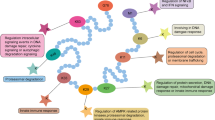
Abstract
Studies over the past decades have elucidated the critical role of autophagy in human health and diseases. Although the processes of autophagy in the cytoplasm have been well studied, the posttranscriptional and epigenetic regulation mechanisms of autophagy are still poorly understood. Protein methylation, including histone methylation and non-histone protein methylation, is the most important type of posttranscriptional and epigenetic modification. Recent studies have shown that protein methylation is associated with effects on autophagosome formation, autophagy-related protein expression, and signaling pathway activation, but the details are still unclear. Thus, it is important to summarize the current status and discuss the future directions of research on protein methylation in the context of autophagy.



Similar content being viewed by others
References
Ashford TP, Porter KR (1962) Cytoplasmic components in hepatic cell lysosomes. J Cell Biol 12:198–202
Mei Y, Glover K, Su M, Sinha SC (2016) Conformational flexibility of BECN1: essential to its key role in autophagy and beyond. Protein Sci 25(10):1767–1785. https://doi.org/10.1002/pro.2984
Xie Z, Klionsky DJ (2007) Autophagosome formation: core machinery and adaptations. Nat Cell Biol 9(10):1102–1109. https://doi.org/10.1038/ncb1007-1102
Mizushima N, Levine B, Cuervo AM, Klionsky DJ (2008) Autophagy fights disease through cellular self-digestion. Nature 451(7182):1069–1075. https://doi.org/10.1038/nature06639
Klionsky DJ, Abdelmohsen K, Abe A, Abedin MJ, Abeliovich H, Acevedo Arozena A et al (2016) Guidelines for the use and interpretation of assays for monitoring autophagy (3rd edition). Autophagy 12(1):1–222. https://doi.org/10.1080/15548627.2015.1100356
Ahmad L, Mashbat B, Leung C, Brookes C, Hamad S, Krokowski S, Shenoy AR, Lorenzo L, Levin M, O’Hare P, Zhang SY, Casanova JL, Mostowy S, Sancho-Shimizu V (2018) Human TANK-binding kinase 1 is required for early autophagy induction upon herpes simplex virus 1 infection. J Allergy Clin Immunol. https://doi.org/10.1016/j.jaci.2018.09.013
Kato H, Perl A (2018) Blockade of Treg cell differentiation and function by the interleukin-21-mechanistic target of rapamycin axis via suppression of autophagy in patients with systemic lupus erythematosus. Arthritis Rheumatol 70(3):427–438. https://doi.org/10.1002/art.40380
Liu H, Fang S, Wang W, Cheng Y, Zhang Y, Liao H, Yao H, Chao J (2016) Macrophage-derived MCPIP1 mediates silica-induced pulmonary fibrosis via autophagy. Part Fibre Toxicol 13(1):55. https://doi.org/10.1186/s12989-016-0167-z
Duan X, Kong Z, Mai X, Lan Y, Liu Y, Yang Z, Zhao Z, Deng T, Zeng T, Cai C, Li S, Zhong W, Wu W, Zeng G (2018) Autophagy inhibition attenuates hyperoxaluria-induced renal tubular oxidative injury and calcium oxalate crystal depositions in the rat kidney. Redox Biol 16:414–425. https://doi.org/10.1016/j.redox.2018.03.019
Zhang Y, Whaley-Connell AT, Sowers JR, Ren J (2018) Autophagy as an emerging target in cardiorenal metabolic disease: from pathophysiology to management. Pharmacol Ther 191:1–22. https://doi.org/10.1016/j.pharmthera.2018.06.004
