Abstract
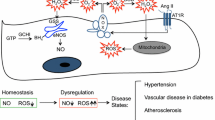
Endothelial cells (ECs) are present throughout blood vessels and have variable roles in both physiological and pathological settings. EC fate is altered and regulated by several key factors in physiological or pathological conditions. Reactive nitrogen species and reactive oxygen species derived from NAD(P)H oxidases, mitochondria, or nitric oxide-producing enzymes are not only cytotoxic but also compose a signaling network in the redox system. The formation, actions, key molecular interactions, and physiological and pathological relevance of redox signals in ECs remain unclear. We review the identities, sources, and biological actions of oxidants and reductants produced during EC function or dysfunction. Further, we discuss how ECs shape key redox sensors and examine the biological functions, transcriptional responses, and post-translational modifications evoked by the redox system in ECs. We summarize recent findings regarding the mechanisms by which redox signals regulate the fate of ECs and address the outcome of altered EC fate in health and disease. Future studies will examine if the redox biology of ECs can be targeted in pathophysiological conditions.




Similar content being viewed by others
Abbreviations
- 3-MST:
-
3-Mercaptopyruvate sulfurtransferase
- AICAR:
-
5-Amino-4-imidazole carboxamide riboside
- AMPK:
-
Adenosine monophosphate-activated protein kinase
- BAT:
-
Brown adipose tissue
- BH4:
-
Tetrahydrobiopterin
- BMP:
-
Bone morphogenetic protein
- CAT:
-
Catalase
- CBS:
-
Cystathionine β-synthase
- CEP:
-
ω-(2-Carboxyethyl)pyrrole
- CSE:
-
Cystathionine γ-lyase
- EPA:
-
Eicosapentaenoic acid
- ER:
-
Endoplasmic reticulum
- ETC:
-
Mitochondrial electron-transport chain
- FoxO:
-
Forkhead homeobox type O
- Gpx:
-
Glutathione peroxidase
- GR:
-
Glutathione reductase
- Grx:
-
Glutaredoxin
- GTPCH I:
-
GTP-cyclohydrolase I
- H2O2 :
-
Hydrogen peroxide
- HIF-1:
-
Hypoxia-inducible factor 1
- HUVEC:
-
Human umbilical vein endothelial cells
- ICAM-1:
-
Intercellular adhesion molecule-1
- IKK:
-
IкB kinase
- JNK:
-
c-Jun N-terminal kinase
- Keap1:
-
Kelch-like ECH-associated protein 1
- LDLR:
-
Low-density lipoprotein receptor
- LKB1:
-
Liver kinase B1
- MCP:
-
Monocyte chemoattractant protein
- NaHS:
-
Sodium hydrosulfide
- NO:
-
Nitric oxide
- NO2-FAs:
-
Nitro-fatty acids
- NOS:
-
Nitric oxide synthase
- NOX:
-
NADPH oxidase
- Nrf2:
-
Nuclear factor erythroid-2-related factor 2
- O ·−2 :
-
Superoxide anion
- OGD:
-
Hypoxia and glucose deprivation
- PAG:
-
DL-propargylglycine
- PI3K:
-
Phosphoinositide 3-kinase
- Prx:
-
Peroxiredoxins
- PTEN:
-
Phosphatase and tensin homolog
- RNS:
-
Reactive nitrogen species
- ROS:
-
Reactive oxygen species
- RSH:
-
Thiol
- RSO2H:
-
Sulfinic acids
- RSO3H:
-
Sulfonic acids
- RSOH:
-
Sulfenic acids
- RS-SG:
-
S-glutathionylation
- RS-SR, RS-SR’:
-
Disulfide bonds
- SIRT1:
-
Sirtuin 1
- SOD:
-
Superoxide dismutase
- SM:
-
Smooth muscle
- SREBP2:
-
Sterol regulatory element binding protein 2
- TP:
-
Thromboxane receptor
- Trx:
-
Thioredoxin
- VEGF:
-
Vascular endothelial growth factor
- VEGFR:
-
VEGF receptor
- VCAM-1:
-
Vascular cell adhesion molecule-1
- WAT:
-
White adipose tissue
References
Cines DB, Pollak ES, Buck CA et al (1998) Endothelial cells in physiology and in the pathophysiology of vascular disorders. Blood 91:3527–3561
Dong Y, Zhang M, Liang B et al (2010) Reduction of AMP-activated protein kinase {alpha}2 increases endoplasmic reticulum stress and atherosclerosis in vivo. Circulation 121:792–803
Budhiraja R, Tuder RM, Hassoun PM (2004) Endothelial dysfunction in pulmonary hypertension. Circulation 109:159–165
Celermajer DS, Sorensen KE, Spiegelhalter DJ, Georgakopoulos D, Robinson J, Deanfield JE (1994) Aging is associated with endothelial dysfunction in healthy men years before the age-related decline in women. J Am Coll Cardiol 24:471–476
Stenvinkel P (2001) Endothelial dysfunction and inflammation-is there a link? Nephrol Dial Transpl 16:1968–1971
Pasula S, Cai X, Dong Y et al (2012) Endothelial epsin deficiency decreases tumor growth by enhancing VEGF signaling. J Clin Invest 122:4424–4438
Cao Y (2013) Angiogenesis and vascular functions in modulation of obesity, adipose metabolism, and insulin sensitivity. Cell Metab 18:478–489
Deanfield JE, Halcox JP, Rabelink TJ (2007) Endothelial function and dysfunction: testing and clinical relevance. Circulation 115:1285–1295
Xu J, Zou MH (2009) Molecular insights and therapeutic targets for diabetic endothelial dysfunction. Circulation 120:1266–1286
Zhang DX, Gutterman DD (2007) Mitochondrial reactive oxygen species-mediated signaling in endothelial cells. Am J Physiol Heart Circ Physiol 292:H2023–H2031
Cai H (2005) Hydrogen peroxide regulation of endothelial function: origins, mechanisms, and consequences. Cardiovasc Res 68:26–36
Beckman JS, Koppenol WH (1996) Nitric oxide, superoxide, and peroxynitrite: the good, the bad, and ugly. Am J Physiol 271:C1424–C1437
Pacher P, Beckman JS, Liaudet L (2007) Nitric oxide and peroxynitrite in health and disease. Physiol Rev 87:315–424
Zou MH, Shi C, Cohen RA (2002) High glucose via peroxynitrite causes tyrosine nitration and inactivation of prostacyclin synthase that is associated with thromboxane/prostaglandin H(2) receptor-mediated apoptosis and adhesion molecule expression in cultured human aortic endothelial cells. Diabetes 51:198–203
Nie H, Wu JL, Zhang M, Xu J, Zou MH (2006) Endothelial nitric oxide synthase-dependent tyrosine nitration of prostacyclin synthase in diabetes in vivo. Diabetes 55:3133–3141
Wang R (2002) Two’s company, three’s a crowd: can H2S be the third endogenous gaseous transmitter? FASEB J 16:1792–1798
Mustafa AK, Gadalla MM, Snyder SH (2009) Signaling by gasotransmitters. Sci Signal 2:re2
Wang K, Ahmad S, Cai M et al (2013) Dysregulation of hydrogen sulfide producing enzyme cystathionine gamma-lyase contributes to maternal hypertension and placental abnormalities in preeclampsia. Circulation 127:2514–2522
Mani S, Li H, Untereiner A et al (2013) Decreased endogenous production of hydrogen sulfide accelerates atherosclerosis. Circulation 127:2523–2534
