Abstract
Influenza viruses are major human pathogens responsible for respiratory diseases affecting millions of people worldwide and characterized by high morbidity and significant mortality. Influenza infections can be controlled by vaccination and antiviral drugs. However, vaccines need annual updating and give limited protection. Only two classes of drugs are currently approved for the treatment of influenza: M2 ion channel blockers and neuraminidase inhibitors. However, they are often associated with limited efficacy and adverse side effects. In addition, the currently available drugs suffer from rapid and extensive emergence of drug resistance. All this highlights the urgent need for developing new antiviral strategies with novel mechanisms of action and with reduced drug resistance potential. Several new classes of antiviral agents targeting viral replication mechanisms or cellular proteins/processes are under development. This review gives an overview of novel strategies targeting the virus and/or the host cell for counteracting influenza virus infection.
Similar content being viewed by others
Introduction
Influenza viruses (IV) represent one of the major threats to public health, as they are responsible for both epidemics and pandemics characterized by high morbidity and mortality. During the past century, the pandemics of Spanish flu (1918), Asian flu (1957), Hong Kong flu (1968), and recently, swine flu (2009) caused millions of deaths worldwide [1]. In addition, the seasonal influenza epidemics result in hundreds of thousands of deaths per year (http://www.who.int). IV belong to the Orthomyxoviridae family and include A, B, and C types, which differ in host range and pathogenicity. In particular, influenza A viruses (IAV) infect a wide range of avian and mammalian hosts, while influenza B viruses (IBV) infect almost exclusively humans. IAV are further classified into subtypes based on the antigenic properties of two viral surface glycoproteins, hemagglutinin (HA) and neuraminidase (NA): 17 HA (H1–H17) and 10 NA (N1–N10) antigenic subtypes have so far been identified [2]. Within a subtype, different strains can arise as a result of point mutations; indeed, IAV evolve constantly and new mutant strains replace the old ones in a process known as “antigenic drift”.
IAV possess a single-stranded, eight-segmented RNA genome of negative polarity, which encodes: the surface glycoproteins HA and NA and the M2 ion channel, that are all inserted into the viral lipid envelope; the matrix protein 1 (M1), that lies beneath the membrane; the three subunits (PB1, PB2, and PA) of the RNA polymerase complex, that is associated with the encapsidated genome; the nucleoprotein (NP), that coats the viral genome; and the nonstructural proteins NS1 and NS2/NEP [2]. In addition, most IAV encode a nonstructural PB1-F2 protein of varying length, which has pro-apoptotic functions [3], and PA-x and N40, two newly identified proteins encoded by the PA and PB1 genes, respectively [4, 5]. As depicted in Fig. 1, the IV replication cycle initiates with the attachment of HA to sialic acid (SA)-containing glycoprotein and glycolipid receptors on the cell surface [6]. The virus particle then enters the cell via clathrin-dependent endocytosis and macropinocytosis. Following entry, the acidic environment of the late endosome triggers a conformational change of HA which drives fusion of the viral envelope with the endosomal membrane. Moreover, the M2 protein creates a proton flow from the endosome into the virion leading to the dissociation of M1 from the viral ribonucleoprotein complexes (vRNPs). The released vRNPs are then transported into the nucleus, wherein the viral RNA polymerase initiates genome transcription and replication. Newly synthesized viral genome segments and proteins (PB1, PB2, PA, and NP) are complexed with M1 and NEP and then exported from the nucleus to the cell membrane for the final assembly and budding phases. Finally, NA cleaves terminal SA residues from HA and the cellular receptors, permitting the release of virions from the cell. In addition to the viral proteins, there are a number of cellular proteins involved in each stage of IV replication, which could represent potential antiviral targets [7].
Antiviral strategies targeting viral functions essential for influenza virus (IV) replication. The first step of IV infection is the interaction between viral hemagglutinin (HA) and cellular sialic acid (SA)-containing receptors, resulting in the attachment of the virion to the target cell. Attachment inhibitors, such as monoclonal antibodies (mAb) directed against the globular head of HA, natural and synthetic compounds containing SA, HA-binding peptides, and compounds that recognize glycosylation sites of HA, interfere with this process and block IV infection. After the internalization of the virion by endocytosis and macropinocytosis, HA mediates the fusion of the viral envelope with the endosomal membrane, a pH-dependent process that can be inhibited by fusion inhibitors such as small molecules that inhibit the low pH-induced conformational change of HA (e.g., arbinol) and neutralizing mAbs directed against the stem region of HA. The activity of the viral protonic pump M2 leads to the acidification of the endosome and to viral uncoating, followed by the release of the viral ribonucleoprotein (vRNP) into the cytoplasm. M2 inhibitors such as adamantanes block IAV (but not IBV) replication at this step. After nuclear translocation of the vRNP, the viral genomic segments are transcribed by the viral RNA-dependent RNA polymerase (RdRP) into mRNAs that are then transported into the cytoplasm and translated into viral proteins necessary for viral genome replication, also catalyzed by viral RdRP. Compounds able to interfere with RdRP activities can inhibit both transcription and replication steps. Transcription, replication, correct assembly of vRNPs, and their nuclear export require the activity of viral nucleoprotein (NP), thus molecules targeting NP functions have demonstrated effective anti-influenza activity. After the assembly of new virions in the cytoplasm, they are transported at the cell membrane and then released by budding. The activity of neuraminidase (NA) present on the virion surface is essential for the cleavage of SA molecules from HA and to allow the release of viral particles. NA inhibitors such as zanamivir and oseltamivir block IAV and IBV replication by interfering with this step
The current options to combat IV infection include vaccination and two classes of antiviral compounds, the M2 ion channel blockers (adamantanes) and the NA inhibitors. However, vaccines need to be reformulated each year due to the genetic variability of the virus and are not always protective; in addition, a rapidly emerging influenza pandemic cannot be contained by vaccination. Adamantanes inhibit IAV replication by blocking virus entry. However, they have no activity against IBV and are often associated with serious side effects. NA inhibitors block the release of virions after budding from the host cell. They exhibit activity against both IAV and IBV but can also cause side effects. In addition, a major problem with both classes of drugs is the rapid emergence of drug-resistant viral strains which have limited the use of the NA inhibitors and rendered the M2 blockers ineffective. Thus, novel IV inhibitors are greatly required.
The need for developing new drugs to overcome resistance and counteract threats of sporadic outbreaks of viruses with pandemic potential has fueled the interest in gaining a deeper knowledge of the structures and functions of the viral components. The body of information coming out of new research initiatives may have the potential to be developed into useful therapeutic strategies. In this review, we present an overview of recent progress in designing and developing new antiviral compounds and strategies to block critical steps of the viral life cycle by inhibiting functions of viral proteins and/or host–virus interactions. In particular, the review focuses mainly on those that in our opinion are the most novel and promising anti-influenza strategies and/or on those compounds that should deserve further attention for developing new therapeutic approaches.
New virus-based anti-influenza virus strategies
In this section, we will discuss current and new anti-influenza approaches from the point of view of targeting the virus itself. A number of novel virus-based anti-influenza strategies are being developed, which include improving currently available drugs in potency, spectrum of activity, or route of delivery, discovering new classes of compounds that target different viral proteins, and the application of combination therapy.
Antiviral strategies targeting the M2 ion channel
Influenza M2 is a homotetrameric protein that acts as a proton channel [8]. After virus endocytosis into the host cell, M2 is activated in response to the low pH in the endosomal lumen and creates a proton flux from the endosome into the virion core (Fig. 1, top left) [8]. M2 is essential for viral replication and its short N-terminal extracellular domain is highly conserved in all human IAV [9]. For these reasons, M2 is considered an excellent target for antiviral agents. Indeed, the M2 protein of IAV (A/M2) is the target of two already licensed drugs for influenza treatment: the M2 blockers amantadine and its methyl derivative rimantadine (Fig. 1, top left) [10, 11]. Both these adamantane derivatives bind the N-terminal channel lumen of the M2 pore and, upon binding, their charged amino group produces a positive electrostatic potential in the channel lumen, which involves an electrostatic repulsion of protons and prevents virus uncoating [10]. Unfortunately, these compounds are inactive against IBV, since the amino acid sequences between the A/M2 and the M2 of IBV are different [8]. Moreover, the rapid emergence of drug-resistant virus variants represents the main limit of these drugs. Indeed, almost all currently circulating IAV are resistant to amantadine and rimantadine, greatly limiting their utility in the clinical practice [12]. Thus, new M2 blockers active against amantadine-resistant viruses are urgently needed.
Drug-resistance to amantadine and rimantadine is associated with single or multiple amino acid substitutions at positions 26, 27, 30, 31, or 34 in the transmembrane region of A/M2 located outside of the H37xxxW41 motif required for channel activity and proton selectivity [13]. More than 95 % of the reported transmissible IAV carry the S31N mutation in the transmembrane region of A/M2 [14, 15]. For this reason, the possibility to target the predominant S31N mutant represents an attractive challenge. Recently, some small molecules were identified as potent inhibitors of the A/M2-S31N variant (Table 1). Among these compounds, M2WJ332 exhibited an antiviral activity against the A/M2-S31N variant higher than that of amantadine against the wild-type A/M2 [15]. In addition to M2WJ332, some benzyl-substituted amantadine derivatives were recently found to inhibit the activity of both S31N and wild-type viruses [16]. Other frequent mutations in the A/M2 protein that confer resistance to amantadine and rimantadine are L26F and V27A [13, 17]. Recently, novel small molecules with inhibitory activity against A/M2 bearing these mutations have been reported (Table 1), including spiroadamantane 9 [18], spiran amine 8 [19], and some organosilane-based compounds [20]. Finally, a neutralizing antibody directed against the A/M2 ion channel, M2-7A, which is able to inhibit the replication of both amantadine-sensitive and amantadine-resistant viruses with similar IC50 values, has been identified [21]. Importantly, passive immunotherapy with M2-7A protected mice from a lethal IV challenge [21]. Although the exact mechanism of action of M2-7A and its binding epitope have not yet been clarified, this inhibitor clearly deserves further investigation.
To date, there is no single M2 blocker capable of targeting both wild-type IV and all circulating amantadine-resistant strains. Nevertheless, the therapy with a combination of these inhibitors could provide an effective strategy to solve the problem of amantadine resistance.
Antiviral strategies targeting the neuraminidase
Another target for the development of new anti-influenza drugs is provided by the viral neuraminidase. NA is a homotetrameric glycoside hydrolase that binds and removes a terminal SA residue from the adjacent oligosaccharide moiety of the cellular receptors recognized by HA, playing a key role in promoting IV infectivity (for a review, see [22]). Indeed, NA is responsible for virus penetration through mucosal secretions, helping the virus to access the target cells by mucus degradation [23]. Moreover, NA allows the detachment of the virion from infected cells and avoids the self-aggregation of progeny virions at late stages of infection by disrupting HA–SA interactions, thus promoting the release and spread of IV (Fig. 1, bottom right) [22].
The most successful NA inhibitors are represented by synthetic analogues of SA. Their mechanism of action is based on the competition with the natural substrate of NA, resulting in a block of the enzyme active site. These compounds are active against both IAV and IBV since NA active site is highly conserved [24, 25]. However, these inhibitors are effective against influenza infection only if administrated within 36-48 h of symptoms onset [25]. Zanamivir and oseltamivir, currently used worldwide as therapeutic and prophylactic agents against IAV and IBV, belong to this class. Zanamivir (GG167) is a 4-deoxy-4-guanidino analogue of SA and was the first approved NA inhibitor [26]. This compound is administrated in patients at least 7 years old via inhalation, due to its poor oral bioavailability (less than 20 %) [27]. Although zanamivir is well tolerated and has few adverse effects, the route of administration of this drug can represent a problem especially for children and elderly patients that might not be able to inhale zanamivir suitably [28]. To circumvent these issues, an intravenous formulation of zanamivir has been developed and is currently under Phase III clinical trial. Oseltamivir (GS4104) is an ethyl ester prodrug, which is orally administrated and is quickly converted into its active form, oseltamivir carboxylate (GS4071), by hepatic esterases. Oseltamivir possesses higher bioavailability (around 80 %) than zanamivir and can be administrated in patients ≥1 year old [29]. More recently, two other SA analogues, peramivir and laninamivir (Table 1), have been licensed in some Asian countries and are currently under clinical evaluation in other countries. Peramivir (BCX-1812, RWJ-270201) is a cyclopentane compound, approved in Japan as Rapiacta and in South Korea as Peramiflu for use in adult and pediatric patients with IAV and IBV infection [30]; in addition, it is currently undergoing clinical trials in the USA and in other countries. This compound is administrated only as an intravenous formulation, due to its very low bioavailability [31]. Laninamivir (R-125489) is an SA analogue structurally similar to zanamivir. Laninamivir is administrated as an octanoyl prodrug, laninamivir octanoate (LO; CS-8958, R-118958), and holds great promise for its long-acting inhibitory activity [32]. LO has been approved in Japan for clinical use (as Inavir) since September 2010, but is still undergoing clinical trials in the USA.
Although the NA active site is a highly conserved target, a number of mutations in the NA of viruses selected in vitro in the presence of NA inhibitors and also in patients have been identified, namely substitutions of residues E105, E119, I122, Q136, D151, R152, D198, R224, S246, H274, R292, N294, and R371 [33]. Some mutations were found to confer resistance to certain NA inhibitors but preserve the susceptibility to others [33]; for example, the main mutation conferring resistance to oseltamivir (H274Y) also confers resistance to peramivir, but not to zanamivir [34]. Currently, no mutation associated with resistance to laninamivir has been identified [33]. Based on NA crystallographic structure and knowledge of the binding mode of these NA inhibitors with the enzyme active site [35], many other analogues of SA have been synthesized and characterized in order to further optimize the binding properties of NA inhibitors. Work on novel anti-NA compounds obtained by structure-based drug design strategies has been well summarized in other recent reviews [35, 36].
An alternative and interesting approach to increase the efficacy of the approved NA drugs is provided by the use of multivalent inhibitors and by conjugating the compounds to a biocompatible polymer [37, 38]. Indeed, several reports have shown that such multivalent presentation of NA inhibitors results in a dramatically higher antiviral potency than that obtained with the monomeric drug [38–40]. As an example, zanamivir attached to the biodegradable polymer poly-l-glutamine exhibited an antiviral activity 1,000- to 10,000-fold higher than that of monomeric zanamivir [38]. In addition, a single dose of zanamivir dimers resulted in an in vivo longer-lasting activity against IV compared to monomeric zanamivir [40].
Antiviral strategies targeting the hemagglutinin
HA is a homotrimeric glycoprotein composed of a stem domain supporting a globular head. Each HA monomer consists of two disulfide-linked polypeptides, HA1 and HA2, derived from proteolytic cleavage of the single immature precursor HA0 by host proteases (see below). HA is essential for the interaction of the virus with cells by binding to SA receptors on host cells and is also involved in the low pH-induced membrane fusion between the viral envelope and the endosomal membrane (Fig. 1, top left) [22].
A possible mechanism of inhibition of viral infection is to block the interaction between viral surface molecules and cellular receptors and the subsequent fusion of the virion with the endosome membranes, thus preventing the entry of the virus into the host cell. Some drugs already approved for the treatment of human immunodeficiency virus (HIV) infection (enfuvirtide and maraviroc) and for the prevention of respiratory syncytial virus (RSV) infection (palivizumab) specifically act by interfering with viral entry [41–43]. In the case of IV, the entry step can be blocked mainly by two strategies: (1) by preventing the binding between the viral HA and the terminal SA of glycoproteins and glycolipids present on the cell membrane; and (2) by blocking the process of fusion between the viral envelope and endosome membrane, necessary for the release of the vRNP into the cytoplasm (Fig. 1, top left).
As for the first strategy, a variety of antiviral agents have been reported that can interfere with IV attachment to target cells. These inhibitors can be divided in different groups based on chemical properties and their mechanism of action:
-
1.
