Abstract
The tubulin cytoskeleton is one of the main components of the cytoarchitecture and is involved in several cellular functions. Here, we examine the interplay between Listeria monocytogenes (Lm) and the tubulin cytoskeleton upon cellular infection. We show that non-polymeric tubulin is present throughout Lm actin comet tails and, to a less extent, in actin clouds. Moreover, we demonstrate that stathmin, a regulator of microtubule dynamics, is also found in these Lm-associated actin structures and is required for tubulin recruitment. Depletion of host stathmin results in longer comets containing less F-actin, which may be correlated with higher levels of inactive cofilin in the comet, thus suggesting a defect on local F-actin dynamics. In addition, intracellular bacterial speed is significantly reduced in stathmin-depleted cells, revealing the importance of stathmin/tubulin in intracellular Lm motility. In agreement, the area of infection foci and the total bacterial loads are also significantly reduced in stathmin-depleted cells. Collectively, our results demonstrate that stathmin promotes efficient cellular infection, possibly through tubulin recruitment and control of actin dynamics at Lm-polymerized actin structures.







Similar content being viewed by others
Abbreviations
- Lm :
-
Listeria monocytogenes
- MT:
-
Microtubule
- ABM:
-
Actin-based motility
- MOI:
-
Multiplicity of infection
- PTM:
-
Post-translational modifications
- STED:
-
Stimulated emission depletion
- ROI:
-
Region of interest
- MFI:
-
Mean fluorescence intensity
- WT:
-
Wild type
- GFP:
-
Green fluorescent protein
References
Haglund CM, Welch MD (2011) Pathogens and polymers: microbe–host interactions illuminate the cytoskeleton. J Cell Biol 195(1):7–17. https://doi.org/10.1083/jcb.201103148
Welch MD, Way M (2013) Arp2/3-mediated actin-based motility: a tail of pathogen abuse. Cell Host Microbe 14(3):242–255. https://doi.org/10.1016/j.chom.2013.08.011
Radhakrishnan GK, Splitter GA (2012) Modulation of host microtubule dynamics by pathogenic bacteria. Biomol Concepts 3(6):571–580. https://doi.org/10.1515/bmc-2012-0030
Geisler F, Leube RE (2016) Epithelial intermediate filaments: guardians against microbial infection? Cells 5(3):29. https://doi.org/10.3390/cells5030029
Torraca V, Mostowy S (2016) Septins and bacterial infection. Front Cell Dev Biol 4:(127). https://doi.org/10.3389/fcell.2016.00127
Rodriguez OC, Schaefer AW, Mandato CA, Forscher P, Bement WM, Waterman-Storer CM (2003) Conserved microtubule-actin interactions in cell movement and morphogenesis. Nat Cell Biol 5(7):599–609. https://doi.org/10.1038/ncb0703-599
Ray K, Marteyn B, Sansonetti PJ, Tang CM (2009) Life on the inside: the intracellular lifestyle of cytosolic bacteria. Nat Rev Microbiol 7(5):333–340. https://doi.org/10.1038/nrmicro2112
Radoshevich L, Cossart P (2018) Listeria monocytogenes: towards a complete picture of its physiology and pathogenesis. Nat Rev Microbiol 16(1):32–46. https://doi.org/10.1038/nrmicro.2017.126
Welch MD, Rosenblatt J, Skoble J, Portnoy DA, Mitchison TJ (1998) Interaction of human Arp2/3 complex and the Listeria monocytogenes ActA protein in actin filament nucleation. Science 281(5373):105–108. https://doi.org/10.1126/science.281.5373.105
Tilney LG, Portnoy DA (1989) Actin filaments and the growth, movement, and spread of the intracellular bacterial parasite, Listeria monocytogenes. J Cell Biol 109(4 Pt 1):1597–1608
Lambrechts A, Gevaert K, Cossart P, Vandekerckhove J, Van Troys M (2008) Listeria comet tails: the actin-based motility machinery at work. Trends Cell Biol 18(5):220–227. https://doi.org/10.1016/j.tcb.2008.03.001
