Abstract
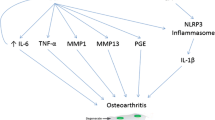
Osteoarthritis (OA) is a whole-joint disease characterized by articular cartilage loss, tissue inflammation, abnormal bone formation and extracellular matrix (ECM) mineralization. Disease-modifying treatments are not yet available and a better understanding of osteoarthritis pathophysiology should lead to the discovery of more effective treatments. Gla-rich protein (GRP) has been proposed to act as a mineralization inhibitor and was recently shown to be associated with OA in vivo. Here, we further investigated the association of GRP with OA mineralization–inflammation processes. Using a synoviocyte and chondrocyte OA cell system, we showed that GRP expression was up-regulated following cell differentiation throughout ECM calcification, and that inflammatory stimulation with IL-1β results in an increased expression of COX2 and MMP13 and up-regulation of GRP. Importantly, while treatment of articular cells with γ-carboxylated GRP inhibited ECM calcification, treatment with either GRP or GRP-coated basic calcium phosphate (BCP) crystals resulted in the down-regulation of inflammatory cytokines and mediators of inflammation, independently of its γ-carboxylation status. Our results strengthen the calcification inhibitory function of GRP and strongly suggest GRP as a novel anti-inflammatory agent, with potential beneficial effects on the main processes responsible for osteoarthritis progression. In conclusion, GRP is a strong candidate target to develop new therapeutic approaches.








Similar content being viewed by others
References
Egloff C, Hügle T, Valderrabano V (2012) Biomechanics and pathomechanisms of osteoarthritis. Eur J Med Sci 142:w13583
Abramson SB, Attur M (2009) Developments in the scientific understanding of osteoarthritis. Arthritis Res Ther 11:227–235
Blanco FJ (2014) Osteoarthritis: something is moving. Reumatol Clin 10:4–5
Fuerst M, Bertrand J, Lammers L, Dreier R, Echtermeyer F, Nitschke Y, Nitschke Y, Rutsch F, Schäfer FK, Niggemeyer O, Steinhagen J, Lohmann CH, Pap T, Rüther W (2009) Calcification of articular cartilage in human osteoarthritis. Arthritis Rheum 60:2694–2703
Rosenthal AK (2011) Crystals, inflammation, and osteoarthritis. Curr Opin Rheumatol 23:170–173
Hernandez-Santana A, Yavorskyy A, Loughran ST, McCarthy GM, McMahon GP (2011) New approaches in the detection of calcium-containing microcrystals in synovial fluid. Bioanalysis 3:1085–1091
Liu YZ, Jackson AP, Cosgrove SD (2009) Contribution of calcium-containing crystals to cartilage degradation and synovial inflammation in osteoarthritis. Osteoarth Cartil 17:1333–1340
Sokolove J, Lepus CM (2013) Role of inflammation in the pathogenesis of osteoarthritis: latest findings and interpretations. Ther Adv Musculoskel Dis 5:77–94
Kraus VB, Blanco F, Englund M, Karsdal MA, Lohmander LS (2015) Call for standardized definitions of osteoarthritis and risk stratification for clinical trials and clinical use. Osteoarth Cartil 23:1233–1241
Shroff RC, Shanahan CM (2007) The vascular biology of calcification. Semin Dial 20:103–109
Luo G, Ducy P, Mckee MD, Pinero GJ, Loyer E, Behringer RR, Karsenty G (1997) Spontaneous calcification of arteries and cartilage in mice lacking matrix Gla protein. Nature 386:78–81
Shea M, Booth SL, Massaro JM, Jacques PF, D’Agostinho RB, Dawson-Hughes B, Ordovas JM, O’Donnell CJ, Kathiresan S, Keaney JF Jr, Vasan RS, Benjamin EJ (2008) Vitamin K and vitamin D status: associations with inflammatory markers in the Framingham Offspring Study. Am J Epidemiol 167:313–320
Wallin R, Wajih N, Hutson SM (2008) VKORC1: a warfarin-sensitive enzyme in vitamin K metabolism and biosynthesis of vitamin K-dependent blood coagulation factors. Vitam Horm 78:227–246