Szabo C, Coletta C, Chao C et al (2013) Tumor-derived hydrogen sulfide, produced by cystathionine-beta-synthase, stimulates bioenergetics, cell proliferation, and angiogenesis in colon cancer. Proc Natl Acad Sci USA 110:12474–12479
Kabil O, Banerjee R (2014) Enzymology of H2S biogenesis, decay and signaling. Antioxid Redox Signal 20:770–782
Modis K, Coletta C, Erdelyi K, Papapetropoulos A, Szabo C (2013) Intramitochondrial hydrogen sulfide production by 3-mercaptopyruvate sulfurtransferase maintains mitochondrial electron flow and supports cellular bioenergetics. FASEB J 27:601–611
Yadav PK, Yamada K, Chiku T, Koutmos M, Banerjee R (2013) Structure and kinetic analysis of H2S production by human mercaptopyruvate sulfurtransferase. J Biol Chem 288:20002–20013
Shibuya N, Mikami Y, Kimura Y, Nagahara N, Kimura H (2009) Vascular endothelium expresses 3-mercaptopyruvate sulfurtransferase and produces hydrogen sulfide. J Biochem 146:623–626
Tang C, Li X, Du J (2006) Hydrogen sulfide as a new endogenous gaseous transmitter in the cardiovascular system. Curr Vasc Pharmacol 4:17–22
Yang G, Wu L, Jiang B et al (2008) H2S as a physiologic vasorelaxant: hypertension in mice with deletion of cystathionine gamma-lyase. Science 322:587–590
Yang G, Wu L, Wang R (2006) Pro-apoptotic effect of endogenous H2S on human aorta smooth muscle cells. FASEB J 20:553–555
Abe K, Kimura H (1996) The possible role of hydrogen sulfide as an endogenous neuromodulator. J Neurosci 16:1066–1071
Beltowski J, Jamroz-Wisniewska A (2012) Modulation of h(2)s metabolism by statins: a new aspect of cardiovascular pharmacology. Antioxid Redox Signal 17:81–94
Olson KR (2012) A practical look at the chemistry and biology of hydrogen sulfide. Antioxid Redox Signal 17:32–44
Greiner R, Palinkas Z, Basell K et al (2013) Polysulfides link H2S to protein thiol oxidation. Antioxid Redox Signal 19:1749–1765
Li L, Whiteman M, Guan YY et al (2008) Characterization of a novel, water-soluble hydrogen sulfide-releasing molecule (GYY4137): new insights into the biology of hydrogen sulfide. Circulation 117:2351–2360
Keefe AD, Miller SL, McDonald G, Bada J (1995) Investigation of the prebiotic synthesis of amino acids and RNA bases from CO2 using FeS/H2S as a reducing agent. Proc Natl Acad Sci USA 92:11904–11906
Tyagi N, Moshal KS, Sen U et al (2009) H2S protects against methionine-induced oxidative stress in brain endothelial cells. Antioxid Redox Signal 11:25–33
Kimura H (2014) Production and physiological effects of hydrogen sulfide. Antioxid Redox Signal 20:783–793
Collman JP, Ghosh S, Dey A, Decreau RA (2009) Using a functional enzyme model to understand the chemistry behind hydrogen sulfide induced hibernation. Proc Natl Acad Sci USA 106:22090–22095
Szabo G, Veres G, Radovits T et al (2011) Cardioprotective effects of hydrogen sulfide. Nitric Oxide 25:201–210
Wen YD, Wang H, Kho SH et al (2013) Hydrogen sulfide protects HUVECs against hydrogen peroxide induced mitochondrial dysfunction and oxidative stress. PLoS ONE 8:e53147
Cai WJ, Wang MJ, Moore PK, Jin HM, Yao T, Zhu YC (2007) The novel proangiogenic effect of hydrogen sulfide is dependent on Akt phosphorylation. Cardiovasc Res 76:29–40
Tao BB, Liu SY, Zhang CC et al (2013) VEGFR2 functions as an H2S-targeting receptor protein kinase with its novel Cys 1045-Cys1024 disulfide bond serving as a specific molecular switch for hydrogen sulfide actions in vascular endothelial cells. Antioxid Redox Signal 19:448–464
Coletta C, Papapetropoulos A, Erdelyi K et al (2012) Hydrogen sulfide and nitric oxide are mutually dependent in the regulation of angiogenesis and endothelium-dependent vasorelaxation. Proc Natl Acad Sci USA 109:9161–9166
Wang MJ, Cai WJ, Li N, Ding YJ, Chen Y, Zhu YC (2010) The hydrogen sulfide donor NaHS promotes angiogenesis in a rat model of hind limb ischemia. Antioxid Redox Signal 12:1065–1077
Polhemus DJ, Kondo K, Bhushan S et al (2013) Hydrogen sulfide attenuates cardiac dysfunction after heart failure via induction of angiogenesis. Circ Heart Fail 6:1077–1086
Mustafa AK, Sikka G, Gazi SK et al (2011) Hydrogen sulfide as endothelium-derived hyperpolarizing factor sulfhydrates potassium channels. Circ Res 109:1259–1268
Tang G, Yang G, Jiang B, Ju Y, Wu L, Wang R (2013) H2S is an endothelium-derived hyperpolarizing factor. Antioxid Redox Signal 19:1634–1646
Guan Q, Zhang Y, Yu C, Liu Y, Gao L, Zhao J (2012) Hydrogen sulfide protects against high-glucose-induced apoptosis in endothelial cells. J Cardiovasc Pharmacol 59:188–193
Wang R (2012) Physiological implications of hydrogen sulfide: a whiff exploration that blossomed. Physiol Rev 92:791–896
Perna AF, Sepe I, Lanza D et al (2013) Hydrogen sulfide reduces cell adhesion and relevant inflammatory triggering by preventing ADAM17-dependent TNF-alpha activation. J Cell Biochem 114:1536–1548
Polhemus DJ, Lefer DJ (2014) Emergence of hydrogen sulfide as an endogenous gaseous signaling molecule in cardiovascular disease. Circ Res 114:730–737
Whiteman M, Moore PK (2009) Hydrogen sulfide and the vasculature: a novel vasculoprotective entity and regulator of nitric oxide bioavailability? J Cell Mol Med 13:488–507
King AL, Polhemus DJ, Bhushan S et al (2014) Hydrogen sulfide cytoprotective signaling is endothelial nitric oxide synthase-nitric oxide dependent. Proc Natl Acad Sci USA 111:3182–3187
Paul BD, Snyder SH (2012) H(2)S signalling through protein sulfhydration and beyond. Nat Rev Mol Cell Biol 13:499–507
Mustafa AK, Gadalla MM, Sen N et al (2009) H2S signals through protein S-sulfhydration. Sci Signal 2:ra72
Hourihan JM, Kenna JG, Hayes JD (2013) The gasotransmitter hydrogen sulfide induces nrf2-target genes by inactivating the keap1 ubiquitin ligase substrate adaptor through formation of a disulfide bond between cys-226 and cys-613. Antioxid Redox Signal 19:465–481
Li W, Busu C, Circu ML, Aw TY (2012) Glutathione in cerebral microvascular endothelial biology and pathobiology: implications for brain homeostasis. Int J Cell Biol 2012:434971
Okouchi M, Okayama N, Aw TY (2009) Preservation of cellular glutathione status and mitochondrial membrane potential by N-acetylcysteine and insulin sensitizers prevent carbonyl stress-induced human brain endothelial cell apoptosis. Curr Neurovasc Res 6:267–278