Loisel TP, Boujemaa R, Pantaloni D, Carlier MF (1999) Reconstitution of actin-based motility of Listeria and Shigella using pure proteins. Nature 401(6753):613–616. https://doi.org/10.1038/44183
Wang J, King JE, Goldrick M, Lowe M, Gertler FB, Roberts IS (2015) Lamellipodin Is important for cell-to-cell spread and actin-based motility in Listeria monocytogenes. Infect Immun 83(9):3740–3748. https://doi.org/10.1128/iai.00193-15
Fattouh R, Kwon H, Czuczman MA, Copeland JW, Pelletier L, Quinlan ME, Muise AM, Higgins DE, Brumell JH (2015) The diaphanous-related formins promote protrusion formation and cell-to-cell spread of Listeria monocytogenes. J Infect Dis 211(7):1185–1195. https://doi.org/10.1093/infdis/jiu546
Greiffenberg L, Goebel W, Kim KS, Weiglein I, Bubert A, Engelbrecht F, Stins M, Kuhn M (1998) Interaction of Listeria monocytogenes with human brain microvascular endothelial cells: InlB-dependent invasion, long-term intracellular growth, and spread from macrophages to endothelial cells. Infect Immun 66(11):5260–5267
Kuhn M (1998) The microtubule depolymerizing drugs nocodazole and colchicine inhibit the uptake of Listeria monocytogenes by P388D1 macrophages. FEMS Microbiol Lett 160(1):87–90. https://doi.org/10.1111/j.1574-6968.1998.tb12895.x
Guzman CA, Rohde M, Chakraborty T, Domann E, Hudel M, Wehland J, Timmis KN (1995) Interaction of Listeria monocytogenes with mouse dendritic cells. Infect Immun 63(9):3665–3673
Lacayo CI, Theriot JA (2004) Listeria monocytogenes actin-based motility varies depending on subcellular location: a kinematic probe for cytoarchitecture. Mol Biol Cell 15(5):2164–2175. https://doi.org/10.1091/mbc.e03-10-0747
Mounier J, Ryter A, Coquis-Rondon M, Sansonetti PJ (1990) Intracellular and cell-to-cell spread of Listeria monocytogenes involves interaction with F-actin in the enterocytelike cell line Caco-2. Infect Immun 58(4):1048–1058
Van Troys M, Lambrechts A, David V, Demol H, Puype M, Pizarro-Cerda J, Gevaert K, Cossart P, Vandekerckhove J (2008) The actin propulsive machinery: the proteome of Listeria monocytogenes tails. Biochem Biophys Res Commun 375(2):194–199. https://doi.org/10.1016/j.bbrc.2008.07.152
Gavet O, Ozon S, Manceau V, Lawler S, Curmi P, Sobel A (1998) The stathmin phosphoprotein family: intracellular localization and effects on the microtubule network. J Cell Sci 111(Pt 22):3333–3346
Gupta KK, Li C, Duan A, Alberico EO, Kim OV, Alber MS, Goodson HV (2013) Mechanism for the catastrophe-promoting activity of the microtubule destabilizer Op18/stathmin. Proc Natl Acad Sci USA 110(51):20449–20454. https://doi.org/10.1073/pnas.1309958110
Maucuer A, Camonis JH, Sobel A (1995) Stathmin interaction with a putative kinase and coiled-coil-forming protein domains. Proc Natl Acad Sci USA 92(8):3100–3104
Pfeuffer T, Goebel W, Laubinger J, Bachmann M, Kuhn M (2000) LaXp180, a mammalian ActA-binding protein, identified with the yeast two-hybrid system, co-localizes with intracellular Listeria monocytogenes. Cell Microbiol 2(2):101–114.https://doi.org/10.1046/j.1462-5822.2000.00034.x
Glaser P, Frangeul L, Buchrieser C, Rusniok C, Amend A, Baquero F, Berche P, Bloecker H, Brandt P, Chakraborty T, Charbit A, Chetouani F, Couvé E, de Daruvar A, Dehoux P, Domann E, Domı́nguez-Bernal G, Duchaud E, Durant L, Dussurget O, Entian K-D, Fsihi H, Portillo FG-D, Garrido P, Gautier L, Goebel W, Gómez-López N, Hain T, Hauf J, Jackson D, Jones L-M, Kaerst U, Kreft J, Kuhn M, Kunst F, Kurapkat G, Madueño E, Maitournam A, Vicente JM, Ng E, Nedjari H, Nordsiek G, Novella S, de Pablos B, Pérez-Diaz J-C, Purcell R, Remmel B, Rose M, Schlueter T, Simoes N, Tierrez A, Vázquez-Boland J-A, Voss H, Wehland J, Cossart P (2001) Comparative genomics of Listeria species. Science 294(5543):849–852. https://doi.org/10.1126/science.1063447
