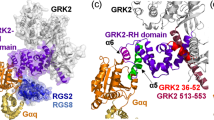
Abstract
The structural basis for the GTPase-accelerating activity of regulators of G protein signaling (RGS) proteins, as well as the mechanistic basis for their specificity in interacting with the heterotrimeric (αβγ) G proteins they inactivate, is not sufficiently understood at the family level. Here, we used biochemical assays to compare RGS domains across the RGS family and map those individual residues that favorably contribute to GTPase-accelerating activity, and those residues responsible for attenuating RGS domain interactions with Gα subunits. We show that conserved interactions of RGS residues with both the Gα switch I and II regions are crucial for RGS activity, while the reciprocal effects of “modulatory” and “disruptor” residues selectively modulate RGS activity. Our results quantify how specific interactions between RGS domains and Gα subunits are set by a balance between favorable RGS residue interactions with particular Gα switch regions, and unfavorable interactions with the Gα helical domain.







Similar content being viewed by others
Availability of data and material
Available upon request.
Code availability
Not applicable.
References
Sprang SR (1997) G protein mechanisms: insights from structural analysis. Annu Rev Biochem 66:639–678
Oldham WM, Hamm HE (2008) Heterotrimeric G protein activation by G-protein-coupled receptors. Nat Rev Mol Cell Biol 9(1):60–71
Berman DM, Kozasa T, Gilman AG (1996) The GTPase-activating protein RGS4 stabilizes the transition state for nucleotide hydrolysis. J Biol Chem 271(44):27209–27212
Hunt TW et al (1996) RGS10 is a selective activator of Gαi GTPase activity. Nature 383(6596):175–177
Koelle MR, Horvitz HR (1996) EGL-10 regulates G protein signaling in the C. elegans nervous system and shares a conserved domain with many mammalian proteins. Cell 84(1):115–125
Siderovski DP et al (1996) A new family of regulators of G-protein-coupled receptors? Curr Biol 6(2):211–212
Watson N et al (1996) RGS family members: GTPase-activating proteins for heterotrimeric G-protein alpha-subunits. Nature 383(6596):172–175
Neubig RR, Siderovski DP (2002) Regulators of G-protein signalling as new central nervous system drug targets. Nat Rev Drug Discov 1(3):187–197
Hollinger S, Hepler JR (2002) Cellular regulation of RGS proteins: modulators and integrators of G protein signaling. Pharmacol Rev 54(3):527–559
Neitzel KL, Hepler JR (2006) Cellular mechanisms that determine selective RGS protein regulation of G protein-coupled receptor signaling. Semin Cell Dev Biol 17(3):383–389
Hurst JH, Hooks SB (2009) Regulator of G-protein signaling (RGS) proteins in cancer biology. Biochem Pharmacol 78(10):1289–1297
Kimple AJ et al (2011) Regulators of G-protein signaling and their Galpha substrates: promises and challenges in their use as drug discovery targets. Pharmacol Rev 63(3):728–749
Ross EM, Wilkie TM (2000) GTPase-activating proteins for heterotrimeric G proteins: regulators of G protein signaling (RGS) and RGS-like proteins. Annu Rev Biochem 69:795–827
Squires KE et al (2018) Genetic analysis of rare human variants of regulators of G protein signaling proteins and their role in human physiology and disease. Pharmacol Rev 70(3):446–474
Masuho I et al (2020) A global map of G protein signaling regulation by RGS proteins. Cell 183(2):503–521 (e19)
Glick JL et al (1998) RGSZ1, a Gz-selective regulator of G protein signaling whose action is sensitive to the phosphorylation state of Gzalpha. J Biol Chem 273(40):26008–26013
Kosloff M et al (2011) Integrating energy calculations with functional assays to decipher the specificity of G protein-RGS protein interactions. Nat Struct Mol Biol 18(7):846–853
Salem-Mansour D et al (2018) Structural motifs in the RGS RZ subfamily combine to attenuate interactions with Gα subunits. Biochem Biophys Res Commun 503(4):2736–2741
Lan KL et al (2000) Rapid kinetics of regulator of G-protein signaling (RGS)-mediated Gαi and Gαo deactivation. Gα specificity of RGS4 AND RGS7. J Biol Chem 275(43):33497–33503
Hooks SB et al (2003) RGS6, RGS7, RGS9, and RGS11 stimulate GTPase activity of Gi family G-proteins with differential selectivity and maximal activity. J Biol Chem 278(12):10087–10093