Neutralizing monoclonal antibodies (mAbs) directed against the membrane-distal globular head domain of HA. The globular head of HA contains three receptor binding sites (RBS) and is responsible for the attachment to cellular receptors [22], thus representing the main target of these inhibitors. The main limit of this antiviral strategy is the hyper-variability of the globular head of HA. In fact, several of the antibodies that target regions of globular head of HA are often able to neutralize a limited range of IV strains [44, 45]. Despite the overall variability of the globular head of HA, the RBS is relatively conserved; thus, antibodies against this site possess a broader spectrum of neutralizing activity. An example is represented by CH65 (Table 1), which is able to neutralize several strains of IV in vitro, by inserting its heavy-chain CDR3 loop into the receptor-binding pocket [45]. Recent studies reported a few examples of RBS-directed antibodies with heterosubtypic anti-influenza activity. In particular, two mAbs, S139/1 and C05 (Table 1), showed neutralization activity against strains from multiple IV subtypes both in vitro and in mice [46, 47].
-
2.
Decoy receptor or SA-containing inhibitors. This class of agents can be subdivided in: (1) polyvalent synthetic SA-containing inhibitors; and (2) natural inhibitors containing SA. Both these types of inhibitors act as receptor mimics and compete with sialylated receptors on the target cells for the binding to HA and thus block the virus attachment (Table 1). In addition, their binding to the virion surface can lead to virus particle aggregation, causing a reduction of virus infectivity. Regarding the first type of inhibitors, the design of polyvalent synthetic SA-containing compounds has been proposed to overcome the low affinity of HA binding to monovalent SA analogues, which are considered ineffective in competing with the highly multivalent interactions between the virus and the host cell [48]. Some of the polyvalent SA-containing inhibitors have also exhibited protective effects against IV infection in mice [49, 50]. As an alternative to multivalent sialosides, several groups have proposed the use of liposomes with SA analogues on the surface in order to allow a multivalent presentation of SA [51, 52]. A second type of inhibitors are constituted by SA-containing natural molecules, such as glycoproteins or proteoglycans. These molecules bind HA and hence create a steric obstacle to the polyvalent interaction of the virus with cells, thus blocking the process of virus adsorption to target cells. An example of these inhibitors is represented by the serum amyloid P component, which contains the α(2,6)-linked SA into the oligosaccharide side chains. This sialylated glycoprotein has been reported to limit IAV infection of airway epithelial cells and to also have therapeutic effects in mice [53].
-
3.
Peptides against HA. Some peptides exhibiting potent and broad-spectrum anti-influenza activity recently emerged as inhibitors of viral attachment [54, 55]. Similar to antibodies, these peptides specifically bind the HA protein and prevent IV adsorption to the host cell. These peptides were found to be effective not only in vitro but also in vivo, even when administered post-infection.
-
4.
Carbohydrate-binding agents that recognize specific glycosylation sites on HA. This class of inhibitors is exemplified by Cyanovirin-N (CV-N). This protein, which derives from the cyanobacterium Nostoc ellipsosporum, recognizes high-mannose oligosaccharide structures on HA (oligomannose-8 and -9) and its binding to HA prevents virus adsorption to the cell [56]. In fact, removal of these glycans from HA causes a decrease of viral sensitivity to CV-N [56]. Interestingly, CV-N showed antiviral activity not only against IAV and IBV but also against a broad range of enveloped viruses, such as HIV [57], Ebola virus [58], human herpesvirus 6 [57, 58], and hepatitis C virus (HCV) [59]. The same mechanism seems to be used by a lectin from green alga Boodlea coacta (BCA) [60]. Indeed, BCA exhibited a strong inhibition of HA activity by specifically interacting with 1–2-linked mannose at the nonreducing terminus of HA [60]. In addition, BCA showed activity against HIV [60]. The antiviral activity of these molecules is closely associated with the degree of glycosylation on the globular head of HA. Indeed, amino acid substitutions in the HA that cause the loss of glycosylation sites are related to a reduced sensitivity to lectin-based inhibitors [61].
Another possible strategy for inhibition of viral entry consists in preventing the fusion of the viral envelope with the endosomal membrane, in order to avoid the release of virion components into the cytoplasm (Fig. 1, top left). There is a heterogeneous group of inhibitors that act at this step of the viral life cycle, with distinct mechanisms of action:
-
1.
Small molecules that inhibit the low pH-induced conformational change of HA. After binding to the cellular receptor, IV is internalized into endosomes by clathrin-independent endocytosis [62]. The low pH inside the late endosomes triggers an irreversible conformational change of HA and enables the extrusion of the fusion peptide and its consequent insertion into the endosomal membrane [63]. Several small molecules that bind pockets in the stem region of the native form of HA have been identified as specific fusion inhibitors of IV [64–70]. Most of them prevent the fusogenic activity of the virus by blocking the low pH-induced conformational change of HA [64, 66–70]. The most recent example of compounds acting by this mechanism is RO5464466, which stabilizes the neutral pH conformation of HA in a pre-fusogenic state and prevents the fusogenic change of HA [70]. In contrast, other compounds, such as C22, inhibit membrane fusion by destabilizing the structure of HA, resulting in a premature and ineffective conformational change [65, 71]. However, these small molecules show a high propensity to select drug-resistant variants and exhibit limited protection against different HA subtypes. An exception is represented by arbidol (ARB; 1-metyl-2-phenyl-thiomethyl-3-carbotoxy-4-dimetylaminomethyl-5-hydroxy-6-bromoindolehydrochloridemonohydrate), which exhibits a broad spectrum of activity not only against IAV and IBV but also against other viruses such as RSV, parainfluenza virus, coxsackie virus, rhinovirus, hepatitis B virus (HBV), and HCV [71, 72]. This drug has been approved in Russia and in China for treatment and prophylaxis of IAV and IBV. Studies with viruses resistant to ARB-bearing mutations which map in the HA2 subunit confirmed that ARB interacts with HA and acts by stabilizing its structure, thus preventing the low pH-induced fusogenic change of HA [73]. Recently, biochemical studies showed that ARB interacts both with cell membrane phospholipids and with aromatic residues of viral glycoproteins on the surface of enveloped viruses [74]. This mechanism of action of ARB can prevent the fusogenic change in viral glycoproteins required for membrane fusion and could explain the broad spectrum of antiviral activity of this compound.
-
2.
Neutralizing mAbs directed against the stem region of HA. Another possible approach to interfere with the fusogenic activity of HA is to develop mAbs directed against its highly conserved stem region, mostly formed by HA2 monomers. Thanks to the high conservation of this region, such a strategy could allow to overcome the problem of the high variability associated to the HA globular head. Recently, a number of studies have reported antibodies that recognize conserved epitopes of the stem region of HA and show a broad activity against different subtypes and groups of IAV both in vitro and in animal models [75–81]. Among these, CR9114 is the only antibody capable of binding and neutralizing all group 1 and group 2 IAV tested, and also different IBV strains both in vitro and in mice [76]. Besides the fact that this antibody may represent a candidate therapeutic agent against IV, these findings suggest that the epitope recognized by CR9114 is highly conserved, not only among the different IAV subtypes but also among IBV. This observation heralds the prospect of a possible universal influenza vaccine against IAV and IBV based on this epitope, able to overcome the genetic variability of IV and to elicit a potent and cross-protective host immunity against all IAV and IBV strains [76].
Antiviral strategies targeting the RNA polymerase
The viral RNA-dependent RNA polymerase (RdRP) is a heterotrimer composed of subunits PB1, PB2, and PA, which carry out both mRNA transcription and replication of the viral genome (Fig. 1, left). During transcription, in a process known as “cap-snatching”, PB2 binds to the 5′-methyl cap of host pre-mRNA molecules, and PA, which has endonuclease activity, cleaves the pre-mRNA to produce a capped primer that is used to start transcription [82]. The PB1 protein possesses the RNA-dependent RNA polymerase activity and it is also responsible for the addition of a poly(A) tail to viral mRNA. PB1 also catalyzes the genome replication, which occurs via a positive sense cRNA intermediate that is an exact copy of the vRNA [82]. The three polymerase subunits interact with each other, in particular the N-terminus of PB1 interacts with the C-terminus of PA [83–85], while the C-terminus of PB1 binds the N-terminus of PB2 [83, 86]; in addition, a weak transient interaction has been proposed for PA and PB2 [87]. Thanks to its multidomain structure and multiple enzymatic activities, the RdRP can be targeted at different sites.
While a number of nucleoside/nucleotide drugs have been developed against other viral polymerases and are commonly used for treating infections caused by HIV, HBV, and herpesviruses, very few compounds have been reported which target the polymerization activity of IAV RdRP. This is in part due to the fact that the RdRP active site of PB1 has not yet been structurally characterized and even the precise boundaries of this domain are not known. The best-characterized anti-influenza nucleoside analogue is favipiravir (T-705; 6-fluoro-3-hydroxy-2-pyrazinecarboxamide; Table 1), which is a pyrazine derivative first identified in 2002 [88]. T-705 has been shown to inhibit influenza A, B, and C viruses in vitro and to be more effective than oseltamivir in protecting mice infected with IAV [88]. By cellular kinases, T-705 is converted to the active form, ribofuranosyl triphosphate, which acts as a nucleoside inhibitor of IV RdRP [89]. Importantly, fapiravir does not inhibit the synthesis of cellular RNA or DNA [90]. T-705 is active against a broad range of IAV and IBV strains, including 2009 pandemic strains, highly pathogenic avian H5N1 viruses, and the recently emerged H7N9 avian virus, and it also inhibits IV strains resistant to current antiviral drugs [90, 91]. Besides IV, T-705 inhibits a number of other RNA viruses, whereas it exhibits no inhibitory effect against DNA viruses. Remarkably, very limited resistance to favipiravir has been reported [90]. A Phase III clinical trial for evaluating favipiravir for influenza therapy began in Japan in October 2009 and has been completed, and two Phase II studies have been conducted in the United States since February 2010 and the results are being reviewed [91]. In the near future, we may therefore see the introduction of favipiravir in the clinical practice for the treatment of influenza.
Another possible strategy for selectively inhibiting IV replication is to target the endonuclease cap-snatching activity of the RdRP complex, which resides in the N-terminal region of PA [92, 93]. In past years, a number of inhibitors (e.g., flutimide and L-735882; Table 1) of PA endonuclease activity have been discovered by Roche, Merck, and other pharmaceutical companies using a structure–activity relationships (SAR) approach [94–96], but most of them have not been developed further, likely because none had sufficiently potent antiviral activity to justify clinical studies. More recently, crystallographic studies revealed that the endonuclease active site of PA contains a conserved deep cleft which could be an excellent target for structure-based design of novel anti-IV drugs [92, 93]. Along this line, two groups have reported co-crystal structures of the PA endonuclease domain with known or predicted inhibitors [97, 98], providing insights that could be useful for the structure-based design of new PA inhibitors. In addition, a fragment screening using a high-resolution crystal structure of the N-terminal endonuclease domain of pandemic 2009 H1N1 IV and structure-based optimization led to the identification of a hydroxypyridinone series of compounds exhibiting promising enzymatic inhibition; a compound from this series was also found to have a significant antiviral activity in cells [99]. Using a different approach, Iwai et al. [100] screened 33 different types of phytochemicals using a PA endonuclease inhibition assay in vitro and identified marchantins as PA inhibitors. In particular, marchantin E docked well into the endonuclease active site and inhibited the growth of both IAV and IBV.
The interactions between the PA and PB1 as well as the PB1 and PB2 subunits have been shown to be essential for polymerase function [101, 102]. In addition, the subunit binding interfaces are highly conserved between different viral strains [2]. Thus, inhibition of these interactions represents an attractive strategy for the development of drugs with broad efficacy against all IV strains [103, 104]. The feasibility of this approach was first proved by studies showing that short N-terminal PB1 peptides, corresponding to the PA-binding domain of PB1, were able to block the activity of IAV polymerase and also inhibit viral replication [105, 106]. Recently, two crystal structures of a truncated form of PA bound to a PB1-derived peptide have been published [107, 108]. Importantly, these structures showed that relatively few residues drive binding of PB1 to PA, suggesting the potential for small molecule-mediated inhibition. Along this line, an in silico screening of 3 million small-molecule structures using one of these crystal structures [107] led to the identification of two compounds (compounds 1 and 5; Table 1) able to interfere with the interaction between PB1 and PA both in vitro and in cells, as well as transcription by the RdRP [109]. One of these molecules (compound 1) also inhibited the replication of a panel of IAV strains, including 2009 pandemic strains and an oseltamivir-resistant isolate, as well as several IBV strains, with EC50 values in the low micromolar range [109]. In addition, the design and synthesis of analogues of another of the hits—compound 10—initially selected in the in silico screening aimed at searching for inhibitors of PA/PB1 interaction led recently to the identification of other compounds endowed with similar antiviral properties [110]. Interestingly, a compound called AL18, previously shown to inhibit subunit interactions of human cytomegalovirus DNA polymerase [111], was found to also block the PA/PB1 interaction as well as the replication of IAV and IBV [112]. In a similar, but more restricted, screening, Fukuoka and colleagues performed a docking simulation using a drug database of ~4,000 compounds and selected candidate compounds targeting the PA/PB1 interface [113]. Among these, benzbromarone, diclazuril, and trenbolone acetate (Table 1) exhibited anti-IAV activity. In addition, benzbromarone and diclazuril were shown to bind the PA subunit and to decrease the transcriptional activity of the viral RdRP. In a different approach, a total of 15,000 molecules were tested in an ELISA-based screening, which led to the identification of benzofurazan compounds that also showed inhibition of viral replication at micromolar concentrations [114]. However, these compounds exhibited significant cytotoxicity, thus likely excluding an in vivo use. Overall, the compounds targeting the PA/PB1 binding interface could provide the basis for the development of a new generation of therapeutic agents against IAV and IBV.
The possibility of targeting other interaction sites in the polymerase complex, e.g., those between the PB1 and PB2 subunits, recently emerged with the publication of the crystal structure of the PB1/PB2 binding interface [102]. The structure showed that only small regions of PB1 (residues 678–757) and of PB2 (residues 1–37) are required for tight binding. Since the PB1/PB2 interface has a crucial function in regulating the polymerase complex and is highly conserved among IV, it appears as a promising target for novel broad-spectrum anti-influenza drugs. As a proof-of-principle, a synthetic peptide corresponding to residues 1–37 of PB2 was shown to inhibit the PB1/PB2 interaction in vitro [115]. However, to date, no small molecule targeting this protein–protein interaction has yet been reported, and indeed the flat PB1/PB2 interface may pose a significant challenge to the development of a nonpeptide small-molecular-weight inhibitor. Recently, Li and colleagues reported that a peptide derived from amino acids 731–757 of PB1 can disrupt the interaction between the C-terminal part of PB1 (aa 676–757) and the N-terminal part of PB2 (aa 1–40), and also inhibit viral RdRP activity and IV replication [116]. Surprisingly, the authors showed that this peptide interacts with PB1 rather than PB2. Furthermore, mutational analyses and computational modeling suggested that the PB1731–757 peptide acts as a competitor of PB2 with respect to binding to PB1. Thus, the inhibitory mechanism of the PB1731–757 peptide is likely different from that of the interfacial peptides PB11–25 and PB21–37, which inhibit complex assembly by binding to its interaction partner PA or PB1, respectively, and could suggest new avenues for antiviral drug discovery.