Misra D, Booth SL, Tolstykh I, Felson DT, Nevitt MC, Lewis CE, Torner J, Neogi T (2013) Vitamin K deficiency is associated with incident knee osteoarthritis. Am J Med 126:243–248
Neogi T, Booth SL, Zhang YQ, Jacques PF, Terkeltaub R, Aliabadi P, Felson DT (2006) Low vitamin K status is associated with osteoarthritis in the hand and knee. Arthritis Rheum 54:1255–1261
Naito K, Watari T, Obayashi O, Katsube S, Nagaoka I, Kaneko K (2011) Relationship between serum undercarboxylated osteocalcin and hyaluronan levels in patients with bilateral knee osteoarthritis. Int J Mol Med 29:756–760
Silaghi C, Fodor D, Cristea V, Crãciun AM (2012) Synovial and serum levels of uncarboxylated matrix Gla-protein (ucMGP) in patients with arthritis. Clin Chem Lab Med 50:125–128
Viegas CSB, Simes DC, Laizè V, Williamson MK, Price PA, Cancela ML (2008) Gla-rich Protein (GRP), a new vitamin K-dependent protein identified from sturgeon cartilage and highly conserved in vertebrates. J Biol Chem 283:36655–36664
Viegas CSB, Cavaco S, Neves PL, Ferreira A, João A, Williamson MK, Price PA, Cancela ML, Simes DC (2009) Gla-rich protein (GRP) is a novel vitamin K dependent protein present in serum and accumulated at sites of pathological calcifications. Am J Pathol 175:2288–2298
Viegas CSB, Herfs M, Rafael MS, Enriquez JL, Teixeira A, Luís I, van ‘t Hoofd C, João A, Maria VL, Cavaco S, Ferreira A, Serra M, Theuwissen E, Vermeer C, Simes, DC (2014) Gla-rich protein is a potential new vitamin K target in cancer: Evidences for a direct GRP-mineral interaction. Biomed Res Int. doi:10.1155/2014/340216
Viegas CSB, Rafael M, Enriquez JL, Teixeira A, Vitorino R, Luís IM, Costa RM, Santos S, Cavaco S, Neves J, Willems B, Vermeer C, Simes DC (2015) Gla-rich protein (GRP) acts as a calcification inhibitor in the human cardiovascular system. Arterioscler Thromb Vasc Biol 35:399–408
Eitzinger N, Surmann-Schmitt C, Bosl M, Schett G, Engelke K, Hess A, von der Mark K, Stock M (2012) Ucma is not necessary for normal development of the mouse skeleton. Bone 50:670–680
Neacsu CD, Grosch M, Tejada M, Winterpacht A, Paulsson M, Wagener R, Tagariello A (2011) Ucmaa (Grp-2) is required for zebrafish skeletal development. Evidence for a functional role of its glutamate γ-carboxylation. Matrix Biol 30:369–378
Surmann-Schmitt C, Dietz U, Kireva T, Adam N, Park J, Tagariello A, Onnerfjord P, Heinegård D, Schlötzer-Schrehardt U, Deutzmann R, von der Mark K, Stock M (2008) Ucma, a novel secreted cartilage-specific protein with implications in osteogenesis. J Biol Chem 283:7082–7093
Tagariello A, Luther J, Streiter M, Didt-Koziel L, Wuelling M, Surmann-Schmitt C, Stock M, Adam N, Vortkamp A, Winterpacht A (2008) Ucma-A novel secreted factor represents a highly specific marker for distal chondrocytes. Matrix Biol 27:3–11
Lee YJ, Park SY, Lee SJ, Boo YC, Choi JY, Kim JE (2015) Ucma, a direct transcriptional target of Runx2 and Osterix, promotes osteoblast differentiation and nodule formation. Osteoarth Cartil. doi:10.1016/j.joca.2015.03.035
Rafael MS, Cavaco S, Viegas CSB, Santos S, Ramos A, Willems B, Herfs M, Theuwissen E, Vermeer C, Simes DC (2014) Insights into the association of Gla-rich protein and osteoarthritis, novel splice variants and γ-carboxylation status. Mol Nutr Food Res. doi:10.1002/mnfr.201300941
Ortiz-Delgado JB, Simes DC, Viegas CSB, Schaff BJ, Sarasquete C, Cancela ML (2006) Cloning of matrix Gla protein in amarine cartilaginous fish, Prionace glauca: preferential protein accumulation in skeletal and vascular systems. Histochem Cell Biol 126:89–101
Burguera EF, Vela AA, Magalhães J, Meijide-Faílde R, Blanco FJ (2014) Effect of hydrogen sulfide sources on inflammation and catabolic markers on interleukin 1β-stimulated human articular chondrocytes. Osteoarth Cartil 22:1026–1035
Cillero PB, Martin M, Arenas J, Lopez-Armada MJ, Blanco FJ (2011) Effect of nitric oxide on mitochondrial activity of human synovial cells. B Musculoskelet Disord 12:1471–2474