Menzies FM, Fleming A, Caricasole A, Bento CF, Andrews SP, Ashkenazi A, Fullgrabe J, Jackson A, Jimenez Sanchez M, Karabiyik C, Licitra F, Lopez Ramirez A, Pavel M, Puri C, Renna M, Ricketts T, Schlotawa L, Vicinanza M, Won H, Zhu Y, Skidmore J, Rubinsztein DC (2017) Autophagy and neurodegeneration: pathogenic mechanisms and therapeutic opportunities. Neuron 93(5):1015–1034. https://doi.org/10.1016/j.neuron.2017.01.022
Sun CY, Zhang QY, Zheng GJ, Feng B (2018) Autophagy and its potent modulators from phytochemicals in cancer treatment. Cancer Chemother Pharmacol. https://doi.org/10.1007/s00280-018-3707-4
Tai S, Hu XQ, Peng DQ, Zhou SH, Zheng XL (2016) The roles of autophagy in vascular smooth muscle cells. Int J Cardiol 211:1–6. https://doi.org/10.1016/j.ijcard.2016.02.128
Mizushima N (2007) Autophagy: process and function. Genes Dev 21(22):2861–2873. https://doi.org/10.1101/gad.1599207
Hansen M, Rubinsztein DC, Walker DW (2018) Autophagy as a promoter of longevity: insights from model organisms. Nat Rev Mol Cell Biol 19(9):579–593. https://doi.org/10.1038/s41580-018-0033-y
Xie Y, Kang R, Sun X, Zhong M, Huang J, Klionsky DJ, Tang D (2015) Posttranslational modification of autophagy-related proteins in macroautophagy. Autophagy 11(1):28–45. https://doi.org/10.4161/15548627.2014.984267
Wani WY, Boyer-Guittaut M, Dodson M, Chatham J, Darley-Usmar V, Zhang J (2015) Regulation of autophagy by protein post-translational modification. Lab Invest 95(1):14–25. https://doi.org/10.1038/labinvest.2014.131
Kuma A, Mizushima N, Ishihara N, Ohsumi Y (2002) Formation of the approximately 350-kDa Apg12–Apg5.Apg16 multimeric complex, mediated by Apg16 oligomerization, is essential for autophagy in yeast. J Biol Chem 277(21):18619–18625. https://doi.org/10.1074/jbc.m111889200
Song H, Pu J, Wang L, Wu L, Xiao J, Liu Q, Chen J, Zhang M, Liu Y, Ni M, Mo J, Zheng Y, Wan D, Cai X, Cao Y, Xiao W, Ye L, Tu E, Lin Z, Wen J, Lu X, He J, Peng Y, Su J, Zhang H, Zhao Y, Lin M, Zhang Z (2015) ATG16L1 phosphorylation is oppositely regulated by CSNK2/casein kinase 2 and PPP1/protein phosphatase 1 which determines the fate of cardiomyocytes during hypoxia/reoxygenation. Autophagy 11(8):1308–1325. https://doi.org/10.1080/15548627.2015.1060386
Pankiv S, Clausen TH, Lamark T, Brech A, Bruun JA, Outzen H, Overvatn A, Bjorkoy G, Johansen T (2007) p62/SQSTM1 binds directly to Atg8/LC3 to facilitate degradation of ubiquitinated protein aggregates by autophagy. J Biol Chem 282(33):24131–24145. https://doi.org/10.1074/jbc.M702824200
Baek SH, Kim KI (2017) Epigenetic control of autophagy: nuclear events gain more attention. Mol Cell 65(5):781–785. https://doi.org/10.1016/j.molcel.2016.12.027
Wu Z, Connolly J, Biggar KK (2017) Beyond histones—the expanding roles of protein lysine methylation. FEBS J 284(17):2732–2744. https://doi.org/10.1111/febs.14056
Murray K (1964) The occurrence of epsilon-N-methyl lysine in histones. Biochemistry 3:10–15
