Abstract
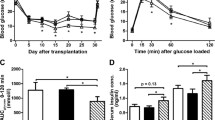
Early apoptosis of grafted islets is one of the main factors affecting the efficacy of islet transplantation. The combined transplantation of islet cells and bone marrow mesenchymal stem cells (BMSCs) can significantly improve the survival rate of grafted islets. Transcription factor insulin gene enhancer binding protein 1 (ISL1) is shown to promote the angiogenesis of grafted islets and the paracrine function of mesenchymal stem cells during the co-transplantation, yet the regulatory mechanism remains unclear. By using ISL1-overexpressing BMSCs and the subtherapeutic doses of islets for co-transplantation, we managed to reduce the apoptosis and improve the survival rate of the grafts. Our metabolomics and proteomics data suggested that ISL1 upregulates aniline (ANLN) and Inhibin beta A chain (INHBA), and stimulated the release of caffeine in the BMSCs. We then demonstrated that the upregulation of ANLN and INHBA was achieved by the binding of ISL1 to the promoter regions of the two genes. In addition, ISL1 could also promote BMSCs to release exosomes with high expression of ANLN, secrete INHBA and caffeine, and reduce streptozocin (STZ)-induced islets apoptosis. Thus, our study provides mechanical insight into the islet/BMSCs co-transplantation and paves the foundation for using conditioned medium to mimic the ISL1-overexpressing BMSCs co-transplantation.
Graphical abstract








Similar content being viewed by others
Data and materials availability
All data associated with this study are present in the paper and/or the Supplementary Materials.
References
Rickels MR, Robertson RP (2019) Pancreatic islet transplantation in humans: recent progress and future directions. Endocr Rev 40(2):631–668. https://doi.org/10.1210/er.2018-00154
Roep BO (2020) Improving clinical islet transplantation outcomes. Diabetes Care 43(4):698–700. https://doi.org/10.2337/dci19-0080
Shapiro AM, Pokrywczynska M, Ricordi C (2017) Clinical pancreatic islet transplantation. Nat Rev Endocrinol 13(5):268–277. https://doi.org/10.1038/nrendo.2016.178
Sakata N, Goto M, Yoshimatsu G, Egawa S, Unno M (2011) Utility of co-transplanting mesenchymal stem cells in islet transplantation. World J Gastroenterol 17(47):5150–5155. https://doi.org/10.3748/wjg.v17.i47.5150
Nalbach L, Roma LP, Schmitt BM et al (2021) Improvement of islet transplantation by the fusion of islet cells with functional blood vessels. EMBO Mol Med 13(1):e12616. https://doi.org/10.15252/emmm.202012616
Shin JY, Jeong JH, Jin H et al (2015) Transplantation of heterospheroids of islet cells and mesenchymal stem cells for effective angiogenesis and anti-apoptosis. Tissue Eng Part A 21(5–6):1024–1035
Ito T, Itakura S, Todorov I et al (2010) Mesenchymal stem cell and islet co-transplantation promotes graft revascularization and function. Transplantation 89(12):1438–1445. https://doi.org/10.1097/tp.0b013e3181db09c4
Cunha JP, Leuckx G, Sterkendries P et al (2017) Human multipotent adult progenitor cells enhance islet function and revascularisation when co-transplanted as a composite pellet in a mouse model of diabetes. Diabetologia 60(1):134–142. https://doi.org/10.1007/s00125-016-4120-3
Hirabaru M, Kuroki T, Adachi T et al (2015) A method for performing islet transplantation using tissue-engineered sheets of islets and mesenchymal stem cells. Tissue Eng Part C Methods 21(12):1205–1215. https://doi.org/10.1089/ten.TEC.2015.0035