Masuho I, Xie K, Martemyanov KA (2013) Macromolecular composition dictates receptor and G protein selectivity of regulator of G protein signaling (RGS) 7 and 9–2 protein complexes in living cells. J Biol Chem 288(35):25129–25142
Israeli R et al (2019) RGS6 and RGS7 discriminate between the highly similar Gαi and Gαo proteins using a two-tiered specificity strategy. J Mol Biol 431(17):3302–3311
Snow BE et al (1998) GTPase activating specificity of RGS12 and binding specificity of an alternatively spliced PDZ (PSD-95/Dlg/ZO-1) domain. J Biol Chem 273(28):17749–17755
Asli A et al (2018) “Disruptor” residues in the regulator of G protein signaling (RGS) R12 subfamily attenuate the inactivation of Galpha subunits. Sci Signal 11(534):eaan3677
Heximer SP et al (1999) G protein selectivity is a determinant of RGS2 function. J Biol Chem 274(48):34253–34259
Kimple AJ et al (2009) Structural determinants of G-protein alpha subunit selectivity by regulator of G-protein signaling 2 (RGS2). J Biol Chem 284(29):19402–19411
Kasom M et al (2018) Interplay between negative and positive design elements in Gα helical domains of G proteins determines interaction specificity toward RGS2. Biochem J 475(14):2293–2304
Natochin M, McEntaffer RL, Artemyev NO (1998) Mutational analysis of the Asn residue essential for RGS protein binding to G-proteins. J Biol Chem 273(12):6731–6735
Wieland T et al (2000) Polarity exchange at the interface of regulators of G protein signaling with G protein alpha-subunits. J Biol Chem 275(37):28500–28506
Moratz C et al (2000) Regulator of G protein signaling 1 (RGS1) markedly impairs Gαi signaling responses of B lymphocytes. J Immunol 164(4):1829–1838
Cladman W, Chidiac P (2002) Characterization and comparison of RGS2 and RGS4 as GTPase-activating proteins for m2 muscarinic receptor-stimulated G(i). Mol Pharmacol 62(3):654–659
Derrien A et al (2003) Src-mediated RGS16 tyrosine phosphorylation promotes RGS16 stability. J Biol Chem 278(18):16107–16116
Cohen SP et al (2012) Regulator of G-protein signaling-21 (RGS21) is an inhibitor of bitter gustatory signaling found in lingual and airway epithelia. J Biol Chem 287(50):41706–41719
Tesmer JJ et al (1997) Structure of RGS4 bound to AlF4–activated Gia1: stabilization of the transition state for GTP hydrolysis. Cell 89(2):251–261
Sprang SR, Chen Z, Du X (2007) Structural basis of effector regulation and signal termination in heterotrimeric Gα proteins. Adv Protein Chem 74:1–65
Mann D et al (2016) Mechanism of the intrinsic arginine finger in heterotrimeric G proteins. Proc Natl Acad Sci USA 113(50):E8041–E8050
Srinivasa SP et al (1998) Mechanism of RGS4, a GTPase-activating protein for G protein alpha subunits. J Biol Chem 273(3):1529–1533
Posner BA et al (1999) Modulation of the affinity and selectivity of RGS protein interaction with Gα subunits by a conserved asparagine/serine residue. Biochemistry 38(24):7773–7779
Slep KC et al (2008) Molecular architecture of Gαo and the structural basis for RGS16-mediated deactivation. Proc Natl Acad Sci USA 105(17):6243–6248
Soundararajan M et al (2008) Structural diversity in the RGS domain and its interaction with heterotrimeric G protein alpha-subunits. Proc Natl Acad Sci USA 105(17):6457–6462
Nance MR et al (2013) Structural and functional analysis of the regulator of G protein signaling 2-Gαq complex. Structure 21(3):438–448
Taylor VG, Bommarito PA, Tesmer JJ (2016) Structure of the regulator of G protein signaling 8 (RGS8)-Gαq complex: molecular basis for Gα selectivity. J Biol Chem 291(10):5138–5145
Coleman DE, Sprang SR (1998) Crystal structures of the G protein Giα1 complexed with GDP and Mg2+: a crystallographic titration experiment. Biochemistry 37(41):14376–14385
Owen VJ et al (2001) Expression of RGS3, RGS4 and Giα2 in acutely failing donor hearts and end-stage heart failure. Eur Heart J 22(12):1015–1020
Li H et al (2010) Regulator of G protein signaling 5 protects against cardiac hypertrophy and fibrosis during biomechanical stress of pressure overload. Proc Natl Acad Sci USA 107(31):13818–13823
Ooe A, Kato K, Noguchi S (2007) Possible involvement of CCT5, RGS3, and YKT6 genes up-regulated in p53-mutated tumors in resistance to docetaxel in human breast cancers. Breast Cancer Res Treat 101(3):305–315
Ganss R (2015) Keeping the balance right: regulator of G protein signaling 5 in vascular physiology and pathology. Prog Mol Biol Transl Sci 133:93–121