Langston W, Chidlow JH Jr, Booth BA et al (2007) Regulation of endothelial glutathione by ICAM-1 governs VEGF-A-mediated eNOS activity and angiogenesis. Free Radic Biol Med 42:720–729
Kugiyama K, Ohgushi M, Motoyama T et al (1998) Intracoronary infusion of reduced glutathione improves endothelial vasomotor response to acetylcholine in human coronary circulation. Circulation 97:2299–2301
Dalle-Donne I, Rossi R, Giustarini D, Colombo R, Milzani A (2007) S-glutathionylation in protein redox regulation. Free Radic Biol Med 43:883–898
Clavreul N, Adachi T, Pimental DR, Ido Y, Schoneich C, Cohen RA (2006) S-glutathiolation by peroxynitrite of p21ras at cysteine-118 mediates its direct activation and downstream signaling in endothelial cells. FASEB J 20:518–520
Li JM, Shah AM (2004) Endothelial cell superoxide generation: regulation and relevance for cardiovascular pathophysiology. Am J Physiol Regul Integr Comp Physiol 287:R1014–R1030
Du XL, Edelstein D, Rossetti L et al (2000) Hyperglycemia-induced mitochondrial superoxide overproduction activates the hexosamine pathway and induces plasminogen activator inhibitor-1 expression by increasing Sp1 glycosylation. Proc Natl Acad Sci USA 97:12222–12226
Quintero M, Colombo SL, Godfrey A, Moncada S (2006) Mitochondria as signaling organelles in the vascular endothelium. Proc Natl Acad Sci USA 103:5379–5384
Song P, Zou MH (2012) Regulation of NAD(P)H oxidases by AMPK in cardiovascular systems. Free Radic Biol Med 52:1607–1619
Lassegue B, San Martin A, Griendling KK (2012) Biochemistry, physiology, and pathophysiology of NADPH oxidases in the cardiovascular system. Circ Res 110:1364–1390
Zou MH, Shi C, Cohen RA (2002) Oxidation of the zinc-thiolate complex and uncoupling of endothelial nitric oxide synthase by peroxynitrite. J Clin Invest 109:817–826
Landmesser U, Dikalov S, Price SR et al (2003) Oxidation of tetrahydrobiopterin leads to uncoupling of endothelial cell nitric oxide synthase in hypertension. J Clin Invest 111:1201–1209
Thum T, Fraccarollo D, Schultheiss M et al (2007) Endothelial nitric oxide synthase uncoupling impairs endothelial progenitor cell mobilization and function in diabetes. Diabetes 56:666–674
Civelek M, Manduchi E, Riley RJ, Stoeckert CJ Jr, Davies PF (2011) Coronary artery endothelial transcriptome in vivo: identification of endoplasmic reticulum stress and enhanced reactive oxygen species by gene connectivity network analysis. Circ Cardiovasc Genet 4:243–252
Malhotra JD, Kaufman RJ (2007) Endoplasmic reticulum stress and oxidative stress: a vicious cycle or a double-edged sword? Antioxid Redox Signal 9:2277–2293
Landmesser U, Spiekermann S, Preuss C et al (2007) Angiotensin II induces endothelial xanthine oxidase activation: role for endothelial dysfunction in patients with coronary disease. Arterioscler Thromb Vasc Biol 27:943–948
Kou B, Ni J, Vatish M, Singer DR (2008) Xanthine oxidase interaction with vascular endothelial growth factor in human endothelial cell angiogenesis. Microcirculation 15:251–267
Turrens JF (2003) Mitochondrial formation of reactive oxygen species. J Physiol 552:335–344
Maranzana E, Barbero G, Falasca AI, Lenaz G, Genova ML (2013) Mitochondrial respiratory supercomplex association limits production of reactive oxygen species from complex I. Antioxid Redox Signal 19:1469–1480
Liu Y, Zhao H, Li H, Kalyanaraman B, Nicolosi AC, Gutterman DD (2003) Mitochondrial sources of H2O2 generation play a key role in flow-mediated dilation in human coronary resistance arteries. Circ Res 93:573–580
Therade-Matharan S, Laemmel E, Carpentier S et al (2005) Reactive oxygen species production by mitochondria in endothelial cells exposed to reoxygenation after hypoxia and glucose depletion is mediated by ceramide. Am J Physiol Regul Integr Comp Physiol 289:R1756–R1762
Xu J, Xie Z, Reece R, Pimental D, Zou MH (2006) Uncoupling of endothelial nitric oxidase synthase by hypochlorous acid: role of NAD(P)H oxidase-derived superoxide and peroxynitrite. Arterioscler Thromb Vasc Biol 26:2688–2695
Wang S, Xu J, Song P, Viollet B, Zou MH (2009) In vivo activation of AMP-activated protein kinase attenuates diabetes-enhanced degradation of GTP cyclohydrolase I. Diabetes 58:1893–1901
Xu J, Wu Y, Song P, Zhang M, Wang S, Zou MH (2007) Proteasome-dependent degradation of guanosine 5’-triphosphate cyclohydrolase I causes tetrahydrobiopterin deficiency in diabetes mellitus. Circulation 116:944–953
Xu J, Wang S, Zhang M, Wang Q, Asfa S, Zou MH (2012) Tyrosine nitration of PA700 links proteasome activation to endothelial dysfunction in mouse models with cardiovascular risk factors. PLoS ONE 7:e29649
Zhao Y, Wu J, Zhu H, Song P, Zou MH (2013) Peroxynitrite-dependent zinc release and inactivation of guanosine 5’-triphosphate cyclohydrolase 1 instigate its ubiquitination in diabetes. Diabetes 62:4247–4256
Hanschmann EM, Godoy JR, Berndt C, Hudemann C, Lillig CH (2013) Thioredoxins, glutaredoxins, and peroxiredoxins-molecular mechanisms and health significance: from cofactors to antioxidants to redox signaling. Antioxid Redox Signal 19:1539–1605
Stangherlin A, Reddy AB (2013) Regulation of circadian clocks by redox homeostasis. J Biol Chem 288:26505–26511
Ma Q (2013) Role of nrf2 in oxidative stress and toxicity. Annu Rev Pharmacol Toxicol 53:401–426
Oelze M, Kroller-Schon S, Steven S et al (2014) Glutathione peroxidase-1 deficiency potentiates dysregulatory modifications of endothelial nitric oxide synthase and vascular dysfunction in aging. Hypertension 63:390–396
Li L, Rose P, Moore PK (2011) Hydrogen sulfide and cell signaling. Annu Rev Pharmacol Toxicol 51:169–187
Whiteman M, Armstrong JS, Chu SH et al (2004) The novel neuromodulator hydrogen sulfide: an endogenous peroxynitrite ‘scavenger’? J Neurochem 90:765–768
Xie Z, Zhang J, Wu J, Viollet B, Zou MH (2008) Upregulation of mitochondrial uncoupling protein-2 by the AMP-activated protein kinase in endothelial cells attenuates oxidative stress in diabetes. Diabetes 57:3222–3230
Potente M, Urbich C, Sasaki K et al (2005) Involvement of Foxo transcription factors in angiogenesis and postnatal neovascularization. J Clin Invest 115:2382–2392
Shen B, Chao L, Chao J (2010) Pivotal role of JNK-dependent FOXO1 activation in downregulation of kallistatin expression by oxidative stress. Am J Physiol Heart Circ Physiol 298:H1048–H1054
