Abstract
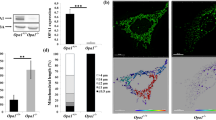
OPA1, a dynamin-related GTPase mutated in autosomal dominant optic atrophy, is essential for the fusion of the inner mitochondrial membrane. Although OPA1 deficiency leads to impaired mitochondrial morphology, the role of OPA1 in central carbon metabolism remains unclear. Here, we aim to explore the functional role and metabolic mechanism of OPA1 in cell fitness beyond the control of mitochondrial fusion. We applied [U-13C]glucose and [U-13C]glutamine isotope tracing techniques to OPA1-knockout (OPA1-KO) mouse embryonic fibroblasts (MEFs) compared to OPA1 wild-type (OPA1-WT) controls. Furthermore, the resulting tracing data were integrated by metabolic flux analysis to understand the underlying metabolic mechanism through which OPA1 deficiency reprograms cellular metabolism. OPA1-deficient MEFs were depleted of intracellular citrate, which was consistent with the decreased oxygen consumption rate in these cells with mitochondrial fission that is not balanced by mitochondrial fusion. Whereas oxidative glucose metabolism was impaired, OPA1-deficient cells activated glutamine-dependent reductive carboxylation and subsequently relied on this reductive metabolism to produce cytosolic citrate as a predominant acetyl-CoA source for de novo fatty acid synthesis. Prevention of cytosolic glutamine reductive carboxylation by GSK321, an inhibitor of isocitrate dehydrogenase 1 (IDH1), largely repressed lipid synthesis and blocked cell proliferation in OPA1-deficient MEFs. Our data support that, when glucose oxidation failed to support lipogenesis and proliferation in cells with unbalanced mitochondrial fission, OPA1 deficiency stimulated metabolic anaplerosis into glutamine-dependent reductive carboxylation in an IDH1-mediated manner.





Similar content being viewed by others
Availability of data and materials
All the metabolic alterations data generated by GC–MS are shown in Supplementary Information.
Abbreviations
- OPA1-WT:
-
OPA1 wild-type
- OPA1-KO:
-
OPA1-knockout
- MEFs:
-
Mouse embryonic fibroblasts
- MFA:
-
Metabolic flux analysis
- IDH1/2:
-
Isocitrate dehydrogenase 1/2
- MPC1/2:
-
Mitochondrial pyruvate carrier 1/2
- PDH:
-
Pyruvate dehydrogenase
- LDH:
-
Lactate dehydrogenase
- CTP:
-
Citrate transport protein
- DIC:
-
Mitochondrial dicarboxylate carrier
- FASN:
-
Fatty acid synthetase
- TFAM:
-
Mitochondrial transcription factor A
- MIDs:
-
Mass isotopologue distributions
- SSR:
-
Sum-of-squared residuals
- α-KG:
-
α-Ketoglutarate
- OXPHOS:
-
Oxidative phosphorylation
- OCR:
-
Oxygen consumption rate
References
Chan DC (2006) Mitochondrial fusion and fission in mammals. Annu Rev Cell Dev Biol 22:79–99. https://doi.org/10.1146/annurev.cellbio.22.010305.104638
Chen H, Chan DC (2017) Mitochondrial dynamics in regulating the unique phenotypes of cancer and stem cells. Cell Metab 26:39–48. https://doi.org/10.1016/j.cmet.2017.05.016
Labbe K, Murley A, Nunnari J (2014) Determinants and functions of mitochondrial behavior. Annu Rev Cell Dev Biol 30:357–391. https://doi.org/10.1146/annurev-cellbio-101011-155756
MacVicar T, Langer T (2016) OPA1 processing in cell death and disease - the long and short of it. J Cell Sci 129:2297–2306. https://doi.org/10.1242/jcs.159186
Song Z, Chen H, Fiket M, Alexander C, Chan DC (2007) OPA1 processing controls mitochondrial fusion and is regulated by mRNA splicing, membrane potential, and Yme1L. J Cell Biol 178:749–755. https://doi.org/10.1083/jcb.200704110
Lenaers G, Reynier P, Elachouri G, Soukkarieh C, Olichon A, Belenguer P, Baricault L, Ducommun B, Hamel C, Delettre C (2009) OPA1 functions in mitochondria and dysfunctions in optic nerve. Int J Biochem Cell Biol 41:1866–1874. https://doi.org/10.1016/j.biocel.2009.04.013