Byvoet P, Shepherd GR, Hardin JM, Noland BJ (1972) The distribution and turnover of labeled methyl groups in histone fractions of cultured mammalian cells. Arch Biochem Biophys 148(2):558–567
Trievel RC, Beach BM, Dirk LM, Houtz RL, Hurley JH (2002) Structure and catalytic mechanism of a SET domain protein methyltransferase. Cell 111(1):91–103
Lee S, Oh S, Jeong K, Jo H, Choi Y, Seo HD, Kim M, Choe J, Kwon CS, Lee D (2018) Dot1 regulates nucleosome dynamics by its inherent histone chaperone activity in yeast. Nat Commun 9(1):240. https://doi.org/10.1038/s41467-017-02759-8
Blanc RS, Richard S (2017) Arginine methylation: the coming of age. Mol Cell 65(1):8–24. https://doi.org/10.1016/j.molcel.2016.11.003
Greer EL, Shi Y (2012) Histone methylation: a dynamic mark in health, disease and inheritance. Nat Rev Genet 13(5):343–357. https://doi.org/10.1038/nrg3173
Mosammaparast N, Shi Y (2010) Reversal of histone methylation: biochemical and molecular mechanisms of histone demethylases. Annu Rev Biochem 79:155–179. https://doi.org/10.1146/annurev.biochem.78.070907.103946
Wei FZ, Cao Z, Wang X, Wang H, Cai MY, Li T, Hattori N, Wang D, Du Y, Song B, Cao LL, Shen C, Wang L, Wang H, Yang Y, Xie D, Wang F, Ushijima T, Zhao Y, Zhu WG (2015) Epigenetic regulation of autophagy by the methyltransferase EZH2 through an MTOR-dependent pathway. Autophagy 11(12):2309–2322. https://doi.org/10.1080/15548627.2015.1117734
Tan JZ, Yan Y, Wang XX, Jiang Y, Xu HE (2014) EZH2: biology, disease, and structure-based drug discovery. Acta Pharmacol Sin 35(2):161–174. https://doi.org/10.1038/aps.2013.161
Liu TP, Hong YH, Tung KY, Yang PM (2016) In silico and experimental analyses predict the therapeutic value of an EZH2 inhibitor GSK343 against hepatocellular carcinoma through the induction of metallothionein genes. Oncoscience 3(1):9–20. https://doi.org/10.18632/oncoscience.285
Hsieh YY, Lo HL, Yang PM (2016) EZH2 inhibitors transcriptionally upregulate cytotoxic autophagy and cytoprotective unfolded protein response in human colorectal cancer cells. Am J Cancer Res 6(8):1661–1680
Sun Y, Jin L, Liu JH, Sui YX, Han LL, Shen XL (2016) Interfering EZH2 expression reverses the cisplatin resistance in human ovarian cancer by inhibiting autophagy. Cancer Biother Radiopharm 31(7):246–252. https://doi.org/10.1089/cbr.2016.2034
Wang Z, Liu H, Xu C (2018) Cellular senescence in the treatment of ovarian cancer. Int J Gynecol Cancer. https://doi.org/10.1097/igc.0000000000001257
Li R, Yi X, Wei X, Huo B, Guo X, Cheng C, Fang ZM, Wang J, Feng X, Zheng P, Su YS, Masau JF, Zhu XH, Jiang DS (2018) EZH2 inhibits autophagic cell death of aortic vascular smooth muscle cells to affect aortic dissection. Cell Death Dis 9(2):180. https://doi.org/10.1038/s41419-017-0213-2
Galluzzi L, Vitale I, Abrams JM, Alnemri ES, Baehrecke EH, Blagosklonny MV, Dawson TM, Dawson VL, El-Deiry WS, Fulda S, Gottlieb E, Green DR, Hengartner MO, Kepp O, Knight RA, Kumar S, Lipton SA, Lu X, Madeo F, Malorni W, Mehlen P, Nunez G, Peter ME, Piacentini M, Rubinsztein DC, Shi Y, Simon HU, Vandenabeele P, White E, Yuan J, Zhivotovsky B, Melino G, Kroemer G (2012) Molecular definitions of cell death subroutines: recommendations of the Nomenclature Committee on Cell Death 2012. Cell Death Differ 19(1):107–120. https://doi.org/10.1038/cdd.2011.96