Antiviral strategies targeting the nucleoprotein
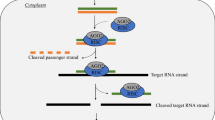
The nucleoprotein (NP) of influenza virus is one of the most abundant viral proteins in the infected cell. It plays a major role in the vRNP assembly by interacting with both RNA and viral polymerase subunits, but there is also evidence of its involvement in vRNP nuclear export and cytoplasmic transport (Fig. 1, left) [117, 118]. Recombinant NP also forms dimers and oligomers in the absence of RNA [119]; however, recent findings revealed that this process seems to be less efficient under these conditions [120]. During viral infection, the oligomerization of NP in ring structures and its interaction with single-stranded viral genomic RNA segments is essential for vRNP assembly. In the working model, NP trimers interact with RNA leaving the nucleotides exposed to the solvent [121]. As a result of the interaction of NP and the heterotrimeric RdRP with viral RNA, the latter is twisted into a double-helical structure with the polymerase complex at one end and an RNA loop at the other [121, 122]. The interaction between the two NP-RNA filaments of the double helix is guaranteed by NP–NP contacts. In recent years, the interactions of NP with itself [123–127], with viral RNA [128], with viral RdRP, and with cellular factors have been investigated for the development of new anti-IV strategies, demonstrating the significant potential of NP as an antiviral target. As an example, short interfering RNA designed for NP gene silencing and possessing a 5′-triphosphate moiety to induce a RIG-I-mediated interferon (IFN) response exhibited potent inhibitory effects both in infected cells and in mice, demonstrating that NP knockdown is a successful strategy to inhibit IAV propagation [129, 130]. Perhaps the most promising feature of NP from a pharmaceutical point of view is the ability of self-interaction mediated by a flexible tail loop present in each monomer that inserts in the neighboring monomer. This interaction is stabilized by electrostatic interactions between E339 and R416 residues [126]. In addition, the phosphorylation at Ser-165 seems to regulate the oligomerization status and RNA binding activity of NP in infected cells [131]. Small molecules able to interfere with correct protein–protein interactions between NP monomers or to stabilize NP in the monomeric form were independently identified by several groups and block IV replication at different steps (Fig. 1) [123, 125–127, 131]. Among these, nucleozin (NCZ) is the most studied. Since NP has two sites for self-interaction [132], it is widely accepted that such compounds bind at least two different sites on NP and act either by stabilizing monomeric NP or by inducing the formation of NP aggregates. The improper interactions of NP monomers occurring in the presence of these antiviral compounds interfere with the different functions of NP in the virus cycle. In fact, NCZ and its derivatives were reported to exert antiviral activity at different times of IAV cycle [133]: there is an early inhibitory effect of NCZ on viral RNA transcription and replication as well as a recently identified antiviral effect exerted on the cytoplasmic trafficking of newly synthesized vRNPs. NCZ is supposed to affect both the transport of vRNPs into the nucleus and, after nuclear export, the transport of newly synthesized vRNP through the cytoplasm that involves the cellular protein Rab11 (Fig. 1) [133]. Interestingly, naproxen, a clinically-approved inhibitor of inducible cyclooxygenase-2 (COX-2), was very recently identified through a structure-based in silico screening as the first inhibitor of the interaction between NP and RNA [128]. It was demonstrated that naproxen targets the RNA-binding groove of NP and blocks it in a monomeric form. Naproxen is effective against IAV H1N1 and H5N1 replication in infected cells and in mice. As prospected by the authors, the dual antiviral effect of this drug against IAV, i.e., inhibition of NP functions and of COX-2-induced cytokine storm, could be particularly useful for the treatment of infections with emerging highly pathogenic viruses characterized by an exacerbated inflammatory response.
Antiviral strategies targeting the nonstructural protein 1
NS1 is a multifunctional viral protein, whose major role is to antagonize the cellular antiviral response and in particular the IFN-mediated response (Fig. 3, bottom) [134, 135]. Actually, NS1 IFN-antagonistic properties seem to be strain-specific [135]. NS1 subverts the cellular IFN response by different strategies: (1) it cooperatively binds to viral dsRNA [136], thus protecting it from the recognition of two cytoplasmic “sentinels” of viral infection, i.e., protein-kinase RNA-activated [137] and 2′-5′-oligo(A) synthetase/RNaseL [138], which are activated by dsRNA and serve to shut off host protein synthesis (and also IFN pathway effectors) and to induce viral RNA degradation, respectively; (2) it binds to cellular mRNA processing factors, such as cellular polyadenylation specificity factor 30 (CPSF30) and polyA-binding protein II (PABP2) [139, 140], thus inhibiting 3′-mRNAs (such as IFN mRNAs) processing and nuclear/cytoplasmic trafficking; (3) it interacts with the ubiquitin-ligase TRIM25 and blocks RIG-I activation, thus preventing subsequent IFN cascade activation [140]; (4) it interacts with cellular IFN-inducible hGBP1 to antagonize its antiviral activity [141]; and, finally, (5) it targets IκB kinase (IKK) to block NF-kB activation [142]. NS1 post-translational modifications such as phosphorylation and SUMOylation may modulate its activity and also the abundance of NS1 dimers and trimers in infected cells [143]. In fact, NS1 dimerization is essential for RNA binding, since mutations that block dimers formation also affect the RNA binding ability of NS1 [144].
To date, no anti-influenza drug targeting this viral protein is under clinical development. However, some molecules were recently identified by a screen aimed at searching compounds that phenotypically suppress NS1 functions [145]. By this approach, 2,000 compounds were tested for their ability to suppress the slow-growth phenotype in yeasts expressing IAV NS1; four molecules able to restore normal growth in yeasts and also to inhibit IAV replication in cells were identified. These compounds and some derivatives showed anti-influenza activity only in IFN-competent cells, and the ability to reverse NS1-mediated block of IFN response [146]. A derivative, JJ3297, resulted to be dependent on cellular RNAseL functions for antiviral activity [147]. These compounds provide the proof-of-principle that NS1 activity can be blocked by small molecules that could also be used in combination with IFN agonists to enhance their antiviral activity. Another target of NS1-based antiviral strategies recently proposed is the interaction of NS1 with viral RNA. Indeed, compounds able to interfere with NS1/viral RNA binding have been identified both by in silico screening [148] and by high-throughput screening (HTS) developed to search for inhibitors of the binding of recombinant NS1 to a viral RNA construct in vitro [149]. By another approach, i.e., a fluorescence polarization-based assay, other inhibitors of the binding between IAV NS1 and dsRNA, a library of quinoxoline derivatives, and a large small molecule library were tested leading to the identification of a compound, epigallocatechine gallate, able to inhibit virus growth [150, 151]. The activity of such compounds suggests that the block of NS1 binding to viral RNA could also be a feasible anti-influenza strategy.
Antiviral strategies using drug combinations
Combination therapies that target multiple viral protein functions have been proposed to achieve greater antiviral effects than each compound given individually, to reduce the development of drug-resistance, and to allow administration of lower drug doses, thereby decreasing adverse effects. Analogous to the treatments used against HIV and HCV, a combination of anti-IV drugs would be expected to be more effective than single-agent chemotherapy in treating serious influenza cases. The combined use of amantadine + oseltamivir [152] and oseltamivir + T-705 [153] has shown therapeutic synergism in infected mice. In addition, association of oseltamivir and amantadine has been demonstrated to reduce the emergence of drug-resistant IAVs [154]. A preliminary, controlled clinical study comparing the therapy with rimantadine plus inhaled zanamivir versus rimantadine alone in hospitalized adult patients with serious influenza showed a higher efficacy for the combination of zanamivir with rimantadine [155]. Several other double-, triple-, even quadruple-drug combinations could be envisaged for achieving additive or synergistic antiviral effects, based not only on the currently available drugs but also on new compounds under development. In addition, besides combinations of therapeutic agents directed against viral targets, combinations with compounds directed against cellular targets have also been proposed (see below).
New host-based anti-influenza virus strategies
IV replication is strictly host-dependent. In fact, a plethora of cellular proteins play an important role in viral replication [156–160]. In recent years, several studies have reported different experimental approaches aimed at identifying the cellular proteins that are essential for IV replication and could be potential targets of new antiviral strategies. Among these, there are, for example, genome-wide RNA interference screenings (reviewed in [161]), proteomic approaches [162], and yeast two-hybrid screenings [159, 163]. Antiviral drugs targeting cellular functions are expected to have some advantages over inhibitors directed against viral targets, in particular for genetically variable viruses such as IAV. In fact, inhibitors of cellular proteins and/or pathways should be less prone to induce the selection of resistant viral strains; on the other hand, perturbing the cellular environment to disrupt viral functions could have adverse side effects that should be carefully considered. We will describe cellular proteins and/or pathways that represent attractive anti-influenza targets due to their active engagement in the viral cycle and some already identified small-molecular-weight inhibitors that are currently under preclinical or clinical investigation as anti-IV agents.
Antiviral strategies involving host factors that are engaged in virus attachment, entry, and release of virus particles
As already mentioned above, the first step in IV infection is the attachment of HA to SA-containing glycoprotein or glycolipid receptors on the host cell surface. An option for influenza therapy is the development of drugs targeting cellular components involved in this step rather than targeting the viral HA protein (Fig. 2, top left). The proteolytic activation of HA by cellular proteases is essential for IV propagation and may thus represent an attractive antiviral target. Human airway trypsin-like protease (HAT) and transmembrane protease serine S1 members (TMPRSS) belong to the type II transmembrane serine protease family, are found in human airway epithelium, and have been proposed as activators of HAs which have a monobasic cleavage site and are expressed by low pathogenic avian viruses and all human H1, H2, and H3 subtypes [164–166]. Processing of HAs with such a monobasic cleavage site restricts the infection to the respiratory airway epithelia in mammals and to the respiratory and intestinal epithelia in birds. In contrast, HAs expressed by highly pathogenic avian strains, such as H5 and H7 strains, contain multibasic cleavage sites that are recognized and cleaved by intracellular proteases, such as furin and proprotein convertase 5 and 6, and by specific transmembrane proteases such as TMPRSS13 [167, 168].
Antiviral strategies involving host factors engaged in IV replication. IV infection can be blocked by inhibitors of transmembrane proteases responsible of HA activation and by Fludase. After the nuclear import of vRNPs, the first step is the transcription of the viral genome into viral mRNA and this process is catalyzed by RdRP with the involvement of cellular proteins such as RNA polymerase II (RNAP-II) and transcription elongation factor pTEF1b. Inhibitors of both RNAP-II and pTEF1b complex are able to interfere with this process and to block viral mRNA synthesis. Viral mRNAs are then exported into the cytoplasm. The soluble lipid mediator PD1 is able to inhibit IV replication by interfering with the nuclear export of viral transcripts mediated by the cellular proteins NFX1 and Nup62. In the cytosol, viral mRNAs are translated into viral proteins required for the viral genome replication, which takes place into the nucleus. At this stage, Hsp90 inhibitors can be effective in blocking the nuclear import of viral proteins. Also, inhibitors of mitochondrial dihydroorotate dehydrogenase (DHODH), an enzyme of the pyrimidine biosynthetic pathway, possess anti-influenza activity. The newly synthesized vRNPs have then to be exported into the cytoplasm to be assembled together with the other viral proteins into the new virions. Antioxidants able to restore the cellular redox potential and intracellular protease inhibitors can interfere with the correct processing and maturation of HA. Finally, compounds that interfere with the proper formation of lipid rafts at the plasma membrane can block IV budding
Given the essential role of cellular proteases in the activation of HA, protease inhibitors have been investigated as potential anti-influenza agents [169]. As an example, aprotinin, which is a 58-amino-acid single-chain globular polypeptide of bovine origin, demonstrated anti-influenza activity in human adenoid epithelial cells [170], in a mouse model of influenza infection [171], and also in humans [172]. Aprotinin is used in clinical practice for the prevention of post-operative bleeding; however, very recently, the use of this protease inhibitor for the treatment of seasonal and swine-originated pandemic influenza has been approved in Russia [173]. In these cases, aerosolized aprotinin can be administered via inhalation at lower doses that those used in the surgical practice. Zhirnov et al. [173] demonstrated that, in the presence of aprotinin, IV prevalently contains uncleaved HA0 and the resulting viral progeny is less infectious. Other protease inhibitors demonstrated anti-influenza activity; for example, a recent screening with the catalytic domain of TMPRSS2 and known trypsin-like serine proteases inhibitors led to the identification of a sulfonylated 3-amindinophenylalanylamide derivative able to block IV propagation in cultured human adenocarcinoma-derived epithelial cells Calu-3 [174]. Finally, as mentioned above, the HAs of highly pathogenic avian IV strains, such as H5 and H7 subtypes [175], contain multibasic cleavage sites and are activated during the transit from ER to the plasma membrane by furin and other cellular proteases located in the trans-Golgi network (Fig. 2, bottom). Thus, the inhibition of furin or furin-like proprotein convertases might also represent an anti-IV strategy [176, 177].
Another possible approach to block virus–host interactions is the removal of SA from the cellular membrane. Sialidases, that catalyze the removal of terminal SA residues from glycoproteins and glycolipids (Fig. 2, top left), have been demonstrated to be effective inhibitors of IV infection [178, 179]. A new candidate drug currently under clinical development as an inhibitor of influenza and parainfluenza infections is DAS181 (also known as Fludase), a recombinant fusion protein that prevents IV attachment by enzymatically removing SA from the receptors of the human epithelia airway [178]. DAS181 is composed of the catalytic domain of Actinomyces viscosus sialidase fused with the respiratory epithelium-anchoring domain of human protein amphiregulin [180, 181]. The advantages of A. viscosus sialidase over other bacterial sialidases are broad substrate specificity, higher specific activity, and good tolerance by the human immune system [178]. Preclinical in vitro and in vivo studies demonstrated the inhibitory activity of DAS181 against various seasonal strains of IAV and IBV and prophylactic and therapeutic effects against H5N1 virus infection in mice [181, 182]. Importantly, this compound is also effective against strains resistant to the existing antiviral drugs, for example against oseltamivir-resistant H1N1 clinical isolates [181]. Moreover, a recent phase II clinical study of inhaled DAS181 showed a significant decrease of viral load in influenza-infected patients [183].
Budding and release of new virus particles occur at specific domains of the plasma membrane, called lipid rafts (Fig. 1, bottom right) [184]. Lipid rafts are dynamic microdomains of the cell membrane characterized by a higher percentage of cholesterol, sphingolipids, and phospholipids containing saturated fatty acids. Viral HA and NA are recruited at these specific domains, together with other cellular proteins responsible of vesicle formation, thus they are considered to be the sites of budding initiation [184]. It was reported that cholesterol depletion [185] or the expression of an IFN-inducible cellular protein, viperin [186], disrupt plasma membrane by perturbing the correct formation of lipid rafts. Viperin interacts with and inhibits farnesyl diphosphate synthase (FPPS), a cellular enzyme involved in the synthesis of various isoprenoid-derived precursors of cholesterol and other essential cellular components [186]. Furthermore, inhibition of FPPS, by chemical agents, siRNA, or viperin expression, also prevents IAV release by perturbing the correct formation of lipid rafts (Fig. 2, bottom right) [186]. However, pamidronate, a clinically-approved FPPS inhibitor, did not show efficacy in vivo in protecting mice from lethal influenza [187]. Thus, FPPS inhibitors may not be suitable anti-influenza agents despite their ability to prevent replication in vitro.
Antiviral strategies involving host factors that are engaged in viral RNA transcription/replication and processing and in vRNP trafficking
Viral RNA synthesis, which entails both primary mRNA transcription and vRNA replication processes, is directed by the virus-encoded RdRP, either transported into the nucleus directly after viral entry or de novo synthesized in the infected cell (Fig. 1, left). The initiation of primary mRNA transcription also depends on the activity of host RNA polymerase II (RNAP-II) and other accessory factors such as the positive transcription elongation factor 1b (pTEF1b), which consists of the cyclin-dependent kinase 9 (Cdk9) and Cyclin T1 complex (Fig. 2, left) [188]. Inhibitors of RNAP-II and Cdk9 that showed anti-influenza activity are listed in Table 2.
The nuclear translocation of vRNP components to promote new vRNP assembly and export also depends on a number of host factors such as importins and molecular chaperones that may represent antiviral targets. In particular, the Hsp90 protein plays a key role in the nuclear translocation of vRNP components PB1 and PB2, by forming a PB1–PB2–Hsp90 complex prior to the assembly of the RdRP complex [189]. Inhibitors of Hsp90, such as geldanamycin and 17-AAG, affect IV replication in cell culture, and the possible mechanism could be the block of the nuclear import of PB1 and PB2 and the induction of their degradation [190]. Other host factors involved in the nuclear export of vRNP represent targets of possible new anti-influenza strategies (Fig. 2, center). In Table 2 are listed a series of inhibitors of cellular proteins that also showed anti-IV activity by interfering with vRNP trafficking.