Skurk C, Maatz H, Kim HS et al (2004) The Akt-regulated forkhead transcription factor FOXO3a controls endothelial cell viability through modulation of the caspase-8 inhibitor FLIP. J Biol Chem 279:1513–1525
Ponugoti B, Dong G, Graves DT (2012) Role of forkhead transcription factors in diabetes-induced oxidative stress. Exp Diabetes Res 2012:939751
Tsuchiya K, Tanaka J, Shuiqing Y et al (2012) FoxOs integrate pleiotropic actions of insulin in vascular endothelium to protect mice from atherosclerosis. Cell Metab 15:372–381
Lee JW, Chen H, Pullikotil P, Quon MJ (2011) Protein kinase A-alpha directly phosphorylates FoxO1 in vascular endothelial cells to regulate expression of vascular cellular adhesion molecule-1 mRNA. J Biol Chem 286:6423–6432
Mortuza R, Chen S, Feng B, Sen S, Chakrabarti S (2013) High glucose induced alteration of SIRTs in endothelial cells causes rapid aging in a p300 and FOXO regulated pathway. PLoS ONE 8:e54514
Olmos Y, Valle I, Borniquel S et al (2009) Mutual dependence of Foxo3a and PGC-1alpha in the induction of oxidative stress genes. J Biol Chem 284:14476–14484
Tanaka J, Qiang L, Banks AS et al (2009) Foxo1 links hyperglycemia to LDL oxidation and endothelial nitric oxide synthase dysfunction in vascular endothelial cells. Diabetes 58:2344–2354
Daly C, Wong V, Burova E et al (2004) Angiopoietin-1 modulates endothelial cell function and gene expression via the transcription factor FKHR (FOXO1). Genes Dev 18:1060–1071
Kim HS, Skurk C, Maatz H et al (2005) Akt/FOXO3a signaling modulates the endothelial stress response through regulation of heat shock protein 70 expression. FASEB J 19:1042–1044
Kensler TW, Wakabayashi N, Biswal S (2007) Cell survival responses to environmental stresses via the Keap1-Nrf2-ARE pathway. Annu Rev Pharmacol Toxicol 47:89–116
Cheng X, Siow RC, Mann GE (2011) Impaired redox signaling and antioxidant gene expression in endothelial cells in diabetes: a role for mitochondria and the nuclear factor-E2-related factor 2-Kelch-like ECH-associated protein 1 defense pathway. Antioxid Redox Signal 14:469–487
Kobayashi A, Kang MI, Okawa H et al (2004) Oxidative stress sensor Keap1 functions as an adaptor for Cul3-based E3 ligase to regulate proteasomal degradation of Nrf2. Mol Cell Biol 24:7130–7139
Zhang DD, Lo SC, Cross JV, Templeton DJ, Hannink M (2004) Keap1 is a redox-regulated substrate adaptor protein for a Cul3-dependent ubiquitin ligase complex. Mol Cell Biol 24:10941–10953
Chen XL, Dodd G, Thomas S et al (2006) Activation of Nrf2/ARE pathway protects endothelial cells from oxidant injury and inhibits inflammatory gene expression. Am J Physiol Heart Circ Physiol 290:H1862–H1870
Hsieh HJ, Cheng CC, Wu ST, Chiu JJ, Wung BS, Wang DL (1998) Increase of reactive oxygen species (ROS) in endothelial cells by shear flow and involvement of ROS in shear-induced c-fos expression. J Cell Physiol 175:156–162
Hsieh CY, Hsiao HY, Wu WY et al (2009) Regulation of shear-induced nuclear translocation of the Nrf2 transcription factor in endothelial cells. J Biomed Sci 16:12
Wei Y, Gong J, Thimmulappa RK, Kosmider B, Biswal S, Duh EJ (2013) Nrf2 acts cell-autonomously in endothelium to regulate tip cell formation and vascular branching. Proc Natl Acad Sci USA 110:E3910–E3918
Wang GL, Jiang BH, Rue EA, Semenza GL (1995) Hypoxia-inducible factor 1 is a basic-helix-loop-helix-PAS heterodimer regulated by cellular O2 tension. Proc Natl Acad Sci USA 92:5510–5514
Jiang BH, Rue E, Wang GL, Roe R, Semenza GL (1996) Dimerization, DNA binding, and transactivation properties of hypoxia-inducible factor 1. J Biol Chem 271:17771–17778
Palmer LA, Gaston B, Johns RA (2000) Normoxic stabilization of hypoxia-inducible factor-1 expression and activity: redox-dependent effect of nitrogen oxides. Mol Pharmacol 58:1197–1203
Loboda A, Stachurska A, Florczyk U et al (2009) HIF-1 induction attenuates Nrf2-dependent IL-8 expression in human endothelial cells. Antioxid Redox Signal 11:1501–1517
Chung HS, Wang SB, Venkatraman V, Murray CI, Van Eyk JE (2013) Cysteine oxidative posttranslational modifications: emerging regulation in the cardiovascular system. Circ Res 112:382–392
Cremers CM, Jakob U (2013) Oxidant sensing by reversible disulfide bond formation. J Biol Chem 288:26489–26496
Connor KM, Subbaram S, Regan KJ et al (2005) Mitochondrial H2O2 regulates the angiogenic phenotype via PTEN oxidation. J Biol Chem 280:16916–16924
Lima B, Forrester MT, Hess DT, Stamler JS (2010) S-nitrosylation in cardiovascular signaling. Circ Res 106:633–646
Ravi K, Brennan LA, Levic S, Ross PA, Black SM (2004) S-nitrosylation of endothelial nitric oxide synthase is associated with monomerization and decreased enzyme activity. Proc Natl Acad Sci USA 101:2619–2624
Hill BG, Bhatnagar A (2007) Role of glutathiolation in preservation, restoration and regulation of protein function. IUBMB Life 59:21–26
Mieyal JJ, Gallogly MM, Qanungo S, Sabens EA, Shelton MD (2008) Molecular mechanisms and clinical implications of reversible protein S-glutathionylation. Antioxid Redox Signal 10:1941–1988
Grek CL, Zhang J, Manevich Y, Townsend DM, Tew KD (2013) Causes and consequences of cysteine S-glutathionylation. J Biol Chem 288:26497–26504
Zmijewski JW, Banerjee S, Bae H, Friggeri A, Lazarowski ER, Abraham E (2010) Exposure to hydrogen peroxide induces oxidation and activation of AMP-activated protein kinase. J Biol Chem 285:33154–33164
Evangelista AM, Thompson MD, Weisbrod RM et al (2012) Redox regulation of SERCA2 is required for vascular endothelial growth factor-induced signaling and endothelial cell migration. Antioxid Redox Signal 17:1099–1108
Evangelista AM, Thompson MD, Bolotina VM, Tong X, Cohen RA (2012) Nox4- and Nox2-dependent oxidant production is required for VEGF-induced SERCA cysteine-674 S-glutathiolation and endothelial cell migration. Free Radic Biol Med 53:2327–2334
Clavreul N, Bachschmid MM, Hou X et al (2006) S-glutathiolation of p21ras by peroxynitrite mediates endothelial insulin resistance caused by oxidized low-density lipoprotein. Arterioscler Thromb Vasc Biol 26:2454–2461
Chen CA, Wang TY, Varadharaj S et al (2010) S-glutathionylation uncouples eNOS and regulates its cellular and vascular function. Nature 468:1115–1118