Wang Y, Wang JW, Li Y et al (2021) Bone marrow-derived mesenchymal stem cells improve rat islet graft revascularization by upregulating ISL1. Stem Cells 39(8):1033–1048. https://doi.org/10.1002/stem.3378
Chen J, Chen J, Cheng Y et al (2020) Mesenchymal stem cell-derived exosomes protect beta cells against hypoxia-induced apoptosis via miR-21 by alleviating ER stress and inhibiting p38 MAPK phosphorylation. Stem Cell Res Ther 11(1):97. https://doi.org/10.1186/s13287-020-01610-0
Wang L, Qing L, Liu H et al (2017) Mesenchymal stromal cells ameliorate oxidative stress-induced islet endothelium apoptosis and functional impairment via Wnt4-beta-catenin signaling. Stem Cell Res Ther 8(1):188. https://doi.org/10.1186/s13287-017-0640-0
Keshtkar S, Kaviani M, Jabbarpour Z et al (2020) Hypoxia-preconditioned Wharton’s Jelly-derived mesenchymal stem cells mitigate stress-induced apoptosis and ameliorate human islet survival and function in direct contact coculture system. Stem Cells Int 2020:8857457. https://doi.org/10.1155/2020/8857457
He Y, Zhang D, Zeng Y et al (2018) Bone marrow-derived mesenchymal stem cells protect islet grafts against endoplasmic reticulum stress-induced apoptosis during the early stage after transplantation. Stem Cells 36(7):1045–1061. https://doi.org/10.1002/stem.2823
Ding Y, Xu D, Feng G, Bushell A, Muschel RJ, Wood KJ (2009) Mesenchymal stem cells prevent the rejection of fully allogenic islet grafts by the immunosuppressive activity of matrix metalloproteinase-2 and -9. Diabetes 58(8):1797–1806. https://doi.org/10.2337/db09-0317
Mohammadi Ayenehdeh J, Niknam B, Rasouli S et al (2017) Immunomodulatory and protective effects of adipose tissue-derived mesenchymal stem cells in an allograft islet composite transplantation for experimental autoimmune type 1 diabetes. Immunol Lett 188:21–31. https://doi.org/10.1016/j.imlet.2017.05.006
Ben Nasr M, Vergani A, Avruch J et al (2015) Co-transplantation of autologous MSCs delays islet allograft rejection and generates a local immunoprivileged site. Acta Diabetol 52(5):917–927. https://doi.org/10.1007/s00592-015-0735-y
Melzi R, Antonioli B, Mercalli A et al (2010) Co-graft of allogeneic immune regulatory neural stem cells (NPC) and pancreatic islets mediates tolerance, while inducing NPC-derived tumors in mice. PLoS ONE 5(4):e10357. https://doi.org/10.1371/journal.pone.0010357
Rackham CL, Amisten S, Persaud SJ, King AJF, Jones PM (2018) Mesenchymal stromal cell secretory factors induce sustained improvements in islet function pre- and post-transplantation. Cytotherapy 20(12):1427–1436. https://doi.org/10.1016/j.jcyt.2018.07.007
Arzouni AA, Vargas-Seymour A, Rackham CL et al (2017) Mesenchymal stromal cells improve human islet function through released products and extracellular matrix. Clin Sci (Lond) 131(23):2835–2845. https://doi.org/10.1042/CS20171251
Song L, Gou W, Wang J et al (2021) Overexpression of alpha-1 antitrypsin in mesenchymal stromal cells improves their intrinsic biological properties and therapeutic effects in nonobese diabetic mice. Stem Cells Transl Med 10:320–331. https://doi.org/10.1002/sctm.20-0122
Liu W, Li L, Rong Y et al (2020) Hypoxic mesenchymal stem cell-derived exosomes promote bone fracture healing by the transfer of miR-126. Acta Biomater 103:196–212. https://doi.org/10.1016/j.actbio.2019.12.020