Shinkai Y (2007) Regulation and function of H3K9 methylation. Subcell Biochem 41:337–350
Jin Q, Yu LR, Wang L, Zhang Z, Kasper LH, Lee JE, Wang C, Brindle PK, Dent SY, Ge K (2011) Distinct roles of GCN5/PCAF-mediated H3K9ac and CBP/p300-mediated H3K18/27ac in nuclear receptor transactivation. EMBO J 30(2):249–262. https://doi.org/10.1038/emboj.2010.318
Artal-Martinez de Narvajas A, Gomez TS, Zhang JS, Mann AO, Taoda Y, Gorman JA, Herreros-Villanueva M, Gress TM, Ellenrieder V, Bujanda L, Kim DH, Kozikowski AP, Koenig A, Billadeau DD (2013) Epigenetic regulation of autophagy by the methyltransferase G9a. Mol Cell Biol 33(20):3983–3993. https://doi.org/10.1128/MCB.00813-13
Collins PL, Oltz EM (2013) Histone methylation keeps the brakes on autophagy. Mol Cell Biol 33(20):3974–3975. https://doi.org/10.1128/MCB.01033-13
Morgan C, Hibben M, Esan O, John S, Patel V, Weiss HA, Murray RM, Hutchinson G, Gureje O, Thara R, Cohen A (2015) Searching for psychosis: INTREPID (1): systems for detecting untreated and first-episode cases of psychosis in diverse settings. Soc Psychiatry Psychiatr Epidemiol 50(6):879–893. https://doi.org/10.1007/s00127-015-1013-6
Li F, Zeng J, Gao Y, Guan Z, Ma Z, Shi Q, Du C, Jia J, Xu S, Wang X, Chang L, He D, Guo P (2015) G9a inhibition induces autophagic cell death via AMPK/mTOR pathway in bladder transitional cell carcinoma. PLoS One 10(9):e0138390. https://doi.org/10.1371/journal.pone.0138390
Ren A, Qiu Y, Cui H, Fu G (2015) Inhibition of H3K9 methyltransferase G9a induces autophagy and apoptosis in oral squamous cell carcinoma. Biochem Biophys Res Commun 459(1):10–17. https://doi.org/10.1016/j.bbrc.2015.01.068
Ke XX, Zhang D, Zhu S, Xia Q, Xiang Z, Cui H (2014) Inhibition of H3K9 methyltransferase G9a repressed cell proliferation and induced autophagy in neuroblastoma cells. PLoS One 9(9):e106962. https://doi.org/10.1371/journal.pone.0106962
Park SE, Yi HJ, Suh N, Park YY, Koh JY, Jeong SY, Cho DH, Kim CS, Hwang JJ (2016) Inhibition of EHMT2/G9a epigenetically increases the transcription of Beclin-1 via an increase in ROS and activation of NF-kappaB. Oncotarget 7(26):39796–39808. https://doi.org/10.18632/oncotarget.9290
Shilatifard A (2008) Molecular implementation and physiological roles for histone H3 lysine 4 (H3K4) methylation. Curr Opin Cell Biol 20(3):341–348. https://doi.org/10.1016/j.ceb.2008.03.019
Ni P, Xu H, Chen C, Wang J, Liu X, Hu Y, Fan Q, Hou Z, Lu Y (2012) Serum starvation induces DRAM expression in liver cancer cells via histone modifications within its promoter locus. PLoS One 7(12):e50502. https://doi.org/10.1371/journal.pone.0050502
Rosenstein DI, Chiodo GT, Bartley MH (1991) Treating recurrent aphthous ulcers in patients with AIDS. J Am Dent Assoc 122(10):64, 67–68
Shen B, Tan M, Mu X, Qin Y, Zhang F, Liu Y, Fan Y (2016) Upregulated SMYD3 promotes bladder cancer progression by targeting BCLAF1 and activating autophagy. Tumour Biol 37(6):7371–7381. https://doi.org/10.1007/s13277-015-4410-2