Wang Y, Yang J, Yi J (2012) Redox sensing by proteins: oxidative modifications on cysteines and the consequent events. Antioxid Redox Signal 16:649–657
Wang K, Zhang T, Dong Q, Nice EC, Huang C, Wei Y (2013) Redox homeostasis: the linchpin in stem cell self-renewal and differentiation. Cell Death Dis 4:e537
Zhang J, Xie Z, Dong Y, Wang S, Liu C, Zou MH (2008) Identification of nitric oxide as an endogenous activator of the AMP-activated protein kinase in vascular endothelial cells. J Biol Chem 283:27452–27461
Han Y, Wang Q, Song P, Zhu Y, Zou MH (2010) Redox regulation of the AMP-activated protein kinase. PLoS ONE 5:e15420
Song P, Wang S, He C, Liang B, Viollet B, Zou MH (2011) AMPKalpha2 deletion exacerbates neointima formation by upregulating Skp2 in vascular smooth muscle cells. Circ Res 109:1230–1239
Song P, Zhou Y, Coughlan KA et al (2013) Adenosine monophosphate-activated protein kinase-alpha2 deficiency promotes vascular smooth muscle cell migration via S-phase kinase-associated protein 2 upregulation and E-cadherin downregulation. Arterioscler Thromb Vasc Biol 33:2800–2809
Wang S, Song P, Zou MH (2012) AMP-activated protein kinase, stress responses and cardiovascular diseases. Clin Sci (Lond) 122:555–573
Zou MH, Hou XY, Shi CM, Nagata D, Walsh K, Cohen RA (2002) Modulation by peroxynitrite of Akt- and AMP-activated kinase-dependent Ser1179 phosphorylation of endothelial nitric oxide synthase. J Biol Chem 277:32552–32557
Song P, Wu Y, Xu J et al (2007) Reactive nitrogen species induced by hyperglycemia suppresses Akt signaling and triggers apoptosis by upregulating phosphatase PTEN (phosphatase and tensin homologue deleted on chromosome 10) in an LKB1-dependent manner. Circulation 116:1585–1595
Song P, Xie Z, Wu Y, Xu J, Dong Y, Zou MH (2008) Protein kinase Czeta-dependent LKB1 serine 428 phosphorylation increases LKB1 nucleus export and apoptosis in endothelial cells. J Biol Chem 283:12446–12455
Wang Q, Zhang M, Ding Y et al (2014) Activation of NAD(P)H oxidase by tryptophan-derived 3-hydroxykynurenine accelerates endothelial apoptosis and dysfunction in vivo. Circ Res 114:480–492
Ido Y, Carling D, Ruderman N (2002) Hyperglycemia-induced apoptosis in human umbilical vein endothelial cells: inhibition by the AMP-activated protein kinase activation. Diabetes 51:159–167
Zhang M, Dong Y, Xu J et al (2008) Thromboxane receptor activates the AMP-activated protein kinase in vascular smooth muscle cells via hydrogen peroxide. Circ Res 102:328–337
Zou MH, Kirkpatrick SS, Davis BJ et al (2004) Activation of the AMP-activated protein kinase by the anti-diabetic drug metformin in vivo. Role of mitochondrial reactive nitrogen species. J Biol Chem 279:43940–43951
Zou MH, Hou XY, Shi CM et al (2003) Activation of 5’-AMP-activated kinase is mediated through c-Src and phosphoinositide 3-kinase activity during hypoxia-reoxygenation of bovine aortic endothelial cells. Role of peroxynitrite. J Biol Chem 278:34003–34010
Choi HC, Song P, Xie Z et al (2008) Reactive nitrogen species is required for the activation of the AMP-activated protein kinase by statin in vivo. J Biol Chem 283:20186–20197
Liu C, Liang B, Wang Q, Wu J, Zou MH (2010) Activation of AMP-activated protein kinase alpha1 alleviates endothelial cell apoptosis by increasing the expression of anti-apoptotic proteins Bcl-2 and survivin. J Biol Chem 285:15346–15355
Colombo SL, Moncada S (2009) AMPKalpha1 regulates the antioxidant status of vascular endothelial cells. Biochem J 421:163–169
Bhatt MP, Lim YC, Kim YM, Ha KS (2013) C-peptide activates AMPKalpha and prevents ROS-mediated mitochondrial fission and endothelial apoptosis in diabetes. Diabetes 62:3851–3862
Cifarelli V, Lee S, Kim DH et al (2012) FOXO1 mediates the autocrine effect of endothelin-1 on endothelial cell survival. Mol Endocrinol 26:1213–1224
Burhans WC, Heintz NH (2009) The cell cycle is a redox cycle: linking phase-specific targets to cell fate. Free Radic Biol Med 47:1282–1293
Sarsour EH, Kumar MG, Chaudhuri L, Kalen AL, Goswami PC (2009) Redox control of the cell cycle in health and disease. Antioxid Redox Signal 11:2985–3011
Menon SG, Goswami PC (2007) A redox cycle within the cell cycle: ring in the old with the new. Oncogene 26:1101–1109
Latella L, Sacco A, Pajalunga D et al (2001) Reconstitution of cyclin D1-associated kinase activity drives terminally differentiated cells into the cell cycle. Mol Cell Biol 21:5631–5643
Ushio-Fukai M (2006) Redox signaling in angiogenesis: role of NADPH oxidase. Cardiovasc Res 71:226–235
Wang Y, Zang QS, Liu Z et al (2011) Regulation of VEGF-induced endothelial cell migration by mitochondrial reactive oxygen species. Am J Physiol Cell Physiol 301:C695–C704
Ashton AW, Ware JA (2004) Thromboxane A2 receptor signaling inhibits vascular endothelial growth factor-induced endothelial cell differentiation and migration. Circ Res 95:372–379
Song P, Zhang M, Wang S, Xu J, Choi HC, Zou MH (2009) Thromboxane A2 receptor activates a rho-associated kinase/LKB1/PTEN pathway to attenuate endothelium insulin signaling. J Biol Chem 284:17120–17128
Xu MJ, Song P, Shirwany N et al (2011) Impaired expression of uncoupling protein 2 causes defective postischemic angiogenesis in mice deficient in AMP-activated protein kinase alpha subunits. Arterioscler Thromb Vasc Biol 31:1757–1765
Zippel N, Malik RA, Fromel T et al (2013) Transforming growth factor-beta-activated kinase 1 regulates angiogenesis via AMP-activated protein kinase-alpha1 and redox balance in endothelial cells. Arterioscler Thromb Vasc Biol 33:2792–2799
Bakkenist CJ, Kastan MB (2003) DNA damage activates ATM through intermolecular autophosphorylation and dimer dissociation. Nature 421:499–506
Okuno Y, Nakamura-Ishizu A, Otsu K, Suda T, Kubota Y (2012) Pathological neoangiogenesis depends on oxidative stress regulation by ATM. Nat Med 18:1208–1216
Jansen F, Yang X, Hoelscher M et al (2013) Endothelial microparticle-mediated transfer of MicroRna-126 promotes vascular endothelial cell repair via SPRED1 and is abrogated in glucose-damaged endothelial microparticles. Circulation 128:2026–2038
Papapetropoulos A, Pyriochou A, Altaany Z et al (2009) Hydrogen sulfide is an endogenous stimulator of angiogenesis. Proc Natl Acad Sci USA 106:21972–21977