Brandhorst H, Brandhorst D, Abraham A et al (2020) Proteomic profiling reveals the ambivalent character of the mesenchymal stem cell secretome: assessing the effect of preconditioned media on isolated human islets. Cell Transplant 29:963689720952332. https://doi.org/10.1177/0963689720952332
Guo T, Wang W, Zhang H et al (2011) ISL1 promotes pancreatic islet cell proliferation. PLoS ONE 6(8):e22387. https://doi.org/10.1371/journal.pone.0022387
Li L, Sun F, Chen X, Zhang M (2018) ISL1 is upregulated in breast cancer and promotes cell proliferation, invasion, and angiogenesis. Onco Targets Ther 11:781–789. https://doi.org/10.2147/OTT.S144241
Gao R, Liang X, Cheedipudi S et al (2019) Pioneering function of Isl1 in the epigenetic control of cardiomyocyte cell fate. Cell Res 29(6):486–501. https://doi.org/10.1038/s41422-019-0168-1
Liu J, Li W, Wang Y et al (2014) Islet-1 overexpression in human mesenchymal stem cells promotes vascularization through monocyte chemoattractant protein-3. Stem Cells 32(7):1843–1854. https://doi.org/10.1002/stem.1682
Barzelay A, Hochhauser E, Entin-Meer M et al (2012) Islet-1 gene delivery improves myocardial performance after experimental infarction. Atherosclerosis 223(2):284–290. https://doi.org/10.1016/j.atherosclerosis.2012.05.025
Ricordi C, Gray DW, Hering BJ et al (1990) Islet isolation assessment in man and large animals. Acta Diabetol Lat 27:185–195
Xiang Q, Liao Y, Chao H et al (2018) ISL1 overexpression enhances the survival of transplanted human mesenchymal stem cells in a murine myocardial infarction model. Stem Cell Res Ther 9(1):51. https://doi.org/10.1186/s13287-018-0803-7
Akulenko N, Ryazansky S, Morgunova V, Komarov PA, Olovnikov I, Vaury C, Jensen S, Kalmykova A (2018) Transcriptional and chromatin changes accompanying de novo formation of transgenic piRNA clusters. RNA 24(4):574–584. https://doi.org/10.1261/rna.062851
Gong L, Zhang Y, Zhao J et al (2022) All-in-one biomimetic nanoplatform based on hollow polydopamine nanoparticles for synergistically enhanced radiotherapy of colon cancer. Small. https://doi.org/10.1002/smll.202107656
Zhang H, Zhu C, Sun Z et al (2019) Linderane protects pancreatic beta cells from streptozotocin (STZ)-induced oxidative damage. Life Sci 233:116732. https://doi.org/10.1016/j.lfs.2019.116732
Deng F, Xu Z, Zhou J, Zhang R, Gong X (2021) ANLN regulated by miR-30a-5p mediates malignant progression of lung adenocarcinoma. Comput Math Methods Med 2021:9549287. https://doi.org/10.1155/2021/9549287
Guo E, Mao X, Wang X et al (2021) Alternatively spliced ANLN isoforms synergistically contribute to the progression of head and neck squamous cell carcinoma. Cell Death Dis 12(8):764. https://doi.org/10.1038/s41419-021-04063-2
Qiu S, Li B, Xia Y et al (2022) CircTHBS1 drives gastric cancer progression by increasing INHBA mRNA expression and stability in a ceRNA- and RBP-dependent manner. Cell Death Dis 13(3):266. https://doi.org/10.1038/s41419-022-04720-0
Brown ML, Ungerleider N, Bonomi L, Andrzejewski D, Burnside A, Schneyer A (2014) Effects of activin A on survival, function and gene expression of pancreatic islets from non-diabetic and diabetic human donors. Islets 6(5–6):e1017226. https://doi.org/10.1080/19382014.2015.1017226
Szabat M, Johnson JD, Piret JM (2010) Reciprocal modulation of adult beta cell maturity by activin A and follistatin. Diabetologia 53(8):1680–1689. https://doi.org/10.1007/s00125-010-1758-0
Nakamura T, Takio K, Eto Y et al (1990) Activin-binding protein from rat ovary is follistatin. Science 247:836–838