Leitao E, Costa AC, Brito C, Costa L, Pombinho R, Cabanes D, Sousa S (2014) Listeria monocytogenes induces host DNA damage and delays the host cell cycle to promote infection. Cell Cycle 13(6):928–940. https://doi.org/10.4161/cc.27780
Mengaud J, Geoffroy C, Cossart P (1991) Identification of a new operon involved in Listeria monocytogenes virulence: its first gene encodes a protein homologous to bacterial metalloproteases. Infect Immun 59(3):1043–1049
Portnoy DA, Jacks PS, Hinrichs DJ (1988) Role of hemolysin for the intracellular growth of Listeria monocytogenes. J Exp Med 167(4):1459–1471. https://doi.org/10.1084/jem.167.4.1459
Schindelin J, Arganda-Carreras I, Frise E, Kaynig V, Longair M, Pietzsch T, Preibisch S, Rueden C, Saalfeld S, Schmid B, Tinevez JY, White DJ, Hartenstein V, Eliceiri K, Tomancak P, Cardona A (2012) Fiji: an open-source platform for biological-image analysis. Nat Methods 9(7):676–682. https://doi.org/10.1038/nmeth.2019
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH image to ImageJ: 25 years of image analysis. Nat Methods 9(7):671–675
Chong R, Squires R, Swiss R, Agaisse H (2011) RNAi screen reveals host cell kinases specifically involved in Listeria monocytogenes spread from cell to cell. PLoS One 6(8):e23399. https://doi.org/10.1371/journal.pone.0023399
Reis O, Sousa S, Camejo A, Villiers V, Gouin E, Cossart P, Cabanes D (2010) LapB, a novel Listeria monocytogenes LPXTG surface adhesin, required for entry into eukaryotic cells and virulence. J Infect Dis 202(4):551–562. https://doi.org/10.1086/654880
Shenoy VB, Tambe DT, Prasad A, Theriot JA (2007) A kinematic description of the trajectories of Listeria monocytogenes propelled by actin comet tails. Proc Natl Acad Sci USA 104(20):8229–8234. https://doi.org/10.1073/pnas.0702454104
Song Y, Brady ST (2015) Post-translational modifications of tubulin: pathways to functional diversity of microtubules. Trends Cell Biol 25(3):125–136. https://doi.org/10.1016/j.tcb.2014.10.004
Bauer S, Pfeuffer T, Kuhn M (2003) Identification and characterisation of regions in the cellular protein LaXp180 and the Listeria monocytogenes surface protein ActA necessary for the interaction of the two proteins. Mol Genet Genom 268(5):607–617. https://doi.org/10.1007/s00438-002-0775-1
Rafelski SM, Theriot JA (2005) Bacterial shape and ActA distribution affect initiation of Listeria monocytogenes actin-based motility. Biophys J 89(3):2146–2158. https://doi.org/10.1529/biophysj.105.061168
Goldberg MB (2001) Actin-based motility of intracellular microbial pathogens. Microbiol Mol Biol Rev 65(4):595–626. https://doi.org/10.1128/mmbr.65.4.595-626.2001 (table of contents)
Nanavati D, Ashton FT, Sanger JM, Sanger JW (1994) Dynamics of actin and alpha-actinin in the tails of Listeria monocytogenes in infected PtK2 cells. Cell Motil Cytoskelet 28(4):346–358. https://doi.org/10.1002/cm.970280408
Byrne FL, Yang L, Phillips PA, Hansford LM, Fletcher JI, Ormandy CJ, McCarroll JA, Kavallaris M (2014) RNAi-mediated stathmin suppression reduces lung metastasis in an orthotopic neuroblastoma mouse model. Oncogene 33(7):882–890. https://doi.org/10.1038/onc.2013.11
Rosenblatt J, Agnew BJ, Abe H, Bamburg JR, Mitchison TJ (1997) Xenopus actin depolymerizing factor/cofilin (XAC) is responsible for the turnover of actin filaments in Listeria monocytogenes tails. J Cell Biol 136(6):1323–1332