Del Dotto V, Mishra P, Vidoni S, Fogazza M, Maresca A, Caporali L, McCaffery JM, Cappelletti M, Baruffini E, Lenaers G et al (2017) OPA1 isoforms in the hierarchical organization of mitochondrial functions. Cell Rep 19:2557–2571. https://doi.org/10.1016/j.celrep.2017.05.073
Lee H, Smith SB, Yoon Y (2017) The short variant of the mitochondrial dynamin OPA1 maintains mitochondrial energetics and cristae structure. J Biol Chem 292:7115–7130. https://doi.org/10.1074/jbc.M116.762567
Bocca C, Kane MS, Veyrat-Durebex C, Chupin S, Alban J, KouassiNzoughet J, Le Mao M, Chao de la Barca JM, Amati-Bonneau P, Bonneau D et al (2018) The metabolomic bioenergetic signature of Opa1-disrupted mouse embryonic fibroblasts highlights aspartate deficiency. Sci Rep 8:11528. https://doi.org/10.1038/s41598-018-29972-9
Chen H, McCaffery JM, Chan DC (2007) Mitochondrial fusion protects against neurodegeneration in the cerebellum. Cell 130:548–562. https://doi.org/10.1016/j.cell.2007.06.026
Lee H, Smith SB, Sheu SS, Yoon Y (2020) The short variant of optic atrophy 1 (OPA1) improves cell survival under oxidative stress. J Biol Chem 295:6543–6560. https://doi.org/10.1074/jbc.RA119.010983
Gomes LC, Di Benedetto G, Scorrano L (2011) During autophagy mitochondria elongate, are spared from degradation and sustain cell viability. Nat Cell Biol 13:589–598. https://doi.org/10.1038/ncb2220
Chao de la Barca JM, Fogazza M, Rugolo M, Chupin S, Del Dotto V, Ghelli AM, Carelli V, Simard G, Procaccio V, Bonneau D et al (2020) Metabolomics hallmarks OPA1 variants correlating with their in vitro phenotype and predicting clinical severity. Hum Mol Genet 29:1319–1329. https://doi.org/10.1093/hmg/ddaa047
Chao de la Barca JM, Arrazola MS, Bocca C, Arnaune-Pelloquin L, Iuliano O, Tcherkez G, Lenaers G, Simard G, Belenguer P, Reynier P (2019) The metabolomic signature of Opa1 deficiency in rat primary cortical neurons shows aspartate/glutamate depletion and phospholipids remodeling. Sci Rep 9:6107. https://doi.org/10.1038/s41598-019-42554-7
Bocca C, Kane MS, Veyrat-Durebex C, Nzoughet JK, Chao de la Barca JM, Chupin S, Alban J, Procaccio V, Bonneau D, Simard G et al (2019) Lipidomics reveals triacylglycerol accumulation due to impaired fatty acid flux in Opa1-disrupted fibroblasts. J Proteome Res 18:2779–2790. https://doi.org/10.1021/acs.jproteome.9b00081
Wise DR, Ward PS, Shay JE, Cross JR, Gruber JJ, Sachdeva UM, Platt JM, DeMatteo RG, Simon MC, Thompson CB (2011) Hypoxia promotes isocitrate dehydrogenase-dependent carboxylation of alpha-ketoglutarate to citrate to support cell growth and viability. Proc Natl Acad Sci USA 108:19611–19616. https://doi.org/10.1073/pnas.1117773108
Metallo CM, Gameiro PA, Bell EL, Mattaini KR, Yang J, Hiller K, Jewell CM, Johnson ZR, Irvine DJ, Guarente L et al (2011) Reductive glutamine metabolism by IDH1 mediates lipogenesis under hypoxia. Nature 481:380–384. https://doi.org/10.1038/nature10602
Mullen AR, Wheaton WW, Jin ES, Chen PH, Sullivan LB, Cheng T, Yang Y, Linehan WM, Chandel NS, DeBerardinis RJ (2011) Reductive carboxylation supports growth in tumour cells with defective mitochondria. Nature 481:385–388. https://doi.org/10.1038/nature10642
Jiang L, Boufersaoui A, Yang C, Ko B, Rakheja D, Guevara G, Hu Z, DeBerardinis RJ (2017) Quantitative metabolic flux analysis reveals an unconventional pathway of fatty acid synthesis in cancer cells deficient for the mitochondrial citrate transport protein. Metab Eng 43:198–207. https://doi.org/10.1016/j.ymben.2016.11.004