Fullgrabe J, Lynch-Day MA, Heldring N, Li W, Struijk RB, Ma Q, Hermanson O, Rosenfeld MG, Klionsky DJ, Joseph B (2013) The histone H4 lysine 16 acetyltransferase hMOF regulates the outcome of autophagy. Nature 500(7463):468–471. https://doi.org/10.1038/nature12313
Wang Z, Long QY, Chen L, Fan JD, Wang ZN, Li LY, Wu M, Chen X (2017) Inhibition of H3K4 demethylation induces autophagy in cancer cell lines. Biochim Biophys Acta 1864 12:2428–2437. https://doi.org/10.1016/j.bbamcr.2017.08.005
Hong F, Wan L, Liu J, Huang K, Xiao Z, Zhang Y, Shi C (2018) Histone methylation regulates Hif-1 signaling cascade in activation of hepatic stellate cells. FEBS Open Bio 8(3):406–415. https://doi.org/10.1002/2211-5463.12379
Shin HJ, Kim H, Oh S, Lee JG, Kee M, Ko HJ, Kweon MN, Won KJ, Baek SH (2016) AMPK–SKP2–CARM1 signalling cascade in transcriptional regulation of autophagy. Nature 534(7608):553–557. https://doi.org/10.1038/nature18014
Selvi BR, Batta K, Kishore AH, Mantelingu K, Varier RA, Balasubramanyam K, Pradhan SK, Dasgupta D, Sriram S, Agrawal S, Kundu TK (2010) Identification of a novel inhibitor of coactivator-associated arginine methyltransferase 1 (CARM1)-mediated methylation of histone H3 Arg-17. J Biol Chem 285(10):7143–7152. https://doi.org/10.1074/jbc.M109.063933
Farooq Z, Banday S, Pandita TK, Altaf M (2016) The many faces of histone H3K79 methylation. Mutat Res Rev Mutat Res 768:46–52. https://doi.org/10.1016/j.mrrev.2016.03.005
Gao Y, Ge W (2018) The histone methyltransferase DOT1L inhibits osteoclastogenesis and protects against osteoporosis. Cell Death Dis 9(2):33. https://doi.org/10.1038/s41419-017-0040-5
Hoeflich KP, Gray DC, Eby MT, Tien JY, Wong L, Bower J, Gogineni A, Zha J, Cole MJ, Stern HM, Murray LJ, Davis DP, Seshagiri S (2006) Oncogenic BRAF is required for tumor growth and maintenance in melanoma models. Cancer Res 66(2):999–1006. https://doi.org/10.1158/0008-5472.CAN-05-2720
Xu J, Wang AH, Oses-Prieto J, Makhijani K, Katsuno Y, Pei M, Yan L, Zheng YG, Burlingame A, Bruckner K, Derynck R (2013) Arginine methylation initiates BMP-induced smad signaling. Mol Cell 51(1):5–19. https://doi.org/10.1016/j.molcel.2013.05.004
Biggar KK, Li SS (2015) Non-histone protein methylation as a regulator of cellular signalling and function. Nat Rev Mol Cell Biol 16(1):5–17. https://doi.org/10.1038/nrm3915
Wu D, Pan W (2010) GSK3: a multifaceted kinase in Wnt signaling. Trends Biochem Sci 35(3):161–168. https://doi.org/10.1016/j.tibs.2009.10.002
Mizushima N, Yoshimori T, Ohsumi Y (2011) The role of Atg proteins in autophagosome formation. Annu Rev Cell Dev Biol 27:107–132. https://doi.org/10.1146/annurev-cellbio-092910-154005
Song H, Feng X, Zhang M, Jin X, Xu X, Wang L, Ding X, Luo Y, Lin F, Wu Q, Liang G, Yu T, Liu Q, Zhang Z (2018) Crosstalk between lysine methylation and phosphorylation of ATG16L1 dictates the apoptosis of hypoxia/reoxygenation-induced cardiomyocytes. Autophagy 14(5):825–844. https://doi.org/10.1080/15548627.2017.1389357