Park S, Jang JS, Hong SM (2007) Long-term consumption of caffeine improves glucose homeostasis by enhancing insulinotropic action through islet insulin/insulin-like growth factor 1 signaling in diabetic rats. Metabolism 56(5):599–607. https://doi.org/10.1016/j.metabol.2006.12.004
Tan Y, Nie W, Chen C et al (2019) Mesenchymal stem cells alleviate hypoxia-induced oxidative stress and enhance the pro-survival pathways in porcine islets. Exp Biol Med (Maywood) 244(9):781–788. https://doi.org/10.1177/1535370219844472
Ishida N, Ishiyama K, Saeki Y, Tanaka Y, Ohdan H (2019) Cotransplantation of preactivated mesenchymal stem cells improves intraportal engraftment of islets by inhibiting liver natural killer cells in mice. Am J Transplant 19(10):2732–2745. https://doi.org/10.1111/ajt.15347
Pelizzo G, Avanzini MA, Folini M et al (2017) CPAM type 2-derived mesenchymal stem cells: malignancy risk study in a 14-month-old boy. Pediatr Pulmonol 52(8):990–999. https://doi.org/10.1002/ppul.23734
Bernardo ME, Zaffaroni N, Novara F et al (2007) Human bone marrow derived mesenchymal stem cells do not undergo transformation after long-term in vitro culture and do not exhibit telomere maintenance mechanisms. Cancer Res 67(19):9142–9149. https://doi.org/10.1158/0008-5472.CAN-06-4690
Chu DT, Phuong TNT, Tien NLB et al (2020) An update on the progress of isolation, culture, storage, and clinical application of human bone marrow mesenchymal stem/stromal cells. Int J Mol Sci. https://doi.org/10.3390/ijms21030708
Forte G, Pietronave S, Nardone G et al (2011) Human cardiac progenitor cell grafts as unrestricted source of supernumerary cardiac cells in healthy murine hearts. Stem Cells 29(12):2051–2061. https://doi.org/10.1002/stem.763
Pang SHM, D’Rozario J, Mendonca S et al (2021) Mesenchymal stromal cell apoptosis is required for their therapeutic function. Nat Commun 12(1):6495. https://doi.org/10.1038/s41467-021-26834-3
Yu SP, Wei Z, Wei L (2013) Preconditioning strategy in stem cell transplantation therapy. Transl Stroke Res 4(1):76–88. https://doi.org/10.1007/s12975-012-0251-0
Liu Q, Lv C, Huang Q et al (2022) ECM1 modified HF-MSCs targeting HSC attenuate liver cirrhosis by inhibiting the TGF-beta/Smad signaling pathway. Cell Death Discov 8(1):51. https://doi.org/10.1038/s41420-022-00846-4
Bao Y, Zhao Z, Gao H (2021) Effect of hTIMP-1 overexpression in human umbilical cord mesenchymal stem cells on the repair of pancreatic islets in type-1 diabetic mice. Cell Biol Int 45(5):1038–1049. https://doi.org/10.1002/cbin.11548
Xiong SQ, Jiang HB, Li YX et al (2016) Role of endogenous insulin gene enhancer protein ISL-1 in angiogenesis. Mol Vis 22:1375–1386
Zhang Y, Chen C, Liu Z et al (2022) PABPC1-induced stabilization of IFI27 mRNA promotes angiogenesis and malignant progression in esophageal squamous cell carcinoma through exosomal miRNA-21-5p. J Exp Clin Cancer Res 41(1):111. https://doi.org/10.1186/s13046-022-02339-9
Wang F, Xiang Z, Huang T, Zhang M, Zhou WB (2020) ANLN directly interacts with RhoA to promote doxorubicin resistance in breast cancer cells. Cancer Manag Res 12:9725–9734. https://doi.org/10.2147/CMAR.S261828
Hamalian S, Güth R, Runa F et al (2021) A SNAI2-PEAK1-INHBA stromal axis drives progression and lapatinib resistance in HER2-positive breast cancer by supporting subpopulations of tumor cells positive for antiapoptotic and stress signaling markers. Oncogene 40(33):5224–5235. https://doi.org/10.1038/s41388-021-01906-2