Giganti A, Plastino J, Janji B, Van Troys M, Lentz D, Ampe C, Sykes C, Friederich E (2005) Actin-filament cross-linking protein T-plastin increases Arp2/3-mediated actin-based movement. J Cell Sci 118(Pt 6):1255–1265. https://doi.org/10.1242/jcs.01698
Bierne H, Gouin E, Roux P, Caroni P, Yin HL, Cossart P (2001) A role for cofilin and LIM kinase in Listeria-induced phagocytosis. J Cell Biol 155(1):101–112. https://doi.org/10.1083/jcb.200104037
Theriot JA, Rosenblatt J, Portnoy DA, Goldschmidt-Clermont PJ, Mitchison TJ (1994) Involvement of profilin in the actin-based motility of L. monocytogenes in cells and in cell-free extracts. Cell 76(3):505–517. https://doi.org/10.1016/0092-8674(94)90114-7
Buchwalow IB, Emoto M, Brich M, Kaufmann SHE (1997) Involvement of tubulin and inhibitory G proteins in the interaction of Listeria monocytogenes with mouse hepatocytes. Infect Immun 65(3):1095–1097
Sanger JM, Sanger JW (2012) Insights into cell division using Listeria monocytogenes infections of PtK2 renal epithelial cells. Cytoskeleton (Hoboken) 69(11):992–999. https://doi.org/10.1002/cm.21076
Zheng K, Kitazato K, Wang Y, He Z (2016) Pathogenic microbes manipulate cofilin activity to subvert actin cytoskeleton. Crit Rev Microbiol 42(5):677–695. https://doi.org/10.3109/1040841x.2015.1010139
Henmi Y, Tanabe K, Takei K (2011) Disruption of microtubule network rescues aberrant actin comets in dynamin2-depleted cells. PLoS One 6(12):e28603. https://doi.org/10.1371/journal.pone.0028603
Yoshida S, Handa Y, Suzuki T, Ogawa M, Suzuki M, Tamai A, Abe A, Katayama E, Sasakawa C (2006) Microtubule-severing activity of Shigella is pivotal for intercellular spreading. Science 314(5801):985–989. https://doi.org/10.1126/science.1133174
Kocks C, Hellio R, Gounon P, Ohayon H, Cossart P (1993) Polarized distribution of Listeria monocytogenes surface protein ActA at the site of directional actin assembly. J Cell Sci 105(Pt 3):699–710
Rafelski SM, Theriot JA (2006) Mechanism of polarization of Listeria monocytogenes surface protein ActA. Mol Microbiol 59(4):1262–1279. https://doi.org/10.1111/j.1365-2958.2006.05025.x
Archuleta TL, Du Y, English CA, Lory S, Lesser C, Ohi MD, Ohi R, Spiller BW (2011) The Chlamydia effector chlamydial outer protein N (CopN) sequesters tubulin and prevents microtubule assembly. J Biol Chem 286(39):33992–33998. https://doi.org/10.1074/jbc.m111.258426
Acknowledgements
This work received funding from Norte-01-0145-FEDER-000012—Structured program on bioengineered therapies for infectious diseases and tissue regeneration, supported by Norte Portugal Regional Operational Programme (NORTE 2020), under the PORTUGAL 2020 Partnership Agreement, through the European Regional Development Fund (FEDER). ACC and FC were supported by a Fundação para a Ciência e Tecnologia (FCT) Post-doctoral Fellowship (SFRH/BPD/88769/2012) and Ph.D. fellowship (SFRH/BD/61825/2009), respectively, through FCT/MEC co-funded by QREN and POPH (Programa Operacional Potencial Humano). SS was supported by FCT Investigator program (COMPETE, POPH, and FCT). We thank ALM unit from IBMC/i3S for technical support, B. Fonseca (FFUP) for the BeWo cell line and H. Maiato, C. Sunkel and J. B. Relvas laboratories (IBMC/i3S) for sharing reagents. We are also thankful to J. Ferreira and A. Pereira for fruitful discussions.
Author information
Authors and Affiliations
Contributions
ACC, DC and SS conceived and designed the experiments; ACC and FC performed the experiments; ACC, FC, DC and SS analyzed the data; DC and SS obtained the funding; ACC and SS wrote the manuscript.