Wen C, Xu M, Mo C, Cheng Z, Guo Q, Zhu X (2018) JMJD6 exerts function in neuropathic pain by regulating NFkappaB following peripheral nerve injury in rats. Int J Mol Med. https://doi.org/10.3892/ijmm.2018.3613
Hsu JM, Chen CT, Chou CK, Kuo HP, Li LY, Lin CY, Lee HJ, Wang YN, Liu M, Liao HW, Shi B, Lai CC, Bedford MT, Tsai CH, Hung MC (2011) Crosstalk between Arg 1175 methylation and Tyr 1173 phosphorylation negatively modulates EGFR-mediated ERK activation. Nat Cell Biol 13(2):174–181. https://doi.org/10.1038/ncb2158
Jiang H, Zhou Z, Jin S, Xu K, Zhang H, Xu J, Sun Q, Wang J, Xu J (2018) PRMT9 promotes hepatocellular carcinoma invasion and metastasis via activating PI3K/Akt/GSK-3beta/Snail signalling. Cancer Sci. https://doi.org/10.1111/cas.13598
Raposo AE, Piller SC (2018) Protein arginine methylation: an emerging regulator of the cell cycle. Cell Div 13:3. https://doi.org/10.1186/s13008-018-0036-2
Wils LJ, Bijlsma MF (2018) Epigenetic regulation of the Hedgehog and Wnt pathways in cancer. Crit Rev Oncol Hematol 121:23–44. https://doi.org/10.1016/j.critrevonc.2017.11.013
Nandy A, Lin L, Velentzas PD, Wu LP, Baehrecke EH, Silverman N (2018) The NF-kappaB factor relish regulates Atg1 expression and controls autophagy. Cell Rep 25(8):2110–2120 e2113. https://doi.org/10.1016/j.celrep.2018.10.076
Kretowski R, Borzym-Kluczyk M, Stypulkowska A, Branska-Januszewska J, Ostrowska H, Cechowska-Pasko M (2016) Low glucose dependent decrease of apoptosis and induction of autophagy in breast cancer MCF-7 cells. Mol Cell Biochem 417(1–2):35–47. https://doi.org/10.1007/s11010-016-2711-4
Copetti T, Bertoli C, Dalla E, Demarchi F, Schneider C (2009) p65/RelA modulates BECN1 transcription and autophagy. Mol Cell Biol 29(10):2594–2608. https://doi.org/10.1128/MCB.01396-08
Lim CB, Fu PY, Ky N, Zhu HS, Feng X, Li J, Srinivasan KG, Hamza MS, Zhao Y (2012) NF-kappaB p65 repression by the sesquiterpene lactone, Helenalin, contributes to the induction of autophagy cell death. BMC Complement Altern Med 12:93. https://doi.org/10.1186/1472-6882-12-93
Verma N, Manna SK (2017) Advanced glycation end products (AGE) potentiates cell death in p53 negative cells via upregulaion of NF-kappa B and impairment of autophagy. J Cell Physiol 232(12):3598–3610. https://doi.org/10.1002/jcp.25828
Gilmore TD (2006) Introduction to NF-kappaB: players, pathways, perspectives. Oncogene 25(51):6680–6684. https://doi.org/10.1038/sj.onc.1209954
Xia L, Tan S, Zhou Y, Lin J, Wang H, Oyang L, Tian Y, Liu L, Su M, Wang H, Cao D, Liao Q (2018) Role of the NFkappaB-signaling pathway in cancer. Onco Targets Ther 11:2063–2073. https://doi.org/10.2147/OTT.S161109
Ea CK, Baltimore D (2009) Regulation of NF-kappaB activity through lysine monomethylation of p65. Proc Natl Acad Sci USA 106(45):18972–18977. https://doi.org/10.1073/pnas.0910439106
Mukherjee N, Cardenas E, Bedolla R, Ghosh R (2017) SETD6 regulates NF-kappaB signaling in urothelial cell survival: implications for bladder cancer. Oncotarget 8(9):15114–15125. https://doi.org/10.18632/oncotarget.14750
Yuan ZL, Guan YJ, Wang L, Wei W, Kane AB, Chin YE (2004) Central role of the threonine residue within the p + 1 loop of receptor tyrosine kinase in STAT3 constitutive phosphorylation in metastatic cancer cells. Mol Cell Biol 24(21):9390–9400. https://doi.org/10.1128/MCB.24.21.9390-9400.2004