Chomczynski P, Sacchi N (1987) Single-step method of RNA isolation by acid guanidinium thiocyanate phenol chloroform extraction. Anal Biochem 162:156–159
Schurgers LJ, Teunissen KJ, Knapen MH, Kwaijtaal M, van Diest R, Appels A, Reutelingsperger CP, Cleutjens JP, Vermeer C (2005) Novel conformation-specific antibodies against matrix gamma-carboxyglutamic acid (Gla) protein: undercarboxylated matrix Gla protein as marker for vascular calcification. Arterioscler Thromb Vasc Biol 25:1629–1633
Pombinho AR, LaizéV Molha DM, Marques SMP, Cancela ML (2004) Development of two bone-derived cell lines from the marine teleost Sparus aurata; evidence for extracellular matrix mineralization and cell-type-specific expression of matrix Gla protein and osteocalcin. Cell Tissue Res 315:393–406
McCarthy GM, Westfall P, Masuda I, Christopherson PA, Cheung HS, Mitchell PG (2001) Basic calcium phosphate crystals activate human osteoarthritic synovial fibroblasts and induce matrix metalloproteinase-13 (collagenase-3) in adult porcine articular chondrocytes.Ann Rheum Dis Aquat Organ 60:399–406
Nakatani S, Mano H, Ryanghyok IM, Shimizu J, Wada M (2006) Excess magnesium inhibits excess calcium-induced matrix-mineralization and production of matrix gla protein (MGP) by ATDC5 cells. Biochem Biophys Res Commun 348:1157–1162
Wallin R, Schurgers LJ, Loeser RF (2010) Biosynthesis of the vitamin K-dependent matrix Gla protein (MGP) in chondrocytes: a fetuin-MGP protein complex is assembled in vesicles shed from normal but not from osteoarthritic chondrocytes. Osteoarth Cartil 16:1096–1103
Ea HK, Nguyen C, Bazin D, Bianchi A, Jérôme G, Pascal R, Daudon M, Frédéric Lioté (2011) Articular cartilage calcification in osteoarthritis. Arthritis Rheum 63:10–18
Smith ER, Hanssen E, McMahon LP, Holt SG (2013) Fetuin-A-containing calciprotein particles reduce mineral stress in the macrophage. PLoS One 8:e60904
Cranenburg EC, Schurgers LJ, Vermeer C (2007) Vitamin K: the coagulation vitamin that became omnipotent. Thromb Haemostasis 98:120–125
Zoch ML, Clemens TL, Riddle RC (2015) New insights into the biology of osteocalcin. Bone. doi:10.1016/j.bone.2015.05.046
Yamaguchi M, Weitzmann M (2010) Vitamin K2 stimulates osteoblastogenesis and suppresses osteoclastogenesis by suppressing NF-κB activation. Int J Mol Med 27:3–14
Roman-Blas JA, Jimenez SA (2006) NF-κB as a potential therapeutic target in osteoarthritis and rheumatoid arthritis. Osteoarthritis Cartilage 14:839–848
Pobirci O, Bogdan F, Pobirci DD, Rosca E, Petcu CA (2011) The study of synovities with articular inflammatory liquid, through clinical-statistical, histological and immunohistochemical methods. Rom J Morphol Embryol 52:333–338
Acknowledgments
This work was funded by projects PTDC/SAU-ORG/112832/2009, PTDC/SAU-ORG/117266/2010 and PTDC/BIM-MEC/1168/2012, and also through Project UID/Multi/04326/2013, all from the Portuguese Science and Technology Foundation (FCT). S. Cavaco, C. S. B. Viegas and M. S. Rafael were the recipients of the FCT fellowships SFRH/BD/60867/2009, SFRH/BPD/70277/2010 and SFRH/BPD/89188/2012, respectively. Authors acknowledge the Orthopedics and Traumatology Service, Algarve Medical Centre (CHAlgarve), Faro, for providing the biological samples used in this study, and to Rheumatologic and Orthopedic Services of CHUAC for their help in obtaining cartilage samples. CIBER-BBN is a Spanish initiative from ISCIII.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cavaco, S., Viegas, C.S.B., Rafael, M.S. et al. Gla-rich protein is involved in the cross-talk between calcification and inflammation in osteoarthritis. Cell. Mol. Life Sci. 73, 1051–1065 (2016). https://doi.org/10.1007/s00018-015-2033-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-015-2033-9