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
18_2018_2977_MOESM1_ESM.tif
Table S1 (relative to Fig. 6b) Stathmin is required for Lm maximal intracellular speed. Control (siC) and stathmin-depleted (siStat) HeLa cells were infected with Lm and imaged by time-lapse microscopy from 7 to 14 h post-infection. Values for Lm speed are mean ± SD of approximately 15 bacteria per condition, in each of the four independent experiments. Fold change (siC/siStat) and statistical significance (p value, Student t test) were calculated (TIFF 156 kb)
18_2018_2977_MOESM2_ESM.tif
Table S2 (relative to Fig. 6d) Stathmin is required for Lm cell-to-cell spreading. Monolayers of control (siC) and stathmin-depleted (siStat) HeLa cells were infected for 24 h with MOI 0.5, fixed and labeled for Lm. Quantification of infection foci areas was performed after multiple image alignment. Values are mean ± SD of at least 60 infection foci areas, per each of the three independent experiments. Fold change (siC/siStat) and statistical significance (p value, Student t test) were calculated (TIFF 114 kb)
18_2018_2977_MOESM3_ESM.tif
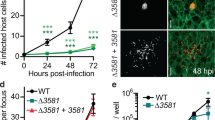
Figure S1 (relative to Fig. 1) Tubulin comets are ubiquitously detected in different Lm strains and cell lines. (a to c) Cells were infected for 7 h and subjected to cold shock prior to fixation for immunofluorescence analysis. Representative Z-stack projections of deconvoluted images are shown. Insets shown as high-magnification composite (merge) and single-channel images displaying tubulin (red), actin (green) and DNA (blue). Scale bars represent 10 µm. (a) Caco-2 cells were infected with Lm strains EGD and 10403S. (b) Human (HeLa, Jeg-3, BeWo), murine (J774) and rat kangaroo (Ptk2) cell lines were infected with Lm EGDe. (c) Quantification of the percentage of Lm actin comets and clouds showing tubulin accumulation in HeLa and Ptk2 cells. Results are mean ± SEM of three independent experiments (n ~ 100 per experiment) (TIFF 3262 kb)
18_2018_2977_MOESM4_ESM.tif
Figure S2 (relative to Fig. 2) Nocodazole treatment does not perturb the formation of Lm-associated tubulin comets. Caco-2 cells infected with Lm. Following the bacterial internalization step (1 h), cells were incubated with nocodazole (or DMSO as control) for 6 h, subjected to cold shock, fixed and labeled for tubulin (red), actin (green) and DNA (blue) to be analyzed by fluorescence microscopy. The micrographs are Z axis projections of deconvolutes images. Quantification of the percentage of Lm showing tubulin accumulation together with actin comets is shown in Fig. 2e. Scale bars correspond to 10 µm (TIFF 3340 kb)
18_2018_2977_MOESM5_ESM.tif
Figure S3 (relative to Fig. 4) Efficiency of stathmin depletion in HeLa cells. (a) Western blot analysis of stathmin expression in HeLa cells treated with control (siC) or with stathmin (siStat) siRNA. GAPDH levels were used as loading control. (b) Quantification of siRNA-mediated silencing of stathmin as evaluated by densitometric analysis of Western blot bands, normalized for GAPDH expression. Results are mean ± SD from four independent experiments. Asterisks indicate statistical significance as determined by Student t test: p < 0.0001 (****) (TIFF 95 kb)
18_2018_2977_MOESM6_ESM.tif
Figure S4 (relative to Fig. 5) Efficiency of stathmin depletion in HeLa cells. Stathmin expression levels evaluated by densitometric analysis of Western blot bands, normalized for GAPDH expression. Results are mean ± SD of four independent experiments. Asterisks indicate statistical significance as determined by Student t test: p < 0.01 (**) (TIFF 50 kb)
18_2018_2977_MOESM7_ESM.avi
Movie S1 (relative to Fig. 3) Tubulin coats actin comet tails polymerized by Lm. HeLa cells infected with Lm were labeled for tubulin (red), Lm (WGA, cyan) and actin (SiRactin, green). Movie shows a 360º rotation of the Lm comet tail obtained by three-dimensional reconstitution of stimulated emission depletion (STED) super-resolution microscopy images (AVI 138248 kb)
18_2018_2977_MOESM8_ESM.avi
Movies S2 and S3 (relative to Fig. 6a, b and Table S1) Stathmin controls Lm speed. Control (Movie S2) and stathmin-depleted cells (Movie S3) infected with Lm were subjected to phase-contrast live-imaging from 7 to 14 h post-infection. Arrows indicate the position of Lm throughout time and the black line shows the described path. Videos are reproduced at 5 frames per second (AVI) (AVI 181 kb)
Rights and permissions
About this article
Cite this article
Costa, A.C., Carvalho, F., Cabanes, D. et al. Stathmin recruits tubulin to Listeria monocytogenes-induced actin comets and promotes bacterial dissemination. Cell. Mol. Life Sci. 76, 961–975 (2019). https://doi.org/10.1007/s00018-018-2977-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-018-2977-7