Antiviral strategies involving host signaling pathways
Many host cell signaling pathways are affected during IV infection. Among these, there are some tyrosine kinase receptors, the downstream mitogen-activated protein kinases (MAPKs) pathway, the phophatidylinositol-3-kinase/Akt/mTOR pathway, and the NF-κB pathway. The activation of host signaling pathways is essential for productive IV infection, since small molecules that inhibit any step of signal transduction are able to block virus propagation by affecting different events in IV life cycle as will be described below.
The MAPKs signaling pathways are activated in response to a variety of stimuli, including infection by IV, and detrimental effects on virus replication are observed if the activation is blocked [191]. The phosphorylating activity of MAPKs pathways effectors plays a major role in vRNP trafficking, particularly in NEP-mediated vRNP nuclear export, and in viral infectious particle production (Fig. 3, center) [192–194]. Of particular interest, MAPKs inhibition can be obtained both pharmacologically (by using inhibitors) and by restoring the physiological intracellular reduced state in the infected cells after the oxidative stress caused by IV infection [195]. In fact, MAPKs pathway, like other cellular signaling pathways activated by IV, such as phosphoinositide 3-kinase (PI3K) and NF-κB-mediated signaling, is strictly dependent on host cell redox state [196–198]. A list of receptor tyrosine kinases and MAPKs signaling inhibitors that block influenza virus replication can be found in Table 2. Along this line, molecules that block IV replication and the pro-inflammatory cascade are represented, for example, by kinase inhibitors, such as the tyrosine kinase receptor inhibitors AG879 and A9 [199], the MEK-specific inhibitor UO126 [191, 200, 201], and antioxidant compounds, such as p38 inhibitor SB203580 and N-acetylcysteine [202, 203], glycyrrizin [204], glutathione, and its derivatives [205, 206], and curcumin and resveratol derivatives [207]. All these compounds are potential new host-based anti-influenza agents that could be used in combination with “old” anti-influenza drugs such as oseltamivir, as prospected in [208].
Antiviral strategies involving host signaling pathways and antiviral defense mechanisms. IV infection triggers the direct activation of a series of cellular signaling pathways such as some tyrosine kinase receptors (TKR) pathways, pro-inflammatory pathways, and the interferon (IFN)-activated antiviral response. Also the oxidative stress that follows viral infection activates several redox-sensitive cellular pathways, such as NF-κB and MAPKs pathways. Furthermore, COX-2 inhibitors have been shown to inhibit both IV replication and viral-induced inflammation and cytokine production. Antioxidants that interfere with redox-sensitive cellular processes also inhibit viral functions such as vRNPs trafficking and nuclear export, as well as HA maturation, thus blocking viral replication. The antiviral response activated by IV is dependent on IFN cascade activation and initiates with the recognition of viral patterns by the Toll-like receptors TLR-3, -7, and -9 on the cell membrane and by cytoplasmic RIG-I-like receptors (RLRs), which act as sensors of viral invasion. TLRs and RLRs agonists as well as CpG oligonucleotides (ODNs) exhibit anti-influenza activity by stimulating the IFN response. The viral NS1 protein antagonizes this antiviral response, thus antiviral strategies either targeted against this protein or involving the activation of IFN pathway have inhibitory effects on IV replication
The redox state of the infected cell also influences other two important processes in the IV life cycle, i.e., HA maturation and NF-kB activation. HA is a disulfide-rich protein and is initially found as a glycosylated monomer in the endoplasmic reticulum (ER). Then, it undergoes trimerization, oxidation catalyzed by the cellular protein disulfide isomerase that needs an oxidative environment, proteolytic cleavage, and is finally inserted into the plasma membrane. Thus, compounds that restore the cellular redox state, such as glutathione, are also able to exert anti-IV activity by two distinct mechanisms: (1) by interference with the activation of redox-sensitive signaling pathways exploited by the virus, such as MAPKs and PI3K pathways (Fig. 3), and (2) by blocking the maturation of viral HA protein (Fig. 1, bottom right) [194, 195]. Also, thiazolides such as nitazoxanide are potential broad-spectrum antivirals that block HA terminal glycosylation and impair the trafficking of HA precursors from ER to Golgi [209]. The exact mechanism of action of these compounds is still to be elucidated, although a cell-mediated effect of thiazolides has been postulated due to the observed inhibition of different viruses by this class of molecules.
The redox-sensitive signaling pathway of PI3K/Akt/mTOR is also activated by IV infection. Its activation transduces the transcriptional signal through a phosphorylation cascade of other downstream kinases such as Akt/PKB, cAMP-dependent kinase (PKA), and ribosomal S6 kinases and mTOR. The role of PI3K pathway activation during IV infection is still controversial; however, compounds targeting PI3K or downstream effectors, such as PI3K inhibitors wortmannin [210] and LY294002 [211] or mTOR inhibitors [212], block IAV replication in vitro. For this reason, considering the increasing availability of PI3K/Akt/mTOR inhibitors already approved or under clinical investigation as anticancer drugs [213], the inhibition of this host cell pathway as a new anti-influenza approach clearly deserves further investigation.
Another pathway targeted by IV infection is that of NF-κB, a family of transcription factors that play a role in induction of inflammation, activation of immune response, proliferation, and apoptosis (Fig. 3, left) [214]. The activation of NF-κB pathway is critical for IV productive replication [215–217] and is the result of the ability of viral HA, NP, and M1 proteins to produce oxidative radicals. The oxidative stress activates redox-sensitive signaling pathways and the NF-κB inhibitor IKK, which is responsible of its derepression and transcriptional activation [218]. The essential nature of active NF-κB for IV replication makes it a promising target of antiviral strategies. On this line, a number of inhibitors of NF-κB activation, such as acetylsalicylic acid [215] and pyrrolidine dithiocarbamate [219], as well as antioxidant agents (see above and [220]), have been reported to block IV replication and propagation. Of particular interest is the NF-κB inhibitor SC75741 [221]. This compound is an efficient blocker of NF-κB module at non-toxic concentrations and also an inhibitor of IAV and IBV replication in MDCK and A549 cells [221, 222]. The effect of SC75741 on virus replication is to prevent efficient nuclear export of vRNP as a result of the lack of NF-κB transcriptional activity, i.e., via inhibition of virus-induced pro-apoptotic genes expression essential for vRNP export. Importantly, no resistant viruses were selected even after 8 passages in the presence of SC75741, thus this inhibitor showed a very low potential in selecting viral resistant variants [221]. Furthermore, SC75741 demonstrated its efficacy in reducing viral replication and cytokine expression in mice infected with highly pathogenic avian IV strains H5N1 and H7N7 [208]. This inhibitor did not show long-term inhibitory effects on cell proliferation [221] and, although more pre-clinical studies are needed, it might represent another candidate drug that targets cellular functions to block IV replication.
The cellular COX pathway is also engaged during IV infection (Fig. 3, left), in particular in the case of human infection with highly pathogenic avian strains such as H5N1 [223]. In fact, the H5N1 strain strongly upregulates COX-2 during the infection of human macrophages, and COX-2-mediated pro-inflammatory signaling could be responsible of the hypercytokinemia that distinguishes the pathogenic mechanism of H5N1 strain in humans. Recently, it was demonstrated that the non-steroidal COX-2 inhibitor nimesulide is able to inhibit H5N1 strain replication in human macrophages; thus, COX-2 could also represent a promising cellular target for new anti-IV strategies [224].
In conclusion, there is increasing evidence that understanding the molecular mechanisms underlying how IV modulates the host cell signaling pathways could be the key that opens the door to innovative therapeutic antiviral approaches.
Antiviral strategies involving host cell metabolism
IV uses cellular constituents to produce its own viral RNAs, proteins, and lipid envelope. However, the virus needs NTPs, amino acids, and membrane components from the host cell to obtain new building blocks to be recycled in the synthesis of viral components. The degradation of cellular nucleic acids is not the only strategy whereby IV enriches the NTPs pool for viral RNA synthesis [225]; in fact, de novo synthesis of pyrimidine and uracil salvage pathways are also engaged during IV infection, depending on the cell type [226]. Pyrimidines are important precursors used for RNA (uracil and cytosine), glycoproteins, and phospholipids biosynthesis. On this line, compounds able to deplete the cellular pyrimidine pool show anti-influenza activity. An example is the small molecule A3, identified by HTS, which possesses broad-spectrum antiviral activity by targeting pyrimidine metabolism and in particular the mitochondrial enzyme dihydroorotate dehydrogenase (DHODH; Fig. 2, right) [226]. DHODH has also emerged as a target of another class of inhibitors, the quinolone carboxylic acid derivatives, which were reported to inhibit IAV replication in infected cells [227]. Furthermore, by studying their effects on DHODH inhibition, it was demonstrated that DHODH block leads to the up-regulation of cellular antiviral factors, such as the NXF1 protein, which are able to reverse the cellular mRNA export block mediated by viral NS1 protein [227]. However, de novo pyrimidine synthesis requirement for efficient viral replication may be cell- and species-specific, since it was reported that the nonnucleoside DHODH inhibitor D282 showed anti-IAV and IBV activity in vitro but not in infected mice [228].
Lipid mediators and lipid metabolic pathways became hot topics in the last year because of the discovery of their role in the modulation of the inflammatory response upon infection by IAV [229, 230]. In particular, the lipid soluble mediator protectin D1 (PD1) is a docosahexaenoic acid-derived protectin D1 isomer product of the 12/15-lipoxigenase that is reduced in lungs of infected mice during infection with IAV [231]. Previously, PD1 was reported to have anti-inflammatory and pro-resolving activity [232, 233]. Morita et al. found that PD1 has potent and specific anti-IV activity in cells and in a mouse model of severe influenza infection with low and highly pathogenic viruses [231]. Mechanistically, PD1 selectively blocks the nuclear export of viral RNA molecules by inhibiting their recruitment to cellular NFX1 protein that binds Nup62 (Fig. 2, center). Importantly, PD1 did not show evident detrimental effects on the transport of cellular mRNAs. Other features make PD1 one of the most promising drug candidate to treat severe influenza infection, such as (1) the activity when also added 48 h post-infection, the time at which the current anti-IV drugs have demonstrated limited efficacy, thus highlighting its potential as both prophylactic and therapeutic agent; (2) the synergic activity with peramivir against IV infection in mice; and (3) the observation that the exogenous administration of PD1 is feasible and attenuates the lung response to viral injuries in infected mice. Antiviral molecules like PD1 and also the aforementioned COX inhibitors, nimesulide and naproxen (clinically approved drugs), belong to an emerging new class of anti-IV agents that couple specific antiviral activity with the ability to block the pro-inflammatory response activated upon infection and responsible for the immunopathogenesis of IV. For all these reasons, the further clinical development of PD1, given its potency and late-stage efficacy, should be greatly encouraged.
Antiviral strategies involving host cell antiviral response
Infection by ssRNA viruses, including IV, is known to be recognized by certain cellular pattern recognition receptors (PRRs), such as transmembrane Toll-like receptors (TLR) 3, 7, and 9, and cytoplasmic RIG-I like receptors (RLRs), which sense the invasion of a pathogen and counteract it both by stimulating an intracellular antiviral state and by sensitizing neighboring cells (Fig. 3, bottom). One of the most important cellular defense strategies against IV infection is the activation of type I and III IFN pathways [234]. Type I IFN expression is the result of the PRRs activation and downstream signaling and leads in turn to the activation of more than 300 IFN-stimulated genes, whose products cooperate to inhibit IV replication at different stages, to trigger the adaptive immune response, and ultimately to induce apoptosis of the infected cell. IVs have evolved various strategies to evade IFN response (reviewed in [234]) with NS1 representing the major player (see above).
Given the pivotal role of IFN pathway in the early anti-IV cellular response, its activation or enhancement represents an important option for antiviral intervention. Compounds that act as agonists of TLRs and RLRs are able to induce IFN and other pro-inflammatory cytokines and chemokines, as well as to function as vaccine adjuvants (Fig. 3, bottom). Indeed, treatment with poly(I·C), CpG oligodeoxynucleotides, and other TLRs ligands proved to be effective in protecting aged mice against lethal IAV infection. Recently, a cell-based HTS aimed at identifying small molecules able to induce IFN was reported [235]. The lead compound 3 identified by this HTS is able to induce an IFN-dependent antiviral state that cannot be counteracted by IAV NS1 protein (Fig. 3, bottom) [235]. Furthermore, a novel small molecule, ASN2, which inhibits viral RdRP subunit PB1, was reported to have additional IFN-inducing properties (Fig. 3) [236]. Short synthetic RNA molecules with an exposed 5′-triphosphate (5′ppp) moiety are substrate for RIG-I and have been reported to be able to induce a potent antiviral response that results in inhibition of IAV replication [129, 237, 238]. In particular, a synthetic 5′ppp-RNA was recently shown not only to inhibit IV replication in A549 cells but also, when intravenously administered, to trigger an antiviral state in infected mice that protected from lethal IV infection [238].
In conclusion, the induction of cellular antiviral responses by small molecules or polymers is another promising strategy for the development of new anti-influenza drugs that could be used in combination with other already available drugs to enhance their efficacy and to counteract synergistically the viral infection.
Concluding remarks
In the last few years, the need to overcome resistance and counteract threats of unforeseeable outbreaks of pandemic viruses has greatly fueled the search for new anti-influenza drugs. Recent research efforts mainly include (1) the improvement of existing drugs directed against M2 and NA; (2) the development of inhibitors against other antiviral targets, in particular RdRP, NP, and NS1; (3) strategies to block virus–cell interactions occurring at different stages of IV replication, such as attachment, entry, viral genome transcription and replication, nuclear export of viral products, and viral particles release; and (4) modulation of cell metabolism and host antiviral response. In particular, in the last few years, the targeting of cellular factors involved in IV replication has received much attention because such an antiviral approach could counteract viral drug resistance, as resistance against host-targeted antivirals would likely not emerge as rapidly as it has for virus-targeted inhibitors. In addition, as for other fast mutating viruses such as HIV and HCV, the application of antiviral drugs, in combination with different mechanisms of action, is being actively pursued, as it could be more effective in treating virulent and pandemic IV strains. Indeed, the possibility of blocking different viral and/or cellular targets may allow the generation of an “antiviral cocktail” that could reduce the probability of generating escape mutants.
Many of the inhibitors described here are still very far from being used in human therapy. Although some of them possess drug-like properties and promising efficacy/toxicity profiles, many preclinical and clinical studies usually undertaken in the drug discovery process [244] will be necessary to determine whether any of these compounds can have the potential to be developed into useful therapeutic strategies. Nevertheless, given the broad array of different anti-IV strategies under development, it is our hope that the discovery of new drugs will very soon provide wider options for improved prophylactic and therapeutic approaches against influenza infection.