Furuya M et al (2004) Expression of regulator of G protein signalling protein 5 (RGS5) in the tumour vasculature of human renal cell carcinoma. J Pathol 203(1):551–558
Altman MK et al (2012) Regulator of G-protein signaling 5 reduces HeyA8 ovarian cancer cell proliferation and extends survival in a murine tumor model. Biochem Res Int 2012:518437
Hu M et al (2013) Over-expression of regulator of G protein signaling 5 promotes tumor metastasis by inducing epithelial-mesenchymal transition in hepatocellular carcinoma cells. J Surg Oncol 108(3):192–196
Druey KM (2009) Regulation of G-protein-coupled signaling pathways in allergic inflammation. Immunol Res 43(1–3):62–76
Hwang IY et al (2013) Rgs13 constrains early B cell responses and limits germinal center sizes. PLoS ONE 8(3):e60139
Wang JH et al (2013) Extension of the germinal center stage of B cell development promotes autoantibodies in BXD2 mice. Arthritis Rheum 65(10):2703–2712
Williams JW et al (2013) RGS3 controls T lymphocyte migration in a model of Th2-mediated airway inflammation. Am J Physiol Lung Cell Mol Physiol 305(10):L693-701
Xie Z, Chan EC, Druey KM (2016) R4 regulator of G protein signaling (RGS) proteins in inflammation and immunity. AAPS J 18(2):294–304
O’Brien JB, Wilkinson JC, Roman DL (2019) Regulator of G-protein signaling (RGS) proteins as drug targets: progress and future potentials. J Biol Chem 294(49):18571–18585
Senese NB et al (2020) Regulator of G-protein signaling (RGS) protein modulation of opioid receptor signaling as a potential target for pain management. Front Mol Neurosci 13:5
DiGiacomo V et al (2020) Probing the mutational landscape of regulators of G protein signaling proteins in cancer. Sci Signal 13(617):eaax8620
Bakhman A et al (2019) Residue-level determinants of angiopoietin-2 interactions with its receptor Tie2. Proteins 87(3):185–197
Shushan A, Kosloff M (2021) Structural design principles for specific ultra-high affinity interactions between colicins/pyocins and immunity proteins. Sci Rep 11(1):3789
Gibson DG et al (2009) Enzymatic assembly of DNA molecules up to several hundred kilobases. Nat Methods 6(5):343–345
Sun D et al (2013) AAscan, PCRdesign and MutantChecker: a suite of programs for primer design and sequence analysis for high-throughput scanning mutagenesis. PLoS ONE 8(10):e78878
Wang J et al (1997) A GTPase-activating protein for the G protein Gαz: identification, purification, and mechanism of action. J Biol Chem 272(9):5732–5740
Ross EM (2002) Quantitative assays for GTPase-activating proteins. Methods Enzymol 344:601–617
Mukhopadhyay S, Ross EM (1999) Rapid GTP binding and hydrolysis by G(q) promoted by receptor and GTPase-activating proteins. Proc Natl Acad Sci USA 96(17):9539–9544
Acknowledgements
We thank John Kehrl (NIH) for RGS3, RGS5, and RGS13 clones, Vadim Arshavsky for the Gαo clone, and Anna Bakhman for technical assistance.
Funding
This work was supported by grants from the Israel Science Foundation (Grant Numbers: 1454/13, 1959/13, 2155/15), the Canadian Institutes of Health Research (CIHR), the International Development Research Centre (IDRC), the Israel Science Foundation (ISF), and the Azrieli Foundation (Grant Number 3512/19) and the DS Research Center at the University of Haifa. The authors acknowledge the contributions of COST Actions CM-1207 (GLISTEN), CA-15126 (ARBIEU), and CA-18133 (ERNEST) and iNEXT actions PID 4101 and 6623 to this work.
Author information
Authors and Affiliations
Contributions
AA designed the research, conducted most of the experiments and structural analysis, analyzed results, and wrote the paper. SHM conducted experiments, analyzed results, and contributed to the paper. MAS conducted experiments, supervised lab work, analyzed results, and contributed to the paper. MK designed and supervised the research and analysis and wrote the paper. All authors were involved in the writing of the paper and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Asli, A., Higazy-Mreih, S., Avital-Shacham, M. et al. Residue-level determinants of RGS R4 subfamily GAP activity and specificity towards the Gi subfamily. Cell. Mol. Life Sci. 78, 6305–6318 (2021). https://doi.org/10.1007/s00018-021-03898-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-021-03898-4