Hu W, Lv J, Han M, Yang Z, Li T, Jiang S, Yang Y (2018) STAT3: the art of multi-tasking of metabolic and immune functions in obesity. Prog Lipid Res 70:17–28. https://doi.org/10.1016/j.plipres.2018.04.002
You L, Wang Z, Li H, Shou J, Jing Z, Xie J, Sui X, Pan H, Han W (2015) The role of STAT3 in autophagy. Autophagy 11(5):729–739. https://doi.org/10.1080/15548627.2015.1017192
Feng Y, Ke C, Tang Q, Dong H, Zheng X, Lin W, Ke J, Huang J, Yeung SC, Zhang H (2014) Metformin promotes autophagy and apoptosis in esophageal squamous cell carcinoma by downregulating Stat3 signaling. Cell Death Dis 5:e1088. https://doi.org/10.1038/cddis.2014.59
Mazure NM, Pouyssegur J (2010) Hypoxia-induced autophagy: cell death or cell survival? Curr Opin Cell Biol 22(2):177–180. https://doi.org/10.1016/j.ceb.2009.11.015
Kim E, Kim M, Woo DH, Shin Y, Shin J, Chang N, Oh YT, Kim H, Rheey J, Nakano I, Lee C, Joo KM, Rich JN, Nam DH, Lee J (2013) Phosphorylation of EZH2 activates STAT3 signaling via STAT3 methylation and promotes tumorigenicity of glioblastoma stem-like cells. Cancer Cell 23(6):839–852. https://doi.org/10.1016/j.ccr.2013.04.008
Kong J, Kong F, Gao J, Zhang Q, Dong S, Gu F, Ke S, Pan B, Shen Q, Sun H, Zheng L, Sun W (2014) YC-1 enhances the anti-tumor activity of sorafenib through inhibition of signal transducer and activator of transcription 3 (STAT3) in hepatocellular carcinoma. Mol Cancer 13:7. https://doi.org/10.1186/1476-4598-13-7
Cargnello M, Roux PP (2011) Activation and function of the MAPKs and their substrates, the MAPK-activated protein kinases. Microbiol Mol Biol Rev 75(1):50–83. https://doi.org/10.1128/MMBR.00031-10
Martinez-Lopez N, Singh R (2014) ATGs: scaffolds for MAPK/ERK signaling. Autophagy 10(3):535–537. https://doi.org/10.4161/auto.27642
Cagnol S, Chambard JC (2010) ERK and cell death: mechanisms of ERK-induced cell death—apoptosis, autophagy and senescence. FEBS J 277(1):2–21. https://doi.org/10.1111/j.1742-4658.2009.07366.x
Andreu-Perez P, Esteve-Puig R, de Torre-Minguela C, Lopez-Fauqued M, Bech-Serra JJ, Tenbaum S, Garcia-Trevijano ER, Canals F, Merlino G, Avila MA, Recio JA (2011) Protein arginine methyltransferase 5 regulates ERK1/2 signal transduction amplitude and cell fate through CRAF. Sci Signal 4(190):ra58. https://doi.org/10.1126/scisignal.2001936
Kruse JP, Gu W (2009) Modes of p53 regulation. Cell 137(4):609–622. https://doi.org/10.1016/j.cell.2009.04.050
Maiuri MC, Galluzzi L, Morselli E, Kepp O, Malik SA, Kroemer G (2010) Autophagy regulation by p53. Curr Opin Cell Biol 22(2):181–185. https://doi.org/10.1016/j.ceb.2009.12.001
Feng Z, Zhang H, Levine AJ, Jin S (2005) The coordinate regulation of the p53 and mTOR pathways in cells. Proc Natl Acad Sci USA 102(23):8204–8209. https://doi.org/10.1073/pnas.0502857102
Feng Z, Hu W, de Stanchina E, Teresky AK, Jin S, Lowe S, Levine AJ (2007) The regulation of AMPK beta1, TSC2, and PTEN expression by p53: stress, cell and tissue specificity, and the role of these gene products in modulating the IGF-1-AKT-mTOR pathways. Cancer Res 67(7):3043–3053. https://doi.org/10.1158/0008-5472.CAN-06-4149