Zhao Z, Wang K, Tan S (2020) microRNA-211-mediated targeting of the INHBA-TGF-β axis suppresses prostate tumor formation and growth. Cancer Gene Ther 28(5):514–528. https://doi.org/10.1038/s41417-020-00237-w
Bao Y, Yao X, Li X et al (2021) INHBA transfection regulates proliferation, apoptosis and hormone synthesis in sheep granulosa cells. Theriogenology 175:111–122. https://doi.org/10.1016/j.theriogenology.2021.09.004
Brown ML, Kimura F, Bonomi LM, Ungerleider NA, Schneyer AL (2011) Differential synthesis and action of TGFss superfamily ligands in mouse and rat islets. Islets 3(6):367–375. https://doi.org/10.4161/isl.3.6.18013
Medina CB, Mehrotra P, Arandjelovic S et al (2020) Metabolites released from apoptotic cells act as tissue messengers. Nature 580(7801):130–135. https://doi.org/10.1038/s41586-020-2121-3
Jiang X, Zhang D, Jiang W (2014) Coffee and caffeine intake and incidence of type 2 diabetes mellitus: a meta-analysis of prospective studies. Eur J Nutr 53(1):25–38. https://doi.org/10.1007/s00394-013-0603-x
Tang X, Sun Y, Xu C et al (2021) Caffeine induces autophagy and apoptosis in auditory hair cells via the SGK1/HIF-1alpha pathway. Front Cell Dev Biol 9:751012. https://doi.org/10.3389/fcell.2021.751012
Ikram M, Park TJ, Ali T, Kim MO (2020) Antioxidant and neuroprotective effects of caffeine against Alzheimer’s and Parkinson’s disease: insight into the role of Nrf-2 and A2AR signaling. Antioxidants (Basel). https://doi.org/10.3390/antiox9090902
Shi CL (1997) Effects of caffeine and acetylcholine on glucose-stimulated insulin release from islet transplants in mice. Cell Transplant 6(1):33–37
Acknowledgements
The authors thank Bing Liu (BioBank, the First Affiliated Hospital of Xi’an Jiaotong University, Shaanxi Province, China) for technical help and revising the manuscript.
Funding
The project was supported by the National Natural Science Foundation of China (no. 81970670, 81970668, 82103213, and 82170768) and the Natural Science Foundation of Shaanxi Province (no. 2020JM-372).
Author information
Authors and Affiliations
Contributions
YW and X-MD developed the concept, designed experiments, and interpreted the data. YW, J-WZ, J-WW, J-LW, R-YM and S-CZ, conducted experiments and collected data. YW and J-WW analyzed data. YW, J-WZ, and X-MD prepared the manuscript. YW, P-JL, J-WZ, J-WW, J-LW, and X-MD made revisions to the manuscript. All the authors approved the final content. YW and X-MD are the guarantors of this work and, as such, had full access to all the data in this study and take responsibility for the integrity of the data and the accuracy of the data analysis.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interests.
Ethics approval
All animal procedures were approved by the Xi'an Jiaotong University Animal Experimentation Ethics Committee.
Consent for publication
N/A.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
18_2022_4571_MOESM1_ESM.docx
ANLN carrying exosome, INHBA, and caffeine reduce the apoptosis of grafted islets during BMSC overexpressing ISL1 co-transplantation, Supplementary Figures S1–5 and Tables S1–4. (DOCX 3541 KB)
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Zhang, JW., Wang, JW. et al. BMSCs overexpressed ISL1 reduces the apoptosis of islet cells through ANLN carrying exosome, INHBA, and caffeine. Cell. Mol. Life Sci. 79, 538 (2022). https://doi.org/10.1007/s00018-022-04571-0
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04571-0