References
Cox NJ, Subbarao K (2000) Global epidemiology of influenza: past and present. Annu Rev Med 51:407–421
Shaw ML, Palese P (2013) Orthomyxoviruses. In: Knipe DM, Howley PM (eds) Fields virology, 6th edn. Lippincott Williams & Wilkins, Philadelphia, pp 1648–1698
Chen W, Calvo PA, Malide D, Gibbs J, Schubert U, Bacik I, Basta S, O’Neill R, Schickli J, Palese P, Henklein P, Bennink JR, Yewdell JW (2001) A novel influenza A virus mitochondrial protein that induces cell death. Nat Med 7(12):1306–1312
Jagger BW, Wise HM, Kash JC, Walters KA, Wills NM, Xiao YL, Dunfee RL, Schwartzman LM, Ozinsky A, Bell GL, Dalton RM, Lo A, Efstathiou S, Atkins JF, Firth AE, Taubenberger JK, Digard P (2012) An overlapping protein-coding region in influenza A virus segment 3 modulates the host response. Science 337(6091):199–204
Wise HM, Foeglein A, Sun J, Dalton RM, Patel S, Howard W, Anderson EC, Barclay WS, Digard P (2009) A complicated message: identification of a novel PB1-related protein translated from influenza A virus segment 2 mRNA. J Virol 83(16):8021–8031
Rumschlag-Booms E, Rong L (2013) Influenza a virus entry: implications in virulence and future therapeutics. Adv Virol 2013:121924
Das K, Aramini JM, Ma LC, Krug RM, Arnold E (2010) Structures of influenza A proteins and insights into antiviral drug targets. Nat Struct Mol Biol 17(5):530–538
Pinto LH, Lamb RA (2006) The M2 proton channels of influenza A and B viruses. J Biol Chem 281(14):8997–9000
Ito T, Gorman OT, Kawaoka Y, Bean WJ, Webster RG (1991) Evolutionary analysis of the influenza A virus M gene with comparison of the M1 and M2 proteins. J Virol 65(10):5491–5498
Leonov H, Astrahan P, Krugliak M, Arkin IT (2011) How do aminoadamantanes block the influenza M2 channel, and how does resistance develop? J Am Chem Soc 133(25):9903–9911
Wang C, Takeuchi K, Pinto LH, Lamb RA (1993) Ion channel activity of influenza A virus M2 protein: characterization of the amantadine block. J Virol 67(9):5585–5594
Moss RB, Davey RT, Steigbigel RT, Fang F (2010) Targeting pandemic influenza: a primer on influenza antivirals and drug resistance. J Antimicrob Chemother 65(6):1086–1093
Bright RA, Medina MJ, Xu X, Perez-Oronoz G, Wallis TR, Davis XM, Povinelli L, Cox NJ, Klimov AI (2005) Incidence of adamantane resistance among influenza A (H3N2) viruses isolated worldwide from 1994 to 2005: a cause for concern. Lancet 366(9492):1175–1181
Krumbholz A, Schmidtke M, Bergmann S, Motzke S, Bauer K, Stech J, Durrwald R, Wutzler P, Zell R (2009) High prevalence of amantadine resistance among circulating European porcine influenza A viruses. J Gen Virol 90(Pt 4):900–908
Wang J, Wu Y, Ma C, Fiorin G, Pinto LH, Lamb RA, Klein ML, Degrado WF (2013) Structure and inhibition of the drug-resistant S31N mutant of the M2 ion channel of influenza A virus. Proc Natl Acad Sci USA 110(4):1315–1320
Wang J, Ma C, Jo H, Canturk B, Fiorin G, Pinto LH, Lamb RA, Klein ML, DeGrado WF (2013) Discovery of novel dual inhibitors of the wild-type and the most prevalent drug-resistant mutant, S31N, of the M2 proton channel from influenza A virus. J Med Chem 56(7):2804–2812
Furuse Y, Suzuki A, Oshitani H (2009) Large-scale sequence analysis of M gene of influenza A viruses from different species: mechanisms for emergence and spread of amantadine resistance. Antimicrob Agents Chemother 53(10):4457–4463
Wang J, Ma C, Fiorin G, Carnevale V, Wang T, Hu F, Lamb RA, Pinto LH, Hong M, Klein ML, DeGrado WF (2011) Molecular dynamics simulation directed rational design of inhibitors targeting drug-resistant mutants of influenza A virus M2. J Am Chem Soc 133(32):12834–12841
Balannik V, Wang J, Ohigashi Y, Jing X, Magavern E, Lamb RA, Degrado WF, Pinto LH (2009) Design and pharmacological characterization of inhibitors of amantadine-resistant mutants of the M2 ion channel of influenza A virus. Biochemistry 48(50):11872–11882
Wang J, Ma C, Wu Y, Lamb RA, Pinto LH, DeGrado WF (2011) Exploring organosilane amines as potent inhibitors and structural probes of influenza a virus M2 proton channel. J Am Chem Soc 133(35):13844–13847
Wei G, Meng W, Guo H, Pan W, Liu J, Peng T, Chen L, Chen CY (2011) Potent neutralization of influenza A virus by a single-domain antibody blocking M2 ion channel protein. PLoS ONE 6(12):e28309
Gamblin SJ, Skehel JJ (2010) Influenza hemagglutinin and neuraminidase membrane glycoproteins. J Biol Chem 285(37):28403–28409
Matrosovich MN, Matrosovich TY, Gray T, Roberts NA, Klenk HD (2004) Human and avian influenza viruses target different cell types in cultures of human airway epithelium. Proc Natl Acad Sci USA 101(13):4620–4624
Russell RJ, Haire LF, Stevens DJ, Collins PJ, Lin YP, Blackburn GM, Hay AJ, Gamblin SJ, Skehel JJ (2006) The structure of H5N1 avian influenza neuraminidase suggests new opportunities for drug design. Nature 443(7107):45–49
Gubareva LV, Kaiser L, Hayden FG (2000) Influenza virus neuraminidase inhibitors. Lancet 355(9206):827–835
von Itzstein M, Wu WY, Kok GB, Pegg MS, Dyason JC, Jin B, Van Phan T, Smythe ML, White HF, Oliver SW et al (1993) Rational design of potent sialidase-based inhibitors of influenza virus replication. Nature 363(6428):418–423
Cass LM, Efthymiopoulos C, Bye A (1999) Pharmacokinetics of zanamivir after intravenous, oral, inhaled or intranasal administration to healthy volunteers. Clin Pharmacokinet 36(Suppl 1):1–11
Tsang KW, Eng P, Liam CK, Shim YS, Lam WK (2005) H5N1 influenza pandemic: contingency plans. Lancet 366(9485):533–534
Davies BE (2010) Pharmacokinetics of oseltamivir: an oral antiviral for the treatment and prophylaxis of influenza in diverse populations. J Antimicrob Chemother 65(Suppl 2):ii5–ii10
Kohno S, Kida H, Mizuguchi M, Hirotsu N, Ishida T, Kadota J, Shimada J (2011) Intravenous peramivir for treatment of influenza A and B virus infection in high-risk patients. Antimicrob Agents Chemother 55(6):2803–2812
Barroso L, Treanor J, Gubareva L, Hayden FG (2005) Efficacy and tolerability of the oral neuraminidase inhibitor peramivir in experimental human influenza: randomized, controlled trials for prophylaxis and treatment. Antivir Ther 10(8):901–910
Koyama K, Takahashi M, Oitate M, Nakai N, Takakusa H, Miura S, Okazaki O (2009) CS-8958, a prodrug of the novel neuraminidase inhibitor R-125489, demonstrates a favorable long-retention profile in the mouse respiratory tract. Antimicrob Agents Chemother 53(11):4845–4851
Samson M, Pizzorno A, Abed Y, Boivin G (2013) Influenza virus resistance to neuraminidase inhibitors. Antiviral Res 98(2):174–185
Pizzorno A, Bouhy X, Abed Y, Boivin G (2011) Generation and characterization of recombinant pandemic influenza A(H1N1) viruses resistant to neuraminidase inhibitors. J Infect Dis 203(1):25–31
Feng E, Ye D, Li J, Zhang D, Wang J, Zhao F, Hilgenfeld R, Zheng M, Jiang H, Liu H (2012) Recent advances in neuraminidase inhibitor development as anti-influenza drugs. ChemMedChem 7(9):1527–1536
Mitrasinovic PM (2010) Advances in the structure-based design of the influenza A neuraminidase inhibitors. Curr Drug Targets 11(3):315–326
Lee CM, Weight AK, Haldar J, Wang L, Klibanov AM, Chen J (2012) Polymer-attached zanamivir inhibits synergistically both early and late stages of influenza virus infection. Proc Natl Acad Sci USA 109(50):20385–20390
Weight AK, Haldar J, Alvarez de Cienfuegos L, Gubareva LV, Tumpey TM, Chen J, Klibanov AM (2011) Attaching zanamivir to a polymer markedly enhances its activity against drug-resistant strains of influenza a virus. J Pharm Sci 100(3):831–835
Honda T, Yoshida S, Arai M, Masuda T, Yamashita M (2002) Synthesis and anti-influenza evaluation of polyvalent sialidase inhibitors bearing 4-guanidino-Neu5Ac2en derivatives. Bioorg Med Chem Lett 12(15):1929–1932
Macdonald SJ, Cameron R, Demaine DA, Fenton RJ, Foster G, Gower D, Hamblin JN, Hamilton S, Hart GJ, Hill AP, Inglis GG, Jin B, Jones HT, McConnell DB, McKimm-Breschkin J, Mills G, Nguyen V, Owens IJ, Parry N, Shanahan SE, Smith D, Watson KG, Wu WY, Tucker SP (2005) Dimeric zanamivir conjugates with various linking groups are potent, long-lasting inhibitors of influenza neuraminidase including H5N1 avian influenza. J Med Chem 48(8):2964–2971
Este JA, Telenti A (2007) HIV entry inhibitors. Lancet 370(9581):81–88
Hu J, Robinson JL (2010) Treatment of respiratory syncytial virus with palivizumab: a systematic review. World J Pediatr 6(4):296–300
Matthews T, Salgo M, Greenberg M, Chung J, DeMasi R, Bolognesi D (2004) Enfuvirtide: the first therapy to inhibit the entry of HIV-1 into host CD4 lymphocytes. Nat Rev Drug Discov 3(3):215–225
Fleury D, Barrere B, Bizebard T, Daniels RS, Skehel JJ, Knossow M (1999) A complex of influenza hemagglutinin with a neutralizing antibody that binds outside the virus receptor binding site. Nat Struct Biol 6(6):530–534
Whittle JR, Zhang R, Khurana S, King LR, Manischewitz J, Golding H, Dormitzer PR, Haynes BF, Walter EB, Moody MA, Kepler TB, Liao HX, Harrison SC (2011) Broadly neutralizing human antibody that recognizes the receptor-binding pocket of influenza virus hemagglutinin. Proc Natl Acad Sci USA 108(34):14216–14221
Ekiert DC, Kashyap AK, Steel J, Rubrum A, Bhabha G, Khayat R, Lee JH, Dillon MA, O’Neil RE, Faynboym AM, Horowitz M, Horowitz L, Ward AB, Palese P, Webby R, Lerner RA, Bhatt RR, Wilson IA (2012) Cross-neutralization of influenza A viruses mediated by a single antibody loop. Nature 489(7417):526–532
Yoshida R, Igarashi M, Ozaki H, Kishida N, Tomabechi D, Kida H, Ito K, Takada A (2009) Cross-protective potential of a novel monoclonal antibody directed against antigenic site B of the hemagglutinin of influenza A viruses. PLoS Pathog 5(3):e1000350
Sun XL (2007) Recent anti-influenza strategies in multivalent sialyloligosaccharides and sialylmimetics approaches. Curr Med Chem 14(21):2304–2313
Gambaryan AS, Tuzikov AB, Chinarev AA, Juneja LR, Bovin NV, Matrosovich MN (2002) Polymeric inhibitor of influenza virus attachment protects mice from experimental influenza infection. Antiviral Res 55(1):201–205
Gambaryan AS, Boravleva EY, Matrosovich TY, Matrosovich MN, Klenk HD, Moiseeva EV, Tuzikov AB, Chinarev AA, Pazynina GV, Bovin NV (2005) Polymer-bound 6′sialyl-N-acetyllactosamine protects mice infected by influenza virus. Antiviral Res 68(3):116–123
Hendricks GL, Weirich KL, Viswanathan K, Li J, Shriver ZH, Ashour J, Ploegh HL, Kurt-Jones EA, Fygenson DK, Finberg RW, Comolli JC, Wang JP (2013) Sialylneolacto-N-tetraose c (LSTc)-bearing liposomal decoys capture influenza A virus. J Biol Chem 288(12):8061–8073
Spevak W, Nagy JO, Charych DH, Schaefer ME, Gilbert JH, Bednarski MD (1993) Polymerized liposomes containing C-glycosides of sialic acid: potent inhibitors of influenza virus in vitro infectivity. J Am Chem Soc 115(3):1146–1147
Job ER, Bottazzi B, Gilbertson B, Edenborough KM, Brown LE, Mantovani A, Brooks AG, Reading PC (2013) Serum amyloid P is a sialylated glycoprotein inhibitor of influenza A viruses. PLoS ONE 8(3):e59623
Jones JC, Turpin EA, Bultmann H, Brandt CR, Schultz-Cherry S (2006) Inhibition of influenza virus infection by a novel antiviral peptide that targets viral attachment to cells. J Virol 80(24):11960–11967
Nicol MQ, Ligertwood Y, Bacon MN, Dutia BM, Nash AA (2012) A novel family of peptides with potent activity against influenza A viruses. J Gen Virol 93(Pt 5):980–986
Smee DF, Bailey KW, Wong MH, O’Keefe BR, Gustafson KR, Mishin VP, Gubareva LV (2008) Treatment of influenza A (H1N1) virus infections in mice and ferrets with cyanovirin-N. Antiviral Res 80(3):266–271
Dey B, Lerner DL, Lusso P, Boyd MR, Elder JH, Berger EA (2000) Multiple antiviral activities of cyanovirin-N: blocking of human immunodeficiency virus type 1 gp120 interaction with CD4 and coreceptor and inhibition of diverse enveloped viruses. J Virol 74(10):4562–4569
Barrientos LG, O’Keefe BR, Bray M, Sanchez A, Gronenborn AM, Boyd MR (2003) Cyanovirin-N binds to the viral surface glycoprotein, GP1,2 and inhibits infectivity of Ebola virus. Antiviral Res 58(1):47–56
Helle F, Wychowski C, Vu-Dac N, Gustafson KR, Voisset C, Dubuisson J (2006) Cyanovirin-N inhibits hepatitis C virus entry by binding to envelope protein glycans. J Biol Chem 281(35):25177–25183
Sato Y, Hirayama M, Morimoto K, Yamamoto N, Okuyama S, Hori K (2011) High mannose-binding lectin with preference for the cluster of alpha1-2-mannose from the green alga Boodlea coacta is a potent entry inhibitor of HIV-1 and influenza viruses. J Biol Chem 286(22):19446–19458
Anders EM, Hartley CA, Jackson DC (1990) Bovine and mouse serum beta inhibitors of influenza A viruses are mannose-binding lectins. Proc Natl Acad Sci USA 87(12):4485–4489
Lakadamyali M, Rust MJ, Zhuang X (2004) Endocytosis of influenza viruses. Microbes Infect 6(10):929–936
Harrison SC (2008) Viral membrane fusion. Nat Struct Mol Biol 15(7):690–698
Bodian DL, Yamasaki RB, Buswell RL, Stearns JF, White JM, Kuntz ID (1993) Inhibition of the fusion-inducing conformational change of influenza hemagglutinin by benzoquinones and hydroquinones. Biochemistry 32(12):2967–2978
Hoffman LR, Kuntz ID, White JM (1997) Structure-based identification of an inducer of the low-pH conformational change in the influenza virus hemagglutinin: irreversible inhibition of infectivity. J Virol 71(11):8808–8820
Luo G, Torri A, Harte WE, Danetz S, Cianci C, Tiley L, Day S, Mullaney D, Yu KL, Ouellet C, Dextraze P, Meanwell N, Colonno R, Krystal M (1997) Molecular mechanism underlying the action of a novel fusion inhibitor of influenza A virus. J Virol 71(5):4062–4070