Budanov AV, Karin M (2008) p53 target genes sestrin1 and sestrin2 connect genotoxic stress and mTOR signaling. Cell 134(3):451–460. https://doi.org/10.1016/j.cell.2008.06.028
Appella E, Anderson CW (2001) Post-translational modifications and activation of p53 by genotoxic stresses. Eur J Biochem 268(10):2764–2772
Chuikov S, Kurash JK, Wilson JR, Xiao B, Justin N, Ivanov GS, McKinney K, Tempst P, Prives C, Gamblin SJ, Barlev NA, Reinberg D (2004) Regulation of p53 activity through lysine methylation. Nature 432(7015):353–360. https://doi.org/10.1038/nature03117
Huang J, Perez-Burgos L, Placek BJ, Sengupta R, Richter M, Dorsey JA, Kubicek S, Opravil S, Jenuwein T, Berger SL (2006) Repression of p53 activity by Smyd2-mediated methylation. Nature 444(7119):629–632. https://doi.org/10.1038/nature05287
Fan JD, Lei PJ, Zheng JY, Wang X, Li S, Liu H, He YL, Wang ZN, Wei G, Zhang X, Li LY, Wu M (2015) The selective activation of p53 target genes regulated by SMYD2 in BIX-01294 induced autophagy-related cell death. PLoS One 10(1):e0116782. https://doi.org/10.1371/journal.pone.0116782
Tasdemir E, Maiuri MC, Galluzzi L, Vitale I, Djavaheri-Mergny M, D’Amelio M, Criollo A, Morselli E, Zhu C, Harper F, Nannmark U, Samara C, Pinton P, Vicencio JM, Carnuccio R, Moll UM, Madeo F, Paterlini-Brechot P, Rizzuto R, Szabadkai G, Pierron G, Blomgren K, Tavernarakis N, Codogno P, Cecconi F, Kroemer G (2008) Regulation of autophagy by cytoplasmic p53. Nat Cell Biol 10(6):676–687. https://doi.org/10.1038/ncb1730
Morselli E, Tasdemir E, Maiuri MC, Galluzzi L, Kepp O, Criollo A, Vicencio JM, Soussi T, Kroemer G (2008) Mutant p53 protein localized in the cytoplasm inhibits autophagy. Cell Cycle 7(19):3056–3061. https://doi.org/10.4161/cc.7.19.6751
Carter S, Bischof O, Dejean A, Vousden KH (2007) C-terminal modifications regulate MDM2 dissociation and nuclear export of p53. Nat Cell Biol 9(4):428–435. https://doi.org/10.1038/ncb1562
Venne AS, Kollipara L, Zahedi RP (2014) The next level of complexity: crosstalk of posttranslational modifications. Proteomics 14(4–5):513–524. https://doi.org/10.1002/pmic.201300344
Gu B, Zhu WG (2012) Surf the post-translational modification network of p53 regulation. Int J Biol Sci 8(5):672–684. https://doi.org/10.7150/ijbs.4283
Acknowledgements
This work was supported by Grants from the National Natural Science Foundation of China (nos. 81600188, 81670050), Tongji Hospital Fund for Distinguished Young Scholars (no. 2016YQ02), and Integrated Innovative Team for Major Human Diseases Program of Tongji Medical College, HUST.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no competing interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Li, R., Wei, X. & Jiang, DS. Protein methylation functions as the posttranslational modification switch to regulate autophagy. Cell. Mol. Life Sci. 76, 3711–3722 (2019). https://doi.org/10.1007/s00018-019-03161-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-019-03161-x