Cacicedo JM, Yagihashi N, Keaney JF Jr, Ruderman NB, Ido Y (2004) AMPK inhibits fatty acid-induced increases in NF-kappaB transactivation in cultured human umbilical vein endothelial cells. Biochem Biophys Res Commun 324:1204–1209
Hattori Y, Nakano Y, Hattori S, Tomizawa A, Inukai K, Kasai K (2008) High molecular weight adiponectin activates AMPK and suppresses cytokine-induced NF-kappaB activation in vascular endothelial cells. FEBS Lett 582:1719–1724
Bess E, Fisslthaler B, Fromel T, Fleming I (2011) Nitric oxide-induced activation of the AMP-activated protein kinase alpha2 subunit attenuates IkappaB kinase activity and inflammatory responses in endothelial cells. PLoS ONE 6:e20848
Delhase M, Hayakawa M, Chen Y, Karin M (1999) Positive and negative regulation of IkappaB kinase activity through IKKbeta subunit phosphorylation. Science 284:309–313
Seldon MP, Silva G, Pejanovic N et al (2007) Heme oxygenase-1 inhibits the expression of adhesion molecules associated with endothelial cell activation via inhibition of NF-kappaB RelA phosphorylation at serine 276. J Immunol 179:7840–7851
Xiao H, Lu M, Lin TY et al (2013) Sterol regulatory element binding protein 2 activation of NLRP3 inflammasome in endothelium mediates hemodynamic-induced atherosclerosis susceptibility. Circulation 128:632–642
Zhou R, Tardivel A, Thorens B, Choi I, Tschopp J (2010) Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat Immunol 11:136–140
Dostert C, Petrilli V, Van Bruggen R, Steele C, Mossman BT, Tschopp J (2008) Innate immune activation through Nalp3 inflammasome sensing of asbestos and silica. Science 320:674–677
Pan LL, Liu XH, Gong QH, Wu D, Zhu YZ (2011) Hydrogen sulfide attenuated tumor necrosis factor-alpha-induced inflammatory signaling and dysfunction in vascular endothelial cells. PLoS ONE 6:e19766
van der Loo B, Schildknecht S, Zee R, Bachschmid MM (2009) Signalling processes in endothelial ageing in relation to chronic oxidative stress and their potential therapeutic implications in humans. Exp Physiol 94:305–310
Burger D, Kwart DG, Montezano AC et al (2012) Microparticles induce cell cycle arrest through redox-sensitive processes in endothelial cells: implications in vascular senescence. J Am Heart Assoc 1:e001842
Levenberg S, Golub JS, Amit M, Itskovitz-Eldor J, Langer R (2002) Endothelial cells derived from human embryonic stem cells. Proc Natl Acad Sci USA 99:4391–4396
Wang ZZ, Au P, Chen T et al (2007) Endothelial cells derived from human embryonic stem cells form durable blood vessels in vivo. Nat Biotechnol 25:317–318
Samuel R, Daheron L, Liao S et al (2013) Generation of functionally competent and durable engineered blood vessels from human induced pluripotent stem cells. Proc Natl Acad Sci USA 110:12774–12779
Margariti A, Winkler B, Karamariti E et al (2012) Direct reprogramming of fibroblasts into endothelial cells capable of angiogenesis and reendothelialization in tissue-engineered vessels. Proc Natl Acad Sci USA 109:13793–13798
Junker JP, Lonnqvist S, Rakar J, Karlsson LK, Grenegard M, Kratz G (2013) Differentiation of human dermal fibroblasts towards endothelial cells. Differentiation 85:67–77
Oswald J, Boxberger S, Jorgensen B et al (2004) Mesenchymal stem cells can be differentiated into endothelial cells in vitro. Stem Cells 22:377–384
Zhang P, Moudgill N, Hager E et al (2011) Endothelial differentiation of adipose-derived stem cells from elderly patients with cardiovascular disease. Stem Cells Dev 20:977–988
Fischer LJ, McIlhenny S, Tulenko T et al (2009) Endothelial differentiation of adipose-derived stem cells: effects of endothelial cell growth supplement and shear force. J Surg Res 152:157–166
Planat-Benard V, Silvestre JS, Cousin B et al (2004) Plasticity of human adipose lineage cells toward endothelial cells: physiological and therapeutic perspectives. Circulation 109:656–663
Soda Y, Marumoto T, Friedmann-Morvinski D et al (2011) Transdifferentiation of glioblastoma cells into vascular endothelial cells. Proc Natl Acad Sci USA 108:4274–4280
Zeng L, Xiao Q, Margariti A et al (2006) HDAC3 is crucial in shear- and VEGF-induced stem cell differentiation toward endothelial cells. J Cell Biol 174:1059–1069
Yamamoto K, Sokabe T, Watabe T et al (2005) Fluid shear stress induces differentiation of Flk-1-positive embryonic stem cells into vascular endothelial cells in vitro. Am J Physiol Heart Circ Physiol 288:H1915–H1924
Zhang C, Zeng L, Emanueli C, Xu Q (2013) Blood flow and stem cells in vascular disease. Cardiovasc Res 99:251–259
Marcelo KL, Goldie LC, Hirschi KK (2013) Regulation of endothelial cell differentiation and specification. Circ Res 112:1272–1287
Myers CT, Krieg PA (2013) BMP-mediated specification of the erythroid lineage suppresses endothelial development in blood island precursors. Blood 122:3929–3939
Wang S, Dale GL, Song P, Viollet B, Zou MH (2010) AMPKalpha1 deletion shortens erythrocyte life span in mice: role of oxidative stress. J Biol Chem 285:19976–19985
Arciniegas E, Sutton AB, Allen TD, Schor AM (1992) Transforming growth factor beta 1 promotes the differentiation of endothelial cells into smooth muscle-like cells in vitro. J Cell Sci 103(Pt 2):521–529
DeRuiter MC, Poelmann RE, VanMunsteren JC, Mironov V, Markwald RR, Gittenberger-de Groot AC (1997) Embryonic endothelial cells transdifferentiate into mesenchymal cells expressing smooth muscle actins in vivo and in vitro. Circ Res 80:444–451
Banerji S, Ni J, Wang SX et al (1999) LYVE-1, a new homologue of the CD44 glycoprotein, is a lymph-specific receptor for hyaluronan. J Cell Biol 144:789–801
Hong YK, Harvey N, Noh YH et al (2002) Prox1 is a master control gene in the program specifying lymphatic endothelial cell fate. Dev Dyn 225:351–357
Adams RH, Alitalo K (2007) Molecular regulation of angiogenesis and lymphangiogenesis. Nat Rev Mol Cell Biol 8:464–478
Cooley LS, Handsley MM, Zhou Z et al (2010) Reversible transdifferentiation of blood vascular endothelial cells to a lymphatic-like phenotype in vitro. J Cell Sci 123:3808–3816
Medici D, Shore EM, Lounev VY, Kaplan FS, Kalluri R, Olsen BR (2010) Conversion of vascular endothelial cells into multipotent stem-like cells. Nat Med 16:1400–1406
Goumans MJ, van Zonneveld AJ, ten Dijke P (2008) Transforming growth factor beta-induced endothelial-to-mesenchymal transition: a switch to cardiac fibrosis? Trends Cardiovasc Med 18:293–298
