity in live T cells. Biochem Biophys Res Commun 459(3):405–410
Fischer G, Wittmann-Liebold B, Lang K, Kiefhaber T, Schmid FX (1989) Cyclophilin and peptidyl-prolyl cis-trans isomerase are probably identical proteins. Nature 337(6206):476–478
Isakov N, Mally MI, Scholz W, Altman A (1987) T-lymphocyte activation: the role of protein kinase C and the bifurcating inositol phospholipid signal transduction pathway. Immunol Rev 95:89–111
McCaffrey PG, Perrino BA, Soderling TR, Rao A (1993) NF-ATp, a T lymphocyte DNA-binding protein that is a target for calcineurin and immunosuppressive drugs. J Biol Chem 268(5):3747–3752
Rooney JW, Sun YL, Glimcher LH, Hoey T (1995) Novel NFAT sites that mediate activation of the interleukin-2 promoter in response to T-cell receptor stimulation. Mol Cell Biol 15(11):6299–6310
Hermann-Kleiter N, Baier G (2010) NFAT pulls the strings during CD4+ T helper cell effector functions. Blood 115(15):2989–2997
Cardenas ME, Hemenway C, Muir RS, Ye R, Fiorentino D, Heitman J (1994) Immunophilins interact with calcineurin in the absence of exogenous immunosuppressive ligands. EMBO J 13(24):5944–5957
Liu J, Farmer JD Jr, Lane WS, Friedman J, Weissman I, Schreiber SL (1991) Calcineurin is a common target of cyclophilin-cyclosporin A and FKBP-FK506 complexes. Cell 66(4):807–815
Letourneau S, Krieg C, Pantaleo G, Boyman O (2009) IL-2- and CD25-dependent immunoregulatory mechanisms in the homeostasis of T-cell subsets. J Allergy Clin Immunol 123(4):758–762
Matsuda S, Shibasaki F, Takehana K, Mori H, Nishida E, Koyasu S (2000) Two distinct action mechanisms of immunophilin-ligand complexes for the blockade of T-cell activation. EMBO Rep 1(5):428–434
Giardino Torchia ML, Dutta D, Mittelstadt PR, Guha J, Gaida MM, Fish K et al (2018) Intensity and duration of TCR signaling is limited by p38 phosphorylation of ZAP-70(T293) and destabilization of the signalosome. Proc Natl Acad Sci USA 115(9):2174–2179
Liao XC, Littman DR (1995) Altered T cell receptor signaling and disrupted T cell development in mice lacking Itk. Immunity 3(6):757–769
Schaeffer EM, Debnath J, Yap G, McVicar D, Liao XC, Littman DR et al (1999) Requirement for Tec kinases Rlk and Itk in T cell receptor signaling and immunity. Science 284(5414):638–641
Tsoukas CD, Grasis JA, Ching KA, Kawakami Y, Kawakami T (2001) Itk/Emt: a link between T cell antigen receptor-mediated Ca2+ events and cytoskeletal reorganization. Trends Immunol 22(1):17–20
Colgan J, Asmal M, Neagu M, Yu B, Schneidkraut J, Lee Y et al (2004) Cyclophilin A regulates TCR signal strength in CD4+ T cells via a proline-directed conformational switch in Itk. Immunity 21(2):189–201
Sarkar P, Reichman C, Saleh T, Birge RB, Kalodimos CG (2007) Proline cis-trans isomerization controls autoinhibition of a signaling protein. Mol Cell 25(3):413–426
Sarkar P, Saleh T, Tzeng SR, Birge RB, Kalodimos CG (2011) Structural basis for regulation of the Crk signaling protein by a proline switch. Nat Chem Biol 7(1):51–57
Nath PR, Dong G, Braiman A, Isakov N (2014) Immunophilins control T lymphocyte adhesion and migration by regulating CrkII binding to C3G. J Immunol 193(8):3966–3977
Nath PR, Dong G, Braiman A, Isakov N (2016) In vivo regulation of human CrkII by cyclophilin A and FK506-binding protein. Biochem Biophys Res Commun 470(2):411–416
Lo WL, Shah NH, Ahsan N, Horkova V, Stepanek O, Salomon AR et al (2018) Lck promotes Zap70-dependent LAT phosphorylation by bridging Zap70 to LAT. Nat Immunol 19(7):733–741