Staschke KA, Hatch SD, Tang JC, Hornback WJ, Munroe JE, Colacino JM, Muesing MA (1998) Inhibition of influenza virus hemagglutinin-mediated membrane fusion by a compound related to podocarpic acid. Virology 248(2):264–274
Yoshimoto J, Kakui M, Iwasaki H, Fujiwara T, Sugimoto H, Hattori N (1999) Identification of a novel HA conformational change inhibitor of human influenza virus. Arch Virol 144(5):865–878
Yu KL, Torri AF, Luo G, Cianci C, Grant-Young K, Danetz S, Tiley L, Krystal M, Meanwell NA (2002) Structure-activity relationships for a series of thiobenzamide influenza fusion inhibitors derived from 1,3,3-trimethyl-5-hydroxy-cyclohexylmethylamine. Bioorg Med Chem Lett 12(23):3379–3382
Zhu L, Li Y, Li S, Li H, Qiu Z, Lee C, Lu H, Lin X, Zhao R, Chen L, Wu JZ, Tang G, Yang W (2011) Inhibition of influenza A virus (H1N1) fusion by benzenesulfonamide derivatives targeting viral hemagglutinin. PLoS ONE 6(12):e29120
Boriskin YS, Leneva IA, Pecheur EI, Polyak SJ (2008) Arbidol: a broad-spectrum antiviral compound that blocks viral fusion. Curr Med Chem 15(10):997–1005
Pecheur EI, Lavillette D, Alcaras F, Molle J, Boriskin YS, Roberts M, Cosset FL, Polyak SJ (2007) Biochemical mechanism of hepatitis C virus inhibition by the broad-spectrum antiviral arbidol. Biochemistry 46(20):6050–6059
Leneva IA, Russell RJ, Boriskin YS, Hay AJ (2009) Characteristics of arbidol-resistant mutants of influenza virus: implications for the mechanism of anti-influenza action of arbidol. Antiviral Res 81(2):132–140
Teissier E, Zandomeneghi G, Loquet A, Lavillette D, Lavergne JP, Montserret R, Cosset FL, Bockmann A, Meier BH, Penin F, Pecheur EI (2011) Mechanism of inhibition of enveloped virus membrane fusion by the antiviral drug arbidol. PLoS ONE 6(1):e15874
Corti D, Voss J, Gamblin SJ, Codoni G, Macagno A, Jarrossay D, Vachieri SG, Pinna D, Minola A, Vanzetta F, Silacci C, Fernandez-Rodriguez BM, Agatic G, Bianchi S, Giacchetto-Sasselli I, Calder L, Sallusto F, Collins P, Haire LF, Temperton N, Langedijk JP, Skehel JJ, Lanzavecchia A (2011) A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science 333(6044):850–856
Dreyfus C, Laursen NS, Kwaks T, Zuijdgeest D, Khayat R, Ekiert DC, Lee JH, Metlagel Z, Bujny MV, Jongeneelen M, van der Vlugt R, Lamrani M, Korse HJ, Geelen E, Sahin O, Sieuwerts M, Brakenhoff JP, Vogels R, Li OT, Poon LL, Peiris M, Koudstaal W, Ward AB, Wilson IA, Goudsmit J, Friesen RH (2012) Highly conserved protective epitopes on influenza B viruses. Science 337(6100):1343–1348
Ekiert DC, Bhabha G, Elsliger MA, Friesen RH, Jongeneelen M, Throsby M, Goudsmit J, Wilson IA (2009) Antibody recognition of a highly conserved influenza virus epitope. Science 324(5924):246–251
Ekiert DC, Friesen RH, Bhabha G, Kwaks T, Jongeneelen M, Yu W, Ophorst C, Cox F, Korse HJ, Brandenburg B, Vogels R, Brakenhoff JP, Kompier R, Koldijk MH, Cornelissen LA, Poon LL, Peiris M, Koudstaal W, Wilson IA, Goudsmit J (2011) A highly conserved neutralizing epitope on group 2 influenza A viruses. Science 333(6044):843–850
Kashyap AK, Steel J, Rubrum A, Estelles A, Briante R, Ilyushina NA, Xu L, Swale RE, Faynboym AM, Foreman PK, Horowitz M, Horowitz L, Webby R, Palese P, Lerner RA, Bhatt RR (2010) Protection from the 2009 H1N1 pandemic influenza by an antibody from combinatorial survivor-based libraries. PLoS Pathog 6(7):e1000990
Sui J, Hwang WC, Perez S, Wei G, Aird D, Chen LM, Santelli E, Stec B, Cadwell G, Ali M, Wan H, Murakami A, Yammanuru A, Han T, Cox NJ, Bankston LA, Donis RO, Liddington RC, Marasco WA (2009) Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses. Nat Struct Mol Biol 16(3):265–273
Throsby M, van den Brink E, Jongeneelen M, Poon LL, Alard P, Cornelissen L, Bakker A, Cox F, van Deventer E, Guan Y, Cinatl J, ter Meulen J, Lasters I, Carsetti R, Peiris M, de Kruif J, Goudsmit J (2008) Heterosubtypic neutralizing monoclonal antibodies cross-protective against H5N1 and H1N1 recovered from human IgM + memory B cells. PLoS ONE 3(12):e3942
Fodor E (2013) The RNA polymerase of influenza a virus: mechanisms of viral transcription and replication. Acta Virol 57(2):113–122
Gonzalez S, Zurcher T, Ortin J (1996) Identification of two separate domains in the influenza virus PB1 protein involved in the interaction with the PB2 and PA subunits: a model for the viral RNA polymerase structure. Nucleic Acids Res 24(22):4456–4463
Ohtsu Y, Honda Y, Sakata Y, Kato H, Toyoda T (2002) Fine mapping of the subunit binding sites of influenza virus RNA polymerase. Microbiol Immunol 46(3):167–175
Perez DR, Donis RO (1995) A 48-amino-acid region of influenza A virus PB1 protein is sufficient for complex formation with PA. J Virol 69(11):6932–6939
Poole EL, Medcalf L, Elton D, Digard P (2007) Evidence that the C-terminal PB2-binding region of the influenza A virus PB1 protein is a discrete alpha-helical domain. FEBS Lett 581(27):5300–5306
Hemerka JN, Wang D, Weng Y, Lu W, Kaushik RS, Jin J, Harmon AF, Li F (2009) Detection and characterization of influenza A virus PA-PB2 interaction through a bimolecular fluorescence complementation assay. J Virol 83(8):3944–3955
Furuta Y, Takahashi K, Fukuda Y, Kuno M, Kamiyama T, Kozaki K, Nomura N, Egawa H, Minami S, Watanabe Y, Narita H, Shiraki K (2002) In vitro and in vivo activities of anti-influenza virus compound T-705. Antimicrob Agents Chemother 46(4):977–981
Furuta Y, Takahashi K, Kuno-Maekawa M, Sangawa H, Uehara S, Kozaki K, Nomura N, Egawa H, Shiraki K (2005) Mechanism of action of T-705 against influenza virus. Antimicrob Agents Chemother 49(3):981–986
Furuta Y, Takahashi K, Shiraki K, Sakamoto K, Smee DF, Barnard DL, Gowen BB, Julander JG, Morrey JD (2009) T-705 (favipiravir) and related compounds: novel broad-spectrum inhibitors of RNA viral infections. Antiviral Res 82(3):95–102
Furuta Y, Gowen BB, Takahashi K, Shiraki K, Smee DF, Barnard DL (2013) Favipiravir (T-705), a novel viral RNA polymerase inhibitor. Antiviral Res 100(2):446–454
Dias A, Bouvier D, Crepin T, McCarthy AA, Hart DJ, Baudin F, Cusack S, Ruigrok RW (2009) The cap-snatching endonuclease of influenza virus polymerase resides in the PA subunit. Nature 458(7240):914–918
Yuan P, Bartlam M, Lou Z, Chen S, Zhou J, He X, Lv Z, Ge R, Li X, Deng T, Fodor E, Rao Z, Liu Y (2009) Crystal structure of an avian influenza polymerase PA(N) reveals an endonuclease active site. Nature 458(7240):909–913
Parkes KE, Ermert P, Fassler J, Ives J, Martin JA, Merrett JH, Obrecht D, Williams G, Klumpp K (2003) Use of a pharmacophore model to discover a new class of influenza endonuclease inhibitors. J Med Chem 46(7):1153–1164
Tomassini J, Selnick H, Davies ME, Armstrong ME, Baldwin J, Bourgeois M, Hastings J, Hazuda D, Lewis J, McClements W et al (1994) Inhibition of cap (m7G pppXm)-dependent endonuclease of influenza virus by 4-substituted 2,4-dioxobutanoic acid compounds. Antimicrob Agents Chemother 38(12):2827–2837
Tomassini JE, Davies ME, Hastings JC, Lingham R, Mojena M, Raghoobar SL, Singh SB, Tkacz JS, Goetz MA (1996) A novel antiviral agent which inhibits the endonuclease of influenza viruses. Antimicrob Agents Chemother 40(5):1189–1193
DuBois RM, Slavish PJ, Baughman BM, Yun MK, Bao J, Webby RJ, Webb TR, White SW (2012) Structural and biochemical basis for development of influenza virus inhibitors targeting the PA endonuclease. PLoS Pathog 8(8):e1002830
Kowalinski E, Zubieta C, Wolkerstorfer A, Szolar OH, Ruigrok RW, Cusack S (2012) Structural analysis of specific metal chelating inhibitor binding to the endonuclease domain of influenza pH1N1 (2009) polymerase. PLoS Pathog 8(8):e1002831
Bauman JD, Patel D, Baker SF, Vijayan RS, Xiang A, Parhi AK, Martinez-Sobrido L, Lavoie EJ, Das K, Arnold E (2013) Crystallographic fragment screening and structure-based optimization yields a new class of influenza endonuclease inhibitors. ACS Chem Biol 8(11):2501–2508
Iwai Y, Murakami K, Gomi Y, Hashimoto T, Asakawa Y, Okuno Y, Ishikawa T, Hatakeyama D, Echigo N, Kuzuhara T (2011) Anti-influenza activity of marchantins, macrocyclic bisbibenzyls contained in liverworts. PLoS ONE 6(5):e19825
Perez DR, Donis RO (2001) Functional analysis of PA binding by influenza a virus PB1: effects on polymerase activity and viral infectivity. J Virol 75(17):8127–8136
Sugiyama K, Obayashi E, Kawaguchi A, Suzuki Y, Tame JR, Nagata K, Park SY (2009) Structural insight into the essential PB1-PB2 subunit contact of the influenza virus RNA polymerase. EMBO J 28(12):1803–1811
Loregian A, Palu G (2005) Disruption of protein–protein interactions: towards new targets for chemotherapy. J Cell Physiol 204(3):750–762. doi:10.1002/jcp.20356
Palu G, Loregian A (2013) Inhibition of herpesvirus and influenza virus replication by blocking polymerase subunit interactions. Antiviral Res 99(3):318–327
Ghanem A, Mayer D, Chase G, Tegge W, Frank R, Kochs G, Garcia-Sastre A, Schwemmle M (2007) Peptide-mediated interference with influenza A virus polymerase. J Virol 81(14):7801–7804
Wunderlich K, Mayer D, Ranadheera C, Holler AS, Manz B, Martin A, Chase G, Tegge W, Frank R, Kessler U, Schwemmle M (2009) Identification of a PA-binding peptide with inhibitory activity against influenza A and B virus replication. PLoS ONE 4(10):e7517
He X, Zhou J, Bartlam M, Zhang R, Ma J, Lou Z, Li X, Li J, Joachimiak A, Zeng Z, Ge R, Rao Z, Liu Y (2008) Crystal structure of the polymerase PA(C)-PB1(N) complex from an avian influenza H5N1 virus. Nature 454(7208):1123–1126
Obayashi E, Yoshida H, Kawai F, Shibayama N, Kawaguchi A, Nagata K, Tame JR, Park SY (2008) The structural basis for an essential subunit interaction in influenza virus RNA polymerase. Nature 454(7208):1127–1131
Muratore G, Goracci L, Mercorelli B, Foeglein A, Digard P, Cruciani G, Palu G, Loregian A (2012) Small molecule inhibitors of influenza A and B viruses that act by disrupting subunit interactions of the viral polymerase. Proc Natl Acad Sci USA 109(16):6247–6252
Massari S, Nannetti G, Goracci L, Sancineto L, Muratore G, Sabatini S, Manfroni G, Mercorelli B, Cecchetti V, Facchini M, Palu G, Cruciani G, Loregian A, Tabarrini O (2013) Structural investigation of cycloheptathiophene-3-carboxamide derivatives targeting influenza virus polymerase assembly. J Med Chem 56(24):10118–10131
Loregian A, Coen DM (2006) Selective anti-cytomegalovirus compounds discovered by screening for inhibitors of subunit interactions of the viral polymerase. Chem Biol 13(2):191–200
Muratore G, Mercorelli B, Goracci L, Cruciani G, Digard P, Palu G, Loregian A (2012) Human cytomegalovirus inhibitor AL18 also possesses activity against influenza A and B viruses. Antimicrob Agents Chemother 56(11):6009–6013
Fukuoka M, Minakuchi M, Kawaguchi A, Nagata K, Kamatari YO, Kuwata K (2012) Structure-based discovery of anti-influenza virus A compounds among medicines. Biochimica et Biophysica Acta 1820(2):90–95
Kessler U, Castagnolo D, Pagano M, Deodato D, Bernardini M, Pilger B, Ranadheera C, Botta M (2013) Discovery and synthesis of novel benzofurazan derivatives as inhibitors of influenza A virus. Bioorg Med Chem Lett 23(20):5575–5577
Chase G, Wunderlich K, Reuther P, Schwemmle M (2011) Identification of influenza virus inhibitors which disrupt of viral polymerase protein–protein interactions. Methods 55(2):188–191
Li C, Ba Q, Wu A, Zhang H, Deng T, Jiang T (2013) A peptide derived from the C-terminus of PB1 inhibits influenza virus replication by interfering with viral polymerase assembly. FEBS J 280(4):1139–1149
Portela A, Digard P (2002) The influenza virus nucleoprotein: a multifunctional RNA-binding protein pivotal to virus replication. J Gen Virol 83(Pt 4):723–734
Amorim MJ, Read EK, Dalton RM, Medcalf L, Digard P (2007) Nuclear export of influenza A virus mRNAs requires ongoing RNA polymerase II activity. Traffic 8(1):1–11
Ye Q, Krug RM, Tao YJ (2006) The mechanism by which influenza A virus nucleoprotein forms oligomers and binds RNA. Nature 444(7122):1078–1082. doi:10.1038/nature05379
Ye Q, Guu TS, Mata DA, Kuo RL, Smith B, Krug RM, Tao YJ (2012) Biochemical and structural evidence in support of a coherent model for the formation of the double-helical influenza A virus ribonucleoprotein. MBio 4(1):e00467
Moeller A, Kirchdoerfer RN, Potter CS, Carragher B, Wilson IA (2012) Organization of the influenza virus replication machinery. Science 338(6114):1631–1634
Zheng W, Tao YJ (2013) Structure and assembly of the influenza A virus ribonucleoprotein complex. FEBS Lett 587(8):1206–1214
Cheng H, Wan J, Lin MI, Liu Y, Lu X, Liu J, Xu Y, Chen J, Tu Z, Cheng YS, Ding K (2012) Design, synthesis, and in vitro biological evaluation of 1H-1,2,3-triazole-4-carboxamide derivatives as new anti-influenza A agents targeting virus nucleoprotein. J Med Chem 55(5):2144–2153
Hung HC, Liu CL, Hsu JT, Horng JT, Fang MY, Wu SY, Ueng SH, Wang MY, Yaw CW, Hou MH (2012) Development of an anti-influenza drug screening assay targeting nucleoproteins with tryptophan fluorescence quenching. Anal Chem 84(15):6391–6399
Kao RY, Yang D, Lau LS, Tsui WH, Hu L, Dai J, Chan MP, Chan CM, Wang P, Zheng BJ, Sun J, Huang JD, Madar J, Chen G, Chen H, Guan Y, Yuen KY (2010) Identification of influenza A nucleoprotein as an antiviral target. Nat Biotechnol 28(6):600–605
Shen YF, Chen YH, Chu SY, Lin MI, Hsu HT, Wu PY, Wu CJ, Liu HW, Lin FY, Lin G, Hsu PH, Yang AS, Cheng YS, Wu YT, Wong CH, Tsai MD (2011) E339…R416 salt bridge of nucleoprotein as a feasible target for influenza virus inhibitors. Proc Natl Acad Sci USA 108(40):16515–16520