Gupta RK, Mepani RJ, Kleiner S et al (2012) Zfp423 expression identifies committed preadipocytes and localizes to adipose endothelial and perivascular cells. Cell Metab 15:230–239
Sena CM, Pereira AM, Seica R (2013) Endothelial dysfunction—a major mediator of diabetic vascular disease. Biochim Biophys Acta 1832:2216–2231
Hopkins PN (2013) Molecular biology of atherosclerosis. Physiol Rev 93:1317–1542
Sukumar P, Viswambharan H, Imrie H et al (2013) Nox2 NADPH oxidase has a critical role in insulin resistance-related endothelial cell dysfunction. Diabetes 62:2130–2134
Gray SP, Di Marco E, Okabe J et al (2013) NADPH oxidase 1 plays a key role in diabetes mellitus-accelerated atherosclerosis. Circulation 127:1888–1902
Wang S, Zhang M, Liang B et al (2010) AMPKalpha2 deletion causes aberrant expression and activation of NAD(P)H oxidase and consequent endothelial dysfunction in vivo: role of 26S proteasomes. Circ Res 106:1117–1128
Dong Y, Zhang M, Wang S et al (2010) Activation of AMP-activated protein kinase inhibits oxidized LDL-triggered endoplasmic reticulum stress in vivo. Diabetes 59:1386–1396
Zhang M, Song P, Xu J, Zou MH (2011) Activation of NAD(P)H oxidases by thromboxane A2 receptor uncouples endothelial nitric oxide synthase. Arterioscler Thromb Vasc Biol 31:125–132
Mani S, Untereiner A, Wu L, Wang R (2014) Hydrogen sulfide and the pathogenesis of atherosclerosis. Antioxid Redox Signal 20:805–817
Wang Y, Zhao X, Jin H et al (2009) Role of hydrogen sulfide in the development of atherosclerotic lesions in apolipoprotein E knockout mice. Arterioscler Thromb Vasc Biol 29:173–179
Wang S, Xu J, Song P et al (2008) Acute inhibition of guanosine triphosphate cyclohydrolase 1 uncouples endothelial nitric oxide synthase and elevates blood pressure. Hypertension 52:484–490
Xu J, Wang S, Wu Y, Song P, Zou MH (2009) Tyrosine nitration of PA700 activates the 26S proteasome to induce endothelial dysfunction in mice with angiotensin II-induced hypertension. Hypertension 54:625–632
Paravicini TM, Touyz RM (2008) NADPH oxidases, reactive oxygen species, and hypertension: clinical implications and therapeutic possibilities. Diabetes Care 31(Suppl 2):S170–S180
Kris-Etherton PM, Harris WS, Appel LJ, American Heart Association, Nutrition C (2002) Fish consumption, fish oil, omega-3 fatty acids, and cardiovascular disease. Circulation 106:2747–2757
Wu Y, Zhang C, Dong Y et al (2012) Activation of the AMP-activated protein kinase by eicosapentaenoic acid (EPA, 20:5 n-3) improves endothelial function in vivo. PLoS ONE 7:e35508
Zhao W, Zhang J, Lu Y, Wang R (2001) The vasorelaxant effect of H(2)S as a novel endogenous gaseous K(ATP) channel opener. EMBO J 20:6008–6016
Yanfei W, Lin S, Junbao D, Chaoshu T (2006) Impact of l-arginine on hydrogen sulfide/cystathionine-gamma-lyase pathway in rats with high blood flow-induced pulmonary hypertension. Biochem Biophys Res Commun 345:851–857
Yan H, Du J, Tang C (2004) The possible role of hydrogen sulfide on the pathogenesis of spontaneous hypertension in rats. Biochem Biophys Res Commun 313:22–27
Peter EA, Shen X, Shah SH et al (2013) Plasma free H2S levels are elevated in patients with cardiovascular disease. J Am Heart Assoc 2:e000387
Albini A, Tosetti F, Li VW, Noonan DM, Li WW (2012) Cancer prevention by targeting angiogenesis. Nat Rev Clin Oncol 9:498–509
Reymond N, d’Agua BB, Ridley AJ (2013) Crossing the endothelial barrier during metastasis. Nat Rev Cancer 13:858–870
Xia C, Meng Q, Liu LZ, Rojanasakul Y, Wang XR, Jiang BH (2007) Reactive oxygen species regulate angiogenesis and tumor growth through vascular endothelial growth factor. Cancer Res 67:10823–10830
Parri M, Chiarugi P (2013) Redox molecular machines involved in tumor progression. Antioxid Redox Signal 19:1828–1845
West XZ, Malinin NL, Merkulova AA et al (2010) Oxidative stress induces angiogenesis by activating TLR2 with novel endogenous ligands. Nature 467:972–976
Daquinag AC, Zhang Y, Kolonin MG (2011) Vascular targeting of adipose tissue as an anti-obesity approach. Trends Pharmacol Sci 32:300–307
Cao Y (2010) Adipose tissue angiogenesis as a therapeutic target for obesity and metabolic diseases. Nat Rev Drug Discov 9:107–115
Kim CS, Park HS, Kawada T et al (2006) Circulating levels of MCP-1 and IL-8 are elevated in human obese subjects and associated with obesity-related parameters. Int J Obes (Lond) 30:1347–1355
Kim SJ, Moon GJ, Cho YH et al (2012) Circulating mesenchymal stem cells microparticles in patients with cerebrovascular disease. PLoS ONE 7:e37036
Roufosse CA, Direkze NC, Otto WR, Wright NA (2004) Circulating mesenchymal stem cells. Int J Biochem Cell Biol 36:585–597
Cao Y (2007) Angiogenesis modulates adipogenesis and obesity. J Clin Invest 117:2362–2368
Villaret A, Galitzky J, Decaunes P et al (2010) Adipose tissue endothelial cells from obese human subjects: differences among depots in angiogenic, metabolic, and inflammatory gene expression and cellular senescence. Diabetes 59:2755–2763
Gealekman O, Guseva N, Hartigan C et al (2011) Depot-specific differences and insufficient subcutaneous adipose tissue angiogenesis in human obesity. Circulation 123:186–194
Brakenhielm E, Cao R, Gao B et al (2004) Angiogenesis inhibitor, TNP-470, prevents diet-induced and genetic obesity in mice. Circ Res 94:1579–1588
Rupnick MA, Panigrahy D, Zhang CY et al (2002) Adipose tissue mass can be regulated through the vasculature. Proc Natl Acad Sci USA 99:10730–10735
Tam J, Duda DG, Perentes JY, Quadri RS, Fukumura D, Jain RK (2009) Blockade of VEGFR2 and not VEGFR1 can limit diet-induced fat tissue expansion: role of local versus bone marrow-derived endothelial cells. PLoS ONE 4:e4974
Lim S, Honek J, Xue Y et al (2012) Cold-induced activation of brown adipose tissue and adipose angiogenesis in mice. Nat Protoc 7:606–615
Xue Y, Lim S, Brakenhielm E, Cao Y (2010) Adipose angiogenesis: quantitative methods to study microvessel growth, regression and remodeling in vivo. Nat Protoc 5:912–920
Bartelt A, Heeren J (2014) Adipose tissue browning and metabolic health. Nat Rev Endocrinol 10:24–36
Cheng X, Chapple SJ, Patel B et al (2013) Gestational diabetes mellitus impairs Nrf2-mediated adaptive antioxidant defenses and redox signaling in fetal endothelial cells in utero. Diabetes 62:4088–4097