Dai W, Wang G, Chwa J, Oh ME, Abeywardana T, Yang Y, Wang QA, Jiang L (2020) Mitochondrial division inhibitor (mdivi-1) decreases oxidative metabolism in cancer. Br J Cancer 122:1288–1297. https://doi.org/10.1038/s41416-020-0778-x
Jiang L, Shestov AA, Swain P, Yang C, Parker SJ, Wang QA, Terada LS, Adams ND, McCabe MT, Pietrak B et al (2016) Reductive carboxylation supports redox homeostasis during anchorage-independent growth. Nature 532:255–258. https://doi.org/10.1038/nature17393
Young JD (2014) INCA: a computational platform for isotopically non-stationary metabolic flux analysis. Bioinformatics (Oxford, England) 30:1333–1335. https://doi.org/10.1093/bioinformatics/btu015
Antoniewicz MR, Kelleher JK, Stephanopoulos G (2007) Elementary metabolite units (EMU): a novel framework for modeling isotopic distributions. Metab Eng 9:68–86. https://doi.org/10.1016/j.ymben.2006.09.001
Young JD, Walther JL, Antoniewicz MR, Yoo H, Stephanopoulos G (2008) An elementary metabolite unit (EMU) based method of isotopically nonstationary flux analysis. Biotechnol Bioeng 99:686–699. https://doi.org/10.1002/bit.21632
Du J, Yanagida A, Knight K, Engel AL, Vo AH, Jankowski C, Sadilek M, Tran VT, Manson MA, Ramakrishnan A et al (2016) Reductive carboxylation is a major metabolic pathway in the retinal pigment epithelium. Proc Natl Acad Sci USA 113:14710–14715. https://doi.org/10.1073/pnas.1604572113
Menendez JA, Lupu R (2007) Fatty acid synthase and the lipogenic phenotype in cancer pathogenesis. Nat Rev Cancer 7:763–777. https://doi.org/10.1038/nrc2222
Mishra P, Carelli V, Manfredi G, Chan DC (2014) Proteolytic cleavage of Opa1 stimulates mitochondrial inner membrane fusion and couples fusion to oxidative phosphorylation. Cell Metab 19:630–641. https://doi.org/10.1016/j.cmet.2014.03.011
Yang L, Tang H, Lin X, Wu Y, Zeng S, Pan Y, Li Y, Xiang G, Lin YF, Zhuang SM et al (2020) OPA1-Exon4b binds to mtDNA D-loop for transcriptional and metabolic modulation, independent of mitochondrial fusion. Front Cell Dev Biol 8:180. https://doi.org/10.3389/fcell.2020.00180
Demers-Lamarche J, Guillebaud G, Tlili M, Todkar K, Bélanger N, Grondin M, Nguyen AP, Michel J, Germain M (2016) Loss of mitochondrial function impairs lysosomes. J Biol Chem 291:10263–10276. https://doi.org/10.1074/jbc.M115.695825
Fendt SM, Bell EL, Keibler MA, Olenchock BA, Mayers JR, Wasylenko TM, Vokes NI, Guarente L, Vander Heiden MG, Stephanopoulos G (2013) Reductive glutamine metabolism is a function of the alpha-ketoglutarate to citrate ratio in cells. Nat Commun 4:2236. https://doi.org/10.1038/ncomms3236
Acknowledgements
We thank the Light Microscopy Core at City of Hope Medical Center for technical assistance.
Funding
The work was supported by Caltech-City of Hope Biomedical Research Initiative awarded to Lei Jiang, David Chan, and Qiong A. Wang, grant NIH R35 GM127147 to David Chan, and P30CA033572 to City of Hope. Qiong A. Wang was also supported by National Institutes of Health grants R01AG063854, R01HD096152, R01DK128907, and the American Diabetes Association Junior Faculty Development Award 1-19-JDF-023.
Author information
Authors and Affiliations
Contributions
WTD and LJ conceptualized the study, designed the experiments, interpreted the data, and revised the manuscript. WTD performed the experiments and wrote the original draft. ZCW helped with the INCA modeling analysis. QAW and DC helped revise the manuscript. LJ supervised the whole study. All authors approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dai, W., Wang, Z., Wang, Q.A. et al. Metabolic reprogramming in the OPA1-deficient cells. Cell. Mol. Life Sci. 79, 517 (2022). https://doi.org/10.1007/s00018-022-04542-5
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00018-022-04542-5