Liu J, Albers MW, Wandless TJ, Luan S, Alberg DG, Belshaw PJ et al (1992) Inhibition of T cell signaling by immunophilin-ligand complexes correlates with loss of calcineurin phosphatase activity. Biochemistry 31(16):3896–3901
Dutta D, Barr VA, Akpan I, Mittelstadt PR, Singha LI, Samelson LE et al (2017) Recruitment of calcineurin to the TCR positively regulates T cell activation. Nat Immunol 18(2):196–204
Pandya P, Pasvolsky R, Babichev Y, Braiman A, Witte S, Altman A et al (2019) PICOT binding to the polycomb group protein, EED, alters H3K27 methylation at the MYT1 PRC2 target gene. Biochem Biophys Res Commun 509(2):469–475
Wange RL, Malek SN, Desiderio S, Samelson LE (1993) Tandem SH2 domains of ZAP-70 bind to T cell antigen receptor zeta and CD3 epsilon from activated Jurkat T cells. J Biol Chem 268(26):19797–19801
Lin TY, Emerman M (2006) Cyclophilin A interacts with diverse lentiviral capsids. Retrovirology 3:70
Bolte S, Cordelieres FP (2006) A guided tour into subcellular colocalization analysis in light microscopy. J Microsc 224(Pt 3):213–232
Villalba M, Coudronniere N, Deckert M, Teixeiro E, Mas P, Altman A (2000) A novel functional interaction between Vav and PKCtheta is required for TCR-induced T cell activation. Immunity 12(2):151–160
Acknowledgements
We thank Ms. Margalit Krup for technical assistance, Ms. Caroline Simon, and Ms. Judith Isakov for editorial assistance, and Drs. Joseph B. Bolen, Michael Emerman, Annemarie Lellouch, and Tony Pawson, for their gifts of reagents. The following reagent was obtained through the NIH HIV Reagent Program, Division of AIDS, NIAID, NIH: Jurkat T-Cells CypA -/-, ARP-10095, contributed by Drs. D. Braaten and J. Luban.
Funding
This work was funded in part by the Israel Science Foundation Grants No. 1235/17 (NI) and 2368/19 (EL), the USA-Israel Binational Science Foundation Grant No. 2013034 (NI), the Jacki and Bruce Barron Cancer Research Scholars’ Program, a partnership between the Israel Cancer Research Fund (ICRF) and the City of Hope (Grant No. 87735611 (NI)), postdoctoral fellowships provided by the Planning and Budgeting Committee (PBC) of the Israel Council for Higher Education (AKA and PRN) and doctoral fellowships provided by the Kreitman School of Advanced Graduate Studies, Ben-Gurion University of the Negev (NPA).
Author information
Authors and Affiliations
Contributions
NI conceived the study, designed experiments, and supervised the project; NPA, AKA, AM, JS, and PRN carried out the experiments; ZS, AB, and NI analyzed the data; EL provided essential tools for the study; NPA, AKA, and NI wrote the manuscript. All authors commented on the previous versions of the manuscript. All authors have read and approved the final manuscript and have given consent for publishing the work.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no relevant financial or non-financial interests to disclose.
Ethics approval
This study was approved in advance by the Ben-Gurion University Institutional Animal Care and Use Committee and conducted in accordance with the Israeli Animal Welfare Act following the guidelines of the Guide for Care and Use of Laboratory Animals (National Research Council 1996). The Animal ethical clearance protocol used for this research is IL-44–08-2017.
Consent to participate
Not Applicable as human subjects were not involved in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Anto, N.P., Arya, A.K., Muraleedharan, A. et al. Cyclophilin A associates with and regulates the activity of ZAP70 in TCR/CD3-stimulated T cells. Cell. Mol. Life Sci. 80, 7 (2023). https://doi.org/10.1007/s00018-022-04657-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04657-9