Sobngwi E, Boudou P, Mauvais-Jarvis F et al (2003) Effect of a diabetic environment in utero on predisposition to type 2 diabetes. Lancet 361:1861–1865
Duplain H, Burcelin R, Sartori C et al (2001) Insulin resistance, hyperlipidemia, and hypertension in mice lacking endothelial nitric oxide synthase. Circulation 104:342–345
Le Gouill E, Jimenez M, Binnert C et al (2007) Endothelial nitric oxide synthase (eNOS) knockout mice have defective mitochondrial beta-oxidation. Diabetes 56:2690–2696
Sansbury BE, Cummins TD, Tang Y et al (2012) Overexpression of endothelial nitric oxide synthase prevents diet-induced obesity and regulates adipocyte phenotype. Circ Res 111:1176–1189
Wang H, Wang AX, Aylor K, Barrett EJ (2013) Nitric oxide directly promotes vascular endothelial insulin transport. Diabetes 62:4030–4042
Nelson ER, Wardell SE, Jasper JS et al (2013) 27-Hydroxycholesterol links hypercholesterolemia and breast cancer pathophysiology. Science 342:1094–1098
Lijnen HR (2008) Angiogenesis and obesity. Cardiovasc Res 78:286–293
Feldmann HM, Golozoubova V, Cannon B, Nedergaard J (2009) UCP1 ablation induces obesity and abolishes diet-induced thermogenesis in mice exempt from thermal stress by living at thermoneutrality. Cell Metab 9:203–209
Xue Y, Petrovic N, Cao R et al (2009) Hypoxia-independent angiogenesis in adipose tissues during cold acclimation. Cell Metab 9:99–109
Wang J, Liu R, Wang F et al (2013) Ablation of LGR4 promotes energy expenditure by driving white-to-brown fat switch. Nat Cell Biol 15:1455–1463
Baker PR, Lin Y, Schopfer FJ et al (2005) Fatty acid transduction of nitric oxide signaling: multiple nitrated unsaturated fatty acid derivatives exist in human blood and urine and serve as endogenous peroxisome proliferator-activated receptor ligands. J Biol Chem 280:42464–42475
Lima ES, Bonini MG, Augusto O, Barbeiro HV, Souza HP, Abdalla DS (2005) Nitrated lipids decompose to nitric oxide and lipid radicals and cause vasorelaxation. Free Radic Biol Med 39:532–539
Trostchansky A, Bonilla L, Gonzalez-Perilli L, Rubbo H (2013) Nitro-fatty acids: formation, redox signaling, and therapeutic potential. Antioxid Redox Signal 19:1257–1265
Wu Y, Dong Y, Song P, Zou MH (2012) Activation of the AMP-activated protein kinase (AMPK) by nitrated lipids in endothelial cells. PLoS ONE 7:e31056
Streeter E, Ng HH, Hart JL (2013) Hydrogen sulfide as a vasculoprotective factor. Med Gas Res 3:9
Kashfi K (2014) Anti-cancer activity of new designer hydrogen sulfide-donating hybrids. Antioxid Redox Signal 20:831–846
Predmore BL, Lefer DJ (2010) Development of hydrogen sulfide-based therapeutics for cardiovascular disease. J Cardiovasc Transl Res 3:487–498
Benavides GA, Squadrito GL, Mills RW et al (2007) Hydrogen sulfide mediates the vasoactivity of garlic. Proc Natl Acad Sci USA 104:17977–17982
Ried K, Frank OR, Stocks NP (2013) Aged garlic extract reduces blood pressure in hypertensives: a dose-response trial. Eur J Clin Nutr 67:64–70
Huang YT, Yao CH, Way CL et al (2013) Diallyl trisulfide and diallyl disulfide ameliorate cardiac dysfunction by suppressing apoptotic and enhancing survival pathways in experimental diabetic rats. J Appl Physiol 114:402–410
Schramm A, Matusik P, Osmenda G, Guzik TJ (2012) Targeting NADPH oxidases in vascular pharmacology. Vasc Pharmacol 56:216–231
Cayatte AJ, Rupin A, Oliver-Krasinski J et al (2001) S17834, a new inhibitor of cell adhesion and atherosclerosis that targets NADPH oxidase. Arterioscler Thromb Vasc Biol 21:1577–1584
Xu S, Jiang B, Hou X et al (2011) High-fat diet increases and the polyphenol, S17834, decreases acetylation of the sirtuin-1-dependent lysine-382 on p53 and apoptotic signaling in atherosclerotic lesion-prone aortic endothelium of normal mice. J Cardiovasc Pharmacol 58:263–271
Yang H, Roberts LJ, Shi MJ et al (2004) Retardation of atherosclerosis by overexpression of catalase or both Cu/Zn-superoxide dismutase and catalase in mice lacking apolipoprotein E. Circ Res 95:1075–1081
Van Assche T, Huygelen V, Crabtree MJ (2011) Targeting vascular redox biology through antioxidant gene delivery: a historical view and current perspectives. Recent Pat Cardiovasc Drug Discovery 6:89–102
Levonen AL, Inkala M, Heikura T et al (2007) Nrf2 gene transfer induces antioxidant enzymes and suppresses smooth muscle cell growth in vitro and reduces oxidative stress in rabbit aorta in vivo. Arterioscler Thromb Vasc Biol 27:741–747
Shuvaev VV, Ilies MA, Simone E et al (2011) Endothelial targeting of antibody-decorated polymeric filomicelles. ACS Nano 5:6991–6999
Zern BJ, Chacko AM, Liu J et al (2013) Reduction of nanoparticle avidity enhances the selectivity of vascular targeting and PET detection of pulmonary inflammation. ACS Nano 7:2461–2469
Pan H, Myerson JW, Hu L et al (2013) Programmable nanoparticle functionalization for in vivo targeting. FASEB J 27:255–264
Hood E, Simone E, Wattamwar P, Dziubla T, Muzykantov V (2011) Nanocarriers for vascular delivery of antioxidants. Nanomedicine 6:1257–1272
Acknowledgments
The authors sincerely apologize to those colleagues whose original work was not cited due to limited space. The authors also thank all current and former members of Dr. Zou’s laboratory for the work described in this review. This study was supported by funding from the following agencies: National Institutes of Health RO1 (HL110488, HL105157, HL096032, HL089920, HL080499, HL079584, and HL074399), the Warren Chair in Diabetes Research of the University of Oklahoma Health Sciences Center (all to Dr. Zou), Scientist Development Grant (11SDG5560036) from National Center of American Heart Association, and Oklahoma Center for the Advancement of Science and Technology (HR12-061) (both to Dr. Song). Dr. Zou is a recipient of the National Established Investigator Award of the American Heart Association.
Conflict of interest
The authors declare that there are no conflicts of interest.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Song, P., Zou, MH. Redox regulation of endothelial cell fate. Cell. Mol. Life Sci. 71, 3219–3239 (2014). https://doi.org/10.1007/s00018-014-1598-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00018-014-1598-z