Su CY, Cheng TJ, Lin MI, Wang SY, Huang WI, Lin-Chu SY, Chen YH, Wu CY, Lai MM, Cheng WC, Wu YT, Tsai MD, Cheng YS, Wong CH (2010) High-throughput identification of compounds targeting influenza RNA-dependent RNA polymerase activity. Proc Natl Acad Sci USA 107(45):19151–19156
Lejal N, Tarus B, Bouguyon E, Chenavas S, Bertho N, Delmas B, Ruigrok RW, Di Primo C, Slama-Schwok A (2013) Structure-based discovery of the novel antiviral properties of naproxen against the nucleoprotein of influenza A virus. Antimicrob Agents Chemother 57(5):2231–2242
Lin L, Liu Q, Berube N, Detmer S, Zhou Y (2012) 5′-Triphosphate-short interfering RNA: potent inhibition of influenza A virus infection by gene silencing and RIG-I activation. J Virol 86(19):10359–10369
Tompkins SM, Lo CY, Tumpey TM, Epstein SL (2004) Protection against lethal influenza virus challenge by RNA interference in vivo. Proc Natl Acad Sci USA 101(23):8682–8686
Chenavas S, Estrozi LF, Slama-Schwok A, Delmas B, Di Primo C, Baudin F, Li X, Crepin T, Ruigrok RW (2013) Monomeric nucleoprotein of influenza A virus. PLoS Pathog 9(3):e1003275
Ruigrok RW, Baudin F (1995) Structure of influenza virus ribonucleoprotein particles. II. Purified RNA-free influenza virus ribonucleoprotein forms structures that are indistinguishable from the intact influenza virus ribonucleoprotein particles. J Gen Virol 76(Pt 4):1009–1014
Amorim MJ, Kao RY, Digard P (2013) Nucleozin targets cytoplasmic trafficking of viral ribonucleoprotein-Rab11 complexes in influenza A virus infection. J Virol 87(8):4694–4703
Hale BG, Randall RE, Ortin J, Jackson D (2008) The multifunctional NS1 protein of influenza A viruses. J Gen Virol 89(Pt 10):2359–2376
Ayllon J, Hale BG, Garcia-Sastre A (2012) Strain-specific contribution of NS1-activated phosphoinositide 3-kinase signaling to influenza A virus replication and virulence. J Virol 86(9):5366–5370
Marc D, Barbachou S, Soubieux D (2013) The RNA-binding domain of influenzavirus non-structural protein-1 cooperatively binds to virus-specific RNA sequences in a structure-dependent manner. Nucleic Acids Res 41(1):434–449
Bergmann M, Garcia-Sastre A, Carnero E, Pehamberger H, Wolff K, Palese P, Muster T (2000) Influenza virus NS1 protein counteracts PKR-mediated inhibition of replication. J Virol 74(13):6203–6206
Min JY, Krug RM (2006) The primary function of RNA binding by the influenza A virus NS1 protein in infected cells: inhibiting the 2′-5′ oligo (A) synthetase/RNase L pathway. Proc Natl Acad Sci USA 103(18):7100–7105
Ramos I, Carnero E, Bernal-Rubio D, Seibert CW, Westera L, Garcia-Sastre A, Fernandez-Sesma A (2013) Contribution of double-stranded RNA and CPSF30 binding domains of influenza virus NS1 to the inhibition of type I interferon production and activation of human dendritic cells. J Virol 87(5):2430–2440
Gack MU, Albrecht RA, Urano T, Inn KS, Huang IC, Carnero E, Farzan M, Inoue S, Jung JU, Garcia-Sastre A (2009) Influenza A virus NS1 targets the ubiquitin ligase TRIM25 to evade recognition by the host viral RNA sensor RIG-I. Cell Host Microbe 5(5):439–449
Zhu Z, Shi Z, Yan W, Wei J, Shao D, Deng X, Wang S, Li B, Tong G, Ma Z (2013) Nonstructural protein 1 of influenza A virus interacts with human guanylate-binding protein 1 to antagonize antiviral activity. PLoS ONE 8(2):e55920
Gao S, Song L, Li J, Zhang Z, Peng H, Jiang W, Wang Q, Kang T, Chen S, Huang W (2012) Influenza A virus-encoded NS1 virulence factor protein inhibits innate immune response by targeting IKK. Cell Microbiol 14(12):1849–1866
Santos A, Pal S, Chacon J, Meraz K, Gonzalez J, Prieto K, Rosas-Acosta G (2013) SUMOylation affects the interferon blocking activity of the influenza A nonstructural protein NS1 without affecting its stability or cellular localization. J Virol 87(10):5602–5620
Wang W, Riedel K, Lynch P, Chien CY, Montelione GT, Krug RM (1999) RNA binding by the novel helical domain of the influenza virus NS1 protein requires its dimer structure and a small number of specific basic amino acids. RNA 5(2):195–205
Basu D, Walkiewicz MP, Frieman M, Baric RS, Auble DT, Engel DA (2009) Novel influenza virus NS1 antagonists block replication and restore innate immune function. J Virol 83(4):1881–1891
Jablonski JJ, Basu D, Engel DA, Geysen HM (2012) Design, synthesis, and evaluation of novel small molecule inhibitors of the influenza virus protein NS1. Bioorg Med Chem 20(1):487–497
Walkiewicz MP, Basu D, Jablonski JJ, Geysen HM, Engel DA (2011) Novel inhibitor of influenza non-structural protein 1 blocks multi-cycle replication in an RNase L-dependent manner. J Gen Virol 92(Pt 1):60–70
Ai H, Zheng F, Zhu C, Sun T, Zhang L, Liu X, Li X, Zhu G, Liu H (2010) Discovery of novel influenza inhibitors targeting the interaction of dsRNA with the NS1 protein by structure-based virtual screening. Int J Bioinform Res Appl 6(5):449–460
Maroto M, Fernandez Y, Ortin J, Pelaez F, Cabello MA (2008) Development of an HTS assay for the search of anti-influenza agents targeting the interaction of viral RNA with the NS1 protein. J Biomol Screen 13(7):581–590
Cho EJ, Xia S, Ma LC, Robertus J, Krug RM, Anslyn EV, Montelione GT, Ellington AD (2012) Identification of influenza virus inhibitors targeting NS1A utilizing fluorescence polarization-based high-throughput assay. J Biomol Screen 17(4):448–459
You L, Cho EJ, Leavitt J, Ma LC, Montelione GT, Anslyn EV, Krug RM, Ellington A, Robertus JD (2011) Synthesis and evaluation of quinoxaline derivatives as potential influenza NS1A protein inhibitors. Bioorg Med Chem Lett 21(10):3007–3011
Ilyushina NA, Hoffmann E, Salomon R, Webster RG, Govorkova EA (2007) Amantadine-oseltamivir combination therapy for H5N1 influenza virus infection in mice. Antivir Ther 12(3):363–370
Smee DF, Hurst BL, Wong MH, Bailey KW, Tarbet EB, Morrey JD, Furuta Y (2010) Effects of the combination of favipiravir (T-705) and oseltamivir on influenza A virus infections in mice. Antimicrob Agents Chemother 54(1):126–133
Ilyushina NA, Bovin NV, Webster RG, Govorkova EA (2006) Combination chemotherapy, a potential strategy for reducing the emergence of drug-resistant influenza A variants. Antiviral Res 70(3):121–131
Ison MG, Gnann JW Jr, Nagy-Agren S, Treannor J, Paya C, Steigbigel R, Elliott M, Weiss HL, Hayden FG (2003) Safety and efficacy of nebulized zanamivir in hospitalized patients with serious influenza. Antivir Ther 8(3):183–190
Karlas A, Machuy N, Shin Y, Pleissner KP, Artarini A, Heuer D, Becker D, Khalil H, Ogilvie LA, Hess S, Maurer AP, Muller E, Wolff T, Rudel T, Meyer TF (2010) Genome-wide RNAi screen identifies human host factors crucial for influenza virus replication. Nature 463(7282):818–822
Konig R, Stertz S, Zhou Y, Inoue A, Hoffmann HH, Bhattacharyya S, Alamares JG, Tscherne DM, Ortigoza MB, Liang Y, Gao Q, Andrews SE, Bandyopadhyay S, De Jesus P, Tu BP, Pache L, Shih C, Orth A, Bonamy G, Miraglia L, Ideker T, Garcia-Sastre A, Young JA, Palese P, Shaw ML, Chanda SK (2010) Human host factors required for influenza virus replication. Nature 463(7282):813–817
Muller KH, Kakkola L, Nagaraj AS, Cheltsov AV, Anastasina M, Kainov DE (2012) Emerging cellular targets for influenza antiviral agents. Trends Pharmacol Sci 33(2):89–99
Shapira SD, Gat-Viks I, Shum BO, Dricot A, de Grace MM, Wu L, Gupta PB, Hao T, Silver SJ, Root DE, Hill DE, Regev A, Hacohen N (2009) A physical and regulatory map of host-influenza interactions reveals pathways in H1N1 infection. Cell 139(7):1255–1267
Hao L, Sakurai A, Watanabe T, Sorensen E, Nidom CA, Newton MA, Ahlquist P, Kawaoka Y (2008) Drosophila RNAi screen identifies host genes important for influenza virus replication. Nature 454(7206):890–893
Shaw ML (2011) The host interactome of influenza virus presents new potential targets for antiviral drugs. Rev Med Virol 21(6):358–369
Jorba N, Juarez S, Torreira E, Gastaminza P, Zamarreno N, Albar JP, Ortin J (2008) Analysis of the interaction of influenza virus polymerase complex with human cell factors. Proteomics 8(10):2077–2088
Tafforeau L, Chantier T, Pradezynski F, Pellet J, Mangeot PE, Vidalain PO, Andre P, Rabourdin-Combe C, Lotteau V (2011) Generation and comprehensive analysis of an influenza virus polymerase cellular interaction network. J Virol 85(24):13010–13018
Bottcher E, Matrosovich T, Beyerle M, Klenk HD, Garten W, Matrosovich M (2006) Proteolytic activation of influenza viruses by serine proteases TMPRSS2 and HAT from human airway epithelium. J Virol 80(19):9896–9898
Bottcher E, Freuer C, Steinmetzer T, Klenk HD, Garten W (2009) MDCK cells that express proteases TMPRSS2 and HAT provide a cell system to propagate influenza viruses in the absence of trypsin and to study cleavage of HA and its inhibition. Vaccine 27(45):6324–6329
Hatesuer B, Bertram S, Mehnert N, Bahgat MM, Nelson PS, Pohlman S, Schughart K (2013) Tmprss2 is essential for influenza H1N1 virus pathogenesis in mice. PLoS Pathog 9(12):e1003774
Horimoto T, Nakayama K, Smeekens SP, Kawaoka Y (1994) Proprotein-processing endoproteases PC6 and furin both activate hemagglutinin of virulent avian influenza viruses. J Virol 68(9):6074–6078
Okumura Y, Takahashi E, Yano M, Ohuchi M, Daidoji T, Nakaya T, Bottcher E, Garten W, Klenk HD, Kido H (2010) Novel type II transmembrane serine proteases, MSPL and TMPRSS13, Proteolytically activate membrane fusion activity of the hemagglutinin of highly pathogenic avian influenza viruses and induce their multicycle replication. J Virol 84(10):5089–5096
Kido H, Okumura Y, Yamada H, Le TQ, Yano M (2007) Proteases essential for human influenza virus entry into cells and their inhibitors as potential therapeutic agents. Curr Pharm Des 13(4):405–414
Zhirnov OP, Ikizler MR, Wright PF (2002) Cleavage of influenza a virus hemagglutinin in human respiratory epithelium is cell associated and sensitive to exogenous antiproteases. J Virol 76(17):8682–8689
Zhirnov OP, Ovcharenko AV, Bukrinskaya AG (1984) Suppression of influenza virus replication in infected mice by protease inhibitors. J Gen Virol 65(Pt 1):191–196
Zhirnov OP, Kirzhner LS, Ovcharenko AV, Malyshev NA (1996) Clinical effectiveness of aprotinin aerosol in influenza and parainfluenza. Vestn Ross Akad Med Nauk 5:26–31
Zhirnov OP, Matrosovich TY, Matrosovich MN, Klenk HD (2011) Aprotinin, a protease inhibitor, suppresses proteolytic activation of pandemic H1N1v influenza virus. Antivir Chem Chemother 21(4):169–174
Meyer D, Sielaff F, Hammami M, Bottcher-Friebertshauser E, Garten W, Steinmetzer T (2013) Identification of the first synthetic inhibitors of the type II transmembrane serine protease TMPRSS2 suitable for inhibition of influenza virus activation. Biochem J 452(2):331–343
Stieneke-Grober A, Vey M, Angliker H, Shaw E, Thomas G, Roberts C, Klenk HD, Garten W (1992) Influenza virus hemagglutinin with multibasic cleavage site is activated by furin, a subtilisin-like endoprotease. EMBO J 11(7):2407–2414
Becker GL, Lu Y, Hardes K, Strehlow B, Levesque C, Lindberg I, Sandvig K, Bakowsky U, Day R, Garten W, Steinmetzer T (2012) Highly potent inhibitors of proprotein convertase furin as potential drugs for treatment of infectious diseases. J Biol Chem 287(26):21992–22003
Becker GL, Sielaff F, Than ME, Lindberg I, Routhier S, Day R, Lu Y, Garten W, Steinmetzer T (2010) Potent inhibitors of furin and furin-like proprotein convertases containing decarboxylated P1 arginine mimetics. J Med Chem 53(3):1067–1075
Malakhov MP, Aschenbrenner LM, Smee DF, Wandersee MK, Sidwell RW, Gubareva LV, Mishin VP, Hayden FG, Kim DH, Ing A, Campbell ER, Yu M, Fang F (2006) Sialidase fusion protein as a novel broad-spectrum inhibitor of influenza virus infection. Antimicrob Agents Chemother 50(4):1470–1479
Nicholls JM, Moss RB, Haslam SM (2013) The use of sialidase therapy for respiratory viral infections. Antiviral Res 98(3):401–409
Triana-Baltzer GB, Sanders RL, Hedlund M, Jensen KA, Aschenbrenner LM, Larson JL, Fang F (2011) Phenotypic and genotypic characterization of influenza virus mutants selected with the sialidase fusion protein DAS181. J Antimicrob Chemother 66(1):15–28
Triana-Baltzer GB, Gubareva LV, Klimov AI, Wurtman DF, Moss RB, Hedlund M, Larson JL, Belshe RB, Fang F (2009) Inhibition of neuraminidase inhibitor-resistant influenza virus by DAS181, a novel sialidase fusion protein. PLoS ONE 4(11):e7838
Nicholls JM, Aschenbrenner LM, Paulson JC, Campbell ER, Malakhov MP, Wurtman DF, Yu M, Fang F (2008) Comment on: concerns of using sialidase fusion protein as an experimental drug to combat seasonal and pandemic influenza. J Antimicrob Chemother 62(2):426–428 (author reply 428–429)
Moss RB, Hansen C, Sanders RL, Hawley S, Li T, Steigbigel RT (2012) A phase II study of DAS181, a novel host directed antiviral for the treatment of influenza infection. J Infect Dis 206(12):1844–1851
Takeda M, Leser GP, Russell CJ, Lamb RA (2003) Influenza virus hemagglutinin concentrates in lipid raft microdomains for efficient viral fusion. Proc Natl Acad Sci USA 100(25):14610–14617
Barman S, Nayak DP (2007) Lipid raft disruption by cholesterol depletion enhances influenza A virus budding from MDCK cells. J Virol 81(22):12169–12178
Wang X, Hinson ER, Cresswell P (2007) The interferon-inducible protein viperin inhibits influenza virus release by perturbing lipid rafts. Cell Host Microbe 2(2):96–105
Tan KS, Ng WC, Seet JE, Olfat F, Engelward BP, Chow VT (2014) Investigating the efficacy of pamidronate, a chemical inhibitor of farnesyl pyrophosphate synthase, in the inhibition of influenza virus infection in vitro and in vivo. Mol Med Rep 9(1):51–56
Nagata K, Kawaguchi A, Naito T (2008) Host factors for replication and transcription of the influenza virus genome. Rev Med Virol 18(4):247–260
Naito T, Momose F, Kawaguchi A, Nagata K (2007) Involvement of Hsp90 in assembly and nuclear import of influenza virus RNA polymerase subunits. J Virol 81(3):1339–1349
Chase G, Deng T, Fodor E, Leung BW, Mayer D, Schwemmle M, Brownlee G (2008) Hsp90 inhibitors reduce influenza virus replication in cell culture. Virology 377(2):431–439
Pleschka S, Wolff T, Ehrhardt C, Hobom G, Planz O, Rapp UR, Ludwig S (2001) Influenza virus propagation is impaired by inhibition of the Raf/MEK/ERK signalling cascade. Nat Cell Biol 3(3):301–305
Kujime K, Hashimoto S, Gon Y, Shimizu K, Horie T (2000) p38 mitogen-activated protein kinase and c-jun-NH2-terminal kinase regulate RANTES production by influenza virus-infected human bronchial epithelial cells. J Immunol 164(6):3222–3228
Marjuki H, Alam MI, Ehrhardt C, Wagner R, Planz O, Klenk HD, Ludwig S, Pleschka S (2006) Membrane accumulation of influenza A virus hemagglutinin triggers nuclear export of the viral genome via protein kinase Calpha-mediated activation of ERK signaling. J Biol Chem 281(24):16707–16715
Nencioni L, Iuvara A, Aquilano K, Ciriolo MR, Cozzolino F, Rotilio G, Garaci E, Palamara AT (2003) Influenza A virus replication is dependent on an antioxidant pathway that involves GSH and Bcl-2. FASEB J 17(6):758–760
Nencioni L, De Chiara G, Sgarbanti R, Amatore D, Aquilano K, Marcocci ME, Serafino A, Torcia M, Cozzolino F, Ciriolo MR, Garaci E, Palamara AT (2009) Bcl-2 expression and p38MAPK activity in cells infected with influenza A virus: impact on virally induced apoptosis and viral replication. J Biol Chem 284(23):16004–16015
Go YM, Kang SM, Roede JR, Orr M, Jones DP (2011) Increased inflammatory signaling and lethality of influenza H1N1 by nuclear thioredoxin-1. PLoS ONE 6(4):e18918
Nencioni L, Sgarbanti R, De Chiara G, Garaci E, Palamara AT (2007) Influenza virus and redox mediated cell signaling: a complex network of virus/host interaction. New Microbiol 30(4):367–375
Shin YK, Liu Q, Tikoo SK, Babiuk LA, Zhou Y (2007) Influenza A virus NS1 protein activates the phosphatidylinositol 3-kinase (PI3K)/Akt pathway by direct interaction with the p85 subunit of PI3K. J Gen Virol 88(Pt 1):13–18
Kumar N, Liang Y, Parslow TG (2011) Receptor tyrosine kinase inhibitors block multiple steps of influenza a virus replication. J Virol 85(6):2818–2827
Droebner K, Pleschka S, Ludwig S, Planz O (2011) Antiviral activity of the MEK-inhibitor U0126 against pandemic H1N1v and highly pathogenic avian influenza virus in vitro and in vivo. Antiviral Res 92(2):195–203
Ludwig S, Wolff T, Ehrhardt C, Wurzer WJ, Reinhardt J, Planz O, Pleschka S (2004) MEK inhibition impairs influenza B virus propagation without emergence of resistant variants. FEBS Lett 561(1–3):37–43
De Flora S, Grassi C, Carati L (1997) Attenuation of influenza-like symptomatology and improvement of cell-mediated immunity with long-term N-acetylcysteine treatment. Eur Respir J 10(7):1535–1541
Geiler J, Michaelis M, Naczk P, Leutz A, Langer K, Doerr HW, Cinatl J Jr (2010) N-acetyl-l-cysteine (NAC) inhibits virus replication and expression of pro-inflammatory molecules in A549 cells infected with highly pathogenic H5N1 influenza A virus. Biochem Pharmacol 79(3):413–420
Michaelis M, Geiler J, Naczk P, Sithisarn P, Leutz A, Doerr HW, Cinatl J Jr (2011) Glycyrrhizin exerts antioxidative effects in H5N1 influenza A virus-infected cells and inhibits virus replication and pro-inflammatory gene expression. PLoS ONE 6(5):e19705
Cai J, Chen Y, Seth S, Furukawa S, Compans RW, Jones DP (2003) Inhibition of influenza infection by glutathione. Free Radic Biol Med 34(7):928–936
Sgarbanti R, Nencioni L, Amatore D, Coluccio P, Fraternale A, Sale P, Mammola CL, Carpino G, Gaudio E, Magnani M, Ciriolo MR, Garaci E, Palamara AT (2011) Redox regulation of the influenza hemagglutinin maturation process: a new cell-mediated strategy for anti-influenza therapy. Antioxid Redox Signal 15(3):593–606
Fioravanti R, Celestino I, Costi R, Cuzzucoli Crucitti G, Pescatori L, Mattiello L, Novellino E, Checconi P, Palamara AT, Nencioni L, Di Santo R (2012) Effects of polyphenol compounds on influenza A virus replication and definition of their mechanism of action. Bioorg Med Chem 20(16):5046–5052
Haasbach E, Hartmayer C, Planz O (2013) Combination of MEK inhibitors and oseltamivir leads to synergistic antiviral effects after influenza A virus infection in vitro. Antiviral Res 98(2):319–324
Rossignol JF, La Frazia S, Chiappa L, Ciucci A, Santoro MG (2009) Thiazolides, a new class of anti-influenza molecules targeting viral hemagglutinin at the post-translational level. J Biol Chem 284(43):29798–29808
Zhou Z, Jiang X, Liu D, Fan Z, Hu X, Yan J, Wang M, Gao GF (2009) Autophagy is involved in influenza A virus replication. Autophagy 5(3):321–328
Shin YK, Liu Q, Tikoo SK, Babiuk LA, Zhou Y (2007) Effect of the phosphatidylinositol 3-kinase/Akt pathway on influenza A virus propagation. J Gen Virol 88(Pt 3):942–950
Murray JL, McDonald NJ, Sheng J, Shaw MW, Hodge TW, Rubin DH, O’Brien WA, Smee DF (2012) Inhibition of influenza A virus replication by antagonism of a PI3K-AKT-mTOR pathway member identified by gene-trap insertional mutagenesis. Antivir Chem Chemother 22(5):205–215
Kurtz JE, Ray-Coquard I (2012) PI3 kinase inhibitors in the clinic: an update. Anticancer Res 32(7):2463–2470
Bonizzi G, Karin M (2004) The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol 25(6):280–288
Mazur I, Wurzer WJ, Ehrhardt C, Pleschka S, Puthavathana P, Silberzahn T, Wolff T, Planz O, Ludwig S (2007) Acetylsalicylic acid (ASA) blocks influenza virus propagation via its NF-kappaB-inhibiting activity. Cell Microbiol 9(7):1683–1694
Nimmerjahn F, Dudziak D, Dirmeier U, Hobom G, Riedel A, Schlee M, Staudt LM, Rosenwald A, Behrends U, Bornkamm GW, Mautner J (2004) Active NF-kappaB signalling is a prerequisite for influenza virus infection. J Gen Virol 85(Pt 8):2347–2356
Wurzer WJ, Ehrhardt C, Pleschka S, Berberich-Siebelt F, Wolff T, Walczak H, Planz O, Ludwig S (2004) NF-kappaB-dependent induction of tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and Fas/FasL is crucial for efficient influenza virus propagation. J Biol Chem 279(30):30931–30937
Flory E, Kunz M, Scheller C, Jassoy C, Stauber R, Rapp UR, Ludwig S (2000) Influenza virus-induced NF-kappaB-dependent gene expression is mediated by overexpression of viral proteins and involves oxidative radicals and activation of IkappaB kinase. J Biol Chem 275(12):8307–8314
Uchide N, Ohyama K, Bessho T, Yuan B, Yamakawa T (2002) Effect of antioxidants on apoptosis induced by influenza virus infection: inhibition of viral gene replication and transcription with pyrrolidine dithiocarbamate. Antiviral Res 56(3):207–217
Uchide N, Toyoda H (2011) Antioxidant therapy as a potential approach to severe influenza-associated complications. Molecules 16(3):2032–2052
Ehrhardt C, Ruckle A, Hrincius ER, Haasbach E, Anhlan D, Ahmann K, Banning C, Reiling SJ, Kuhn J, Strobl S, Vitt D, Leban J, Planz O, Ludwig S (2013) The NF-kappaB inhibitor SC75741 efficiently blocks influenza virus propagation and confers a high barrier for development of viral resistance. Cell Microbiol 15(7):1198–1211
Leban J, Baierl M, Mies J, Trentinaglia V, Rath S, Kronthaler K, Wolf K, Gotschlich A, Seifert MH (2007) A novel class of potent NF-kappaB signaling inhibitors. Bioorg Med Chem Lett 17(21):5858–5862
Lee SM, Cheung CY, Nicholls JM, Hui KP, Leung CY, Uiprasertkul M, Tipoe GL, Lau YL, Poon LL, Ip NY, Guan Y, Peiris JS (2008) Hyperinduction of cyclooxygenase-2-mediated proinflammatory cascade: a mechanism for the pathogenesis of avian influenza H5N1 infection. J Infect Dis 198(4):525–535
Lee SM, Gai WW, Cheung TK, Peiris JS (2011) Antiviral effect of a selective COX-2 inhibitor on H5N1 infection in vitro. Antiviral Res 91(3):330–334
Beloso A, Martinez C, Valcarcel J, Santaren JF, Ortin J (1992) Degradation of cellular mRNA during influenza virus infection: its possible role in protein synthesis shutoff. J Gen Virol 73(Pt 3):575–581
Hoffmann HH, Kunz A, Simon VA, Palese P, Shaw ML (2011) Broad-spectrum antiviral that interferes with de novo pyrimidine biosynthesis. Proc Natl Acad Sci USA 108(14):5777–5782
Zhang L, Das P, Schmolke M, Manicassamy B, Wang Y, Deng X, Cai L, Tu BP, Forst CV, Roth MG, Levy DE, Garcia-Sastre A, de Brabander J, Phillips MA, Fontoura BM (2012) Inhibition of pyrimidine synthesis reverses viral virulence factor-mediated block of mRNA nuclear export. J Cell Biol 196(3):315–326
Smee DF, Hurst BL, Day CW (2012) D282, a non-nucleoside inhibitor of influenza virus infection that interferes with de novo pyrimidine biosynthesis. Antivir Chem Chemother 22(6):263–272
Garcia-Sastre A (2013) Lessons from lipids in the fight against influenza. Cell 154(1):22–23
Tam VC, Quehenberger O, Oshansky CM, Suen R, Armando AM, Treuting PM, Thomas PG, Dennis EA, Aderem A (2013) Lipidomic profiling of influenza infection identifies mediators that induce and resolve inflammation. Cell 154(1):213–227
Morita M, Kuba K, Ichikawa A, Nakayama M, Katahira J, Iwamoto R, Watanebe T, Sakabe S, Daidoji T, Nakamura S, Kadowaki A, Ohto T, Nakanishi H, Taguchi R, Nakaya T, Murakami M, Yoneda Y, Arai H, Kawaoka Y, Penninger JM, Arita M, Imai Y (2013) The lipid mediator protectin D1 inhibits influenza virus replication and improves severe influenza. Cell 153(1):112–125
Levy BD, Kohli P, Gotlinger K, Haworth O, Hong S, Kazani S, Israel E, Haley KJ, Serhan CN (2007) Protectin D1 is generated in asthma and dampens airway inflammation and hyperresponsiveness. J Immunol 178(1):496–502
Serhan CN (2007) Resolution phase of inflammation: novel endogenous anti-inflammatory and proresolving lipid mediators and pathways. Annu Rev Immunol 25:101–137
Garcia-Sastre A (2011) Induction and evasion of type I interferon responses by influenza viruses. Virus Res 162(1–2):12–18
Martinez-Gil L, Ayllon J, Ortigoza MB, Garcia-Sastre A, Shaw ML, Palese P (2012) Identification of small molecules with type I interferon inducing properties by high-throughput screening. PLoS ONE 7(11):e49049
Ortigoza MB, Dibben O, Maamary J, Martinez-Gil L, Leyva-Grado VH, Abreu P Jr, Ayllon J, Palese P, Shaw ML (2012) A novel small molecule inhibitor of influenza A viruses that targets polymerase function and indirectly induces interferon. PLoS Pathog 8(4):e1002668
Ranjan P, Jayashankar L, Deyde V, Zeng H, Davis WG, Pearce MB, Bowzard JB, Hoelscher MA, Jeisy-Scott V, Wiens ME, Gangappa S, Gubareva L, Garcia-Sastre A, Katz JM, Tumpey TM, Fujita T, Sambhara S (2010) 5′PPP-RNA induced RIG-I activation inhibits drug-resistant avian H5N1 as well as 1918 and 2009 pandemic influenza virus replication. Virol J 7:102
Goulet ML, Olagnier D, Xu Z, Paz S, Belgnaoui SM, Lafferty EI, Janelle V, Arguello M, Paquet M, Ghneim K, Richards S, Smith A, Wilkinson P, Cameron M, Kalinke U, Qureshi S, Lamarre A, Haddad EK, Sekaly RP, Peri S, Balachandran S, Lin R, Hiscott J (2013) Systems analysis of a RIG-I agonist inducing broad spectrum inhibition of virus infectivity. PLoS Pathog 9(4):e1003298
Tamm I, Folkers K, Horsfall FL Jr (1953) Inhibition of influenza virus multiplication by alkyl derivatives of benzimidazole. II. Measurement of inhibitory activity by hemagglutination titrations. J Exp Med 98(3):229–243
Wang S, Zhang J, Ye X (2012) Protein kinase inhibitor flavopiridol inhibits the replication of influenza virus in vitro. Wei Sheng Wu Xue Bao 52(9):1137–1142
Watanabe K, Takizawa N, Katoh M, Hoshida K, Kobayashi N, Nagata K (2001) Inhibition of nuclear export of ribonucleoprotein complexes of influenza virus by leptomycin B. Virus Res 77(1):31–42
Alamares-Sapuay JG, Martinez-Gil L, Stertz S, Miller MS, Shaw ML, Palese P (2013) Serum- and glucocorticoid-regulated kinase 1 is required for nuclear export of the ribonucleoprotein of influenza A virus. J Virol 87(10):6020–6026
Martinez-Gil L, Alamares-Sapuay JG, Ramana Reddy MV, Goff PH, Premkumar Reddy E, Palese P (2013) A small molecule multi-kinase inhibitor reduces influenza A virus replication by restricting viral RNA synthesis. Antiviral Res 100(1):29–37
Loregian A, Palù G (2013) How academic labs can approach the drug discovery process as a way to synergize with big pharma. Trends Microbiol 21(6):261–264
Acknowledgments
Work in the authors’ laboratory was supported by MURST EX60 %, Progetto di Ricerca di Ateneo 2007 (grant no. CPDA074945), and PRIN 2008 (grant no. 20085FF4J4) to A.L., by Regione Veneto and Progetto Strategico di Ateneo 2008 to G.P., by Italian Ministry of Health and Istituto Superiore Sanità, Progetto finalizzato 2009 “Studio e sviluppo di nuovi farmaci antivirali contro infezioni da virus influenzale A-H1N1” to A.L. and G.P., and by ESCMID Research Grant 2013 to B.M.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Loregian and B. Mercorelli contributed equally to this work.
Rights and permissions
About this article
Cite this article
Loregian, A., Mercorelli, B., Nannetti, G. et al. Antiviral strategies against influenza virus: towards new therapeutic approaches. Cell. Mol. Life Sci. 71, 3659–3683 (2014). https://doi.org/10.1007/s00018-014-1615-